Team:BCCS-Bristol/Project
From 2009.igem.org
iGEM 2009
Contents |
Outer Membrane Vesicles
We are attempting to express (non-toxic!) heterologus proteins in bacterial outer membrane vesicles for the purposes of 'digital' communication, swarm immunity and pattern formation:
- Special delivery: vesicle trafficking in prokaryotes (image credit)
- Engineered Bacterial Outer Membrane Vesicles with Enhanced Functionality
Magnetotaxis
Inspired by papers on controlled manipulation and actuation of micro-objects with magnetotactic bacteria, we are also looking into magnetotaxis via attatchment of bacteria to external magnetic particles:
- Effect of quantity and configuration of attached bacteria on bacterial propulsion of microbeads (image credit)
Proposal
Steve 2nd draft
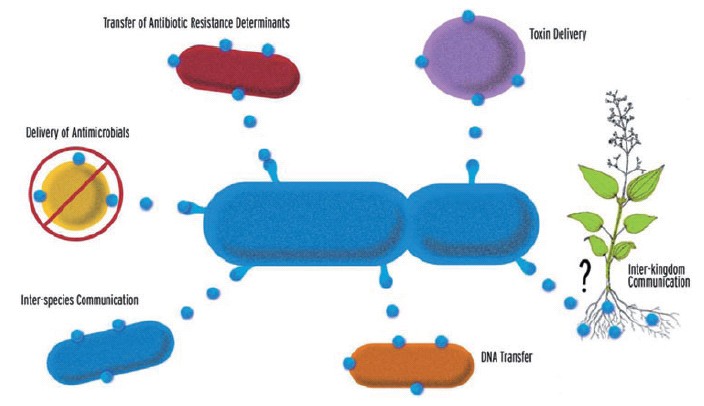
Gram-negative bacteria naturally produce outer member vesicles (OMVs): spherical, bilayered proteolipids with an average diameter of 20-200nm. OMVs can carry outer membrane, periplasmic and cytoplasmic proteins, DNA, RNA and other factors associated with virulence. They have been implicated in the delivery of toxins to host cells, in the transfer of proteins and genetic materials between bacterial cells and in cell-to-cell signalling [1].
Recently it was demonstrated that chimeric proteins formed by a fusion to the vesicle-associated toxin ClyA are exported to OMVs, and that fusion partners can retain their activity in the OMVs [2]. Protein secretion via OMVs offers several distinct engineering opportunities not present when employing traditional recombinant secretory mechanisms. Firstly, vesicle fusion with the cell membrane offers a way of delivering proteins into other cells; P. aeruginosa uses OMVs to transfer antibiotic resistance proteins from one bacterial cell to another, whilst several strains of pathogenic bacteria are capable of delivering toxins to eukaryotic target cells via OMVs [3]. Secondly, OMVs offer the possibility of delivering localised concentrations of several different proteins simultaneously; OMVs from some pathogenic bacteria contain a potent cocktail of virulence factors including toxins, adhesins and invasins [1]. Thirdly, the ease with which it is possible to isolate OMVs from the supernatant via vacuum filtration suggests a new method of harvesting proteins.
Elsewhere, external magnetic fields have been used for fine control over the movement of naturally magnetotactic bacteria, offering an alternative to directed movement via chemical gradients [4]. However, understanding of magnetosome synthesis is still rudimentary, blocking efforts to engineer magnetotaxis in vivo [5].
In this project we aim to construct a system for directed delivery of proteins into cells by combining ex vivo magnetotaxis with OMV protein secretion. Bacteria will preferentially attach to one hemisphere of a half-coated magnetic bead (through the usage of lock-and-key mechanisms e.g. streptavidin-biotin), exerting a propulsion force. The orientation of the bead will be controlled by a uniform external magnetic field so that the bead is steered towards a target cell population. Upon sensing the target cells, vesicle budding will commence, delivering a protein or cocktail of proteins. A toxic cocktail may be appropriate in the case of a cancerous population, alternatively, the cocktail may contain proteins to replace those missing as a result of genetic defect. In the first instance, we shall seek to transfer antibiotic resistance proteins in OMVs from the magnetotactic bacteria to cells otherwise lacking the means to defend themselves against the antibiotic. If this proves successful we hope to study OMV interactions with simple eukaryotic organisms.
As part of the project we will design Biobricks allowing the secretion of any protein in OMVs via fusion with novel, non-toxic partners known to be enhanced in OMVs. We hope that the Biobricks will become widely used as a simple and effective means of protein secretion, whilst also conveying the unique engineering advantages of vesicular encapsulation.
The mechanisms of magnetotaxis and OMV secretion will be implemented into BSim, the award-winning agent-based modelling framework developed by last year’s Bristol team, for the purposes of informing lab work and testing speculative ideas. A key new feature will allow users to ‘plug in’ SBML models and see their effect at the population level [6]. We will use this feature to simulate the processes of vesicle production and environmental sensing in biochemical detail whilst not sacrificing BSim’s unique ability to draw inferences at the population level.
1. Global proteomic profiling of native outer membrane vesicles derived from Escherichia coli, DOI 10.1002/pmic.200700196
2. Engineered Bacterial Outer Membrane Vesicles with Enhanced Functionality, doi:10.1016/j.jmb.2008.03.076
3. Special delivery: vesicle trafficking in prokaryotes, doi:10.1111/j.1365-2958.2006.05272.x
4. Controlled manipulation and actuation of micro-objects with magnetotactic bacteria, APPLIED PHYSICS LETTERS 89, 233904 2006
5. Bazylinski A.D. and Frankel R.B. (2004), Magnetosome formation in prokaryotes, Nat. Rev. Microb. 2, 217-230
6. CellDesigner 3.5: A Versatile Modeling Toolfor Biochemical Networks, IEEE Vol. 96, No. 8, August 2008
Petros 1st draft
Knowledge and arguments regarding what we are doing
All biologically important chemistry occurs in the nano-scale level. This ranges from receptor-ligand interactions that occur between cells and their environment to protein-protein chemistry occurring intracellularly. The implications of this is that research efforts need to concentrate on the production and development of nano-sized particles with the correct stereochemistry, with prime examples being [1] and [2].
The development of nano-sized particles with correct stereochemistry is currently limited by current organic chemistry and rational engineering of organic molecules has been employed by harnessing the chemical abilities of living organisms like in the synthesis of polyketide antibiotics [3]. A major obstacle that currently limits such techniques, but is not limited to, is the harvesting of such nano-scale chemicals or their direct application from the synthesizing organism to the target location. Problems include the diffusive nature of chemicals and their subsequent exposure to the environmental degradation, thus limiting the communication capabilities in the dimensions of time and distance.
This also places limits on the ability of controlling or forming chemotactic gradients. However some organisms can be manipulated not only by chemical gradients but by physical forces, in particular they show preference in orientation towards the earth’s magnetic field, a property called magnetotaxis [4]. The application of physical forces provides potentially greater control over chemical gradients.
With all these issues in mind we aim to implement in our project the increased survival of nanosized chemicals, thus increasing the communication capabilities at least in the dimension of time and possibly distance, by encapsulating the chemicals within or on the surface of small lipid bilayers (vesicles). We also aim to implement the control over actuation of microobjects by application of an external magnetic field to the bacterial population (ref from patterns and magnetic objects).
Milestones & Approach
Realising that magnetotaxis cannot be implemented in vivo by expressing the relevant genes from magnetotactic able organisms inside E.coli in the given time frame of the project we aim as a proof of principle to show the abilities of magnetotaxis by an ex vivo approach. This will be implemented by the preferential attachment of E.coli (through the usage of lock-and-key mechanisms e.g. streptavidin-biotin [5]) to one hemisphere of a magnetic bead, thus simulating the effect of preferential polarity. As a second step to this we need to clearly control the flagellar genetic networks that control motion as attachment onto surfaces may incapacitate movement [6].
As it is difficult to import the molecular machinery that guides vesicle formation from eukaryotic organisms to the prokaryotic context we aim to exploit the fact that vesicles bud off naturally from gram negative bacteria during their growth (any vesicle paper). Some work has been done to demonstrate the possibilities of such scenario by creating chimeric proteins with a haemolytic toxin, ClyA [7,8]. Our aim is to create chimeric proteins with a non-toxic protein to be used as a carrier to enable us to tag proteins of interest (cargo) into vesicular encapsulations. Proteins of interest may fall under the following categories:
(a) Exposed to the environment for preferential attachment to target cells by protein-protein interactions. (b) Encapsulated within the vesicle for delivery to target cells via membrane fusion. (c) Potential interaction of proteins encapsulated within vesicles by vesicle fusion.
Along with magnetotaxis we can implement a directed delivery system by guiding the bacterial organisms to locations of interest by the usage of external magnetic fields and the preferential budding of vesicles by controlling the expression of the carrier protein through genetic regulatory networks (GRNs). In a similar frame of mind the microorganisms can be used for nano-scale printing onto surfaces in a pattern of interest. An example of useful printing might be the laying down of tracks by having a filament like protein within vesicles, and vesicle fusion will give a track to be used by other protein motors.
Methodologies
Experimental and computational methods that aim to both self-complement and go beyond of the limitations of each method on its own will be pursuit. An extension to the stochastic simulator developed last year (BSim) to include features of bacterial population dynamics and the implementation of vesicle budding will give a more “real-world” approach to the simulation environment. Also the shape of bacteria will be updated to include a rod-like or ellipsoid shape which is closer to the real-life scenario but also will affect collision physics. Computations will be developed in Java and/or Matlab. Experimental techniques will test the simulation predictions at a later stage, and at earlier stages will aim to develop biobricks suitable for the usage by the synthetic biology community that will allow the incorporation of any protein by using one of our non-toxic carriers.
The application of vesicular encapsulations of targeted chemicals is of great interest to many research disciplines and our work will be used as grounding for others to follow.
[1] Yotaro Izumi, Lei Xu, Emmanuelle di Tomaso, Dai Fukumura & Rakesh K. Jain (2002), Tumour biology: Herceptin acts as an anti-angiogenic cocktail, Nature 416, 279-280
[2] Erythropoietin,http://www.cancerbackup.org.uk/treatments/supportivetherapies/erythropoietin, true on 28 July 2009
[3] Weissman, K. J. and Leadlay, P. F. (2005) Combinatorial biosynthesis of reduced polyketides. Nature Reviews Microbiol. 3, 925-936.
[4] Bazylinski A.D. and Frankel R.B. (2004), Magnetosomeformation in prokaryotes, Nat. Rev. Microb. 2, 217-230
[5]Streptavidin expressed on Outer Membtrane, http://partsregistry.org/Part:BBa_J36844
[6] Meadows P.S. (1970), The attachment of bacteria to solid surfaces, Arch. Mikrobiol. 75, 374-381
[7] Kim J., Doody A.M., Chen D.J., Cremona G.H., Shuler M. L., Putnam D. and DeLisa M.P. (2008) , Engineered Bacterial Outer Membrane Vesicles with Enhanced functionality, J. Mol. Biol. 380, 51-66.
[8] Sun Nyunt Wai, Barbro Lindmark, Tomas Söderblom, Akemi Takade, Marie Westermark, Jan Oscarsson, Jana Jass, Agneta Richter-Dahlfors, Yoshimitsu Mizunoe and Bernt Eric Uhlin (2003), Vesicle-Mediated Export and Assembly of Pore-Forming Oligomers of the Enterobacterial ClyA Cytotoxin, Cell 115, 25–35,
 "
"