Team:Imperial College London/Autoinduction
From 2009.igem.org
JamesField (Talk | contribs) (→Rationale) |
(→Autoinduction Contents) |
||
| (43 intermediate revisions not shown) | |||
| Line 1: | Line 1: | ||
{{Imperial/09/TemplateTop}} | {{Imperial/09/TemplateTop}} | ||
| + | <html> | ||
| + | <center> | ||
| + | <div class="highslide-gallery"> | ||
| + | <a href="https://static.igem.org/mediawiki/2009/c/ca/II09_MapIndicator_Autoinduction.png" class="highslide" onclick="return hs.expand(this, config1)" title="When glucose in the system runs out, the glucose-sensitive promoter is activated"> | ||
| + | <img src="https://static.igem.org/mediawiki/2009/c/ca/II09_MapIndicator_Autoinduction.png" alt="" title="Click to enlarge" width="75%"/> | ||
| + | </a> | ||
| + | </div> | ||
| + | </center> | ||
| + | </html> | ||
| + | =<font size='5'><b>Module Integration - Autoinduction</b></font>= | ||
| + | ==Overview== | ||
| - | + | The autoinduction process switches on <b>Module 2</b> following the accumulation of a predetermined concentration of polypeptide. The term <i>'autoinduction'</i> was chosen as the flipping of this switch is triggered by the chemical composition of the media in which our chassis is grown. | |
| - | + | ||
| - | + | The <i>'on signal'</i> orginates from inside the system: hence <i>'autoinduction'</i>. | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | |||
| - | |||
| - | |||
<html> | <html> | ||
<center> | <center> | ||
<div class="highslide-gallery"> | <div class="highslide-gallery"> | ||
| - | <a href=" | + | <a href="https://static.igem.org/mediawiki/2009/e/e3/Module2Timeline.png" class="highslide" onclick="return hs.expand(this, config1)"> |
| - | <img src=" | + | <img src="https://static.igem.org/mediawiki/2009/e/e3/Module2Timeline.png" alt="" title="Click to enlarge" width="75%"/> |
</a> | </a> | ||
<div class="highslide-caption"> | <div class="highslide-caption"> | ||
| - | Module 2 | + | Module 2 Timeline |
</div> | </div> | ||
</div> | </div> | ||
| Line 27: | Line 30: | ||
<br> | <br> | ||
| - | = | + | ==Rationale== |
| - | + | ||
| - | + | ||
| - | + | ||
| - | =Rationale= | + | |
| - | + | ||
| - | + | ||
| + | Before embarking on the creation of an autoinduction switch, we reviewed a number of possible alternatives. A genetically encoded timer was initially considered however these present a considerable number of problems that were absent from an autoinduction switch. | ||
<!--template:----------------------------------------------------------------------------> | <!--template:----------------------------------------------------------------------------> | ||
| Line 44: | Line 42: | ||
| Existing genetic timers are unable to provide a long enough delay. | | Existing genetic timers are unable to provide a long enough delay. | ||
| The length of our delay can be tuned by altering the composition of our media | | The length of our delay can be tuned by altering the composition of our media | ||
| - | |||
|- style="color:#333; background-color:#CCCCFF;" cellpadding="6" cellspacing="0" border="1" | |- style="color:#333; background-color:#CCCCFF;" cellpadding="6" cellspacing="0" border="1" | ||
| - | + | | They are liable to fall out of phase. | |
| - | | | + | | Since the whole population are exposed to the same media, a greater proportion will begin <b>Module 2</b> in synchrony. |
| - | | | + | |- style="color:#333; background-color:#CCCCFF;" cellpadding="6" cellspacing="0" border="1" |
| + | | They require multiple genes. | ||
| + | | Only a single promoter is required for our autoinduction switch. | ||
|} | |} | ||
| + | We believe that the advantages and modularity of our autoinduction switch is such that future iGEM teams will be able to deploy it in a broad range of senarios. | ||
| + | ==Initiation== | ||
| - | + | <b>Module 2</b> is triggered by a rise in levels of <i>cyclic Adenosine Mono Phosphate</i> (cAMP). This is because cAMP facilitates the dimerisation of the transcription factor CRP which induces transcription downstream of the PcstA promoter ([http://partsregistry.org/Part:BBa_K118011 BBa_K118011]). The rise in cAMP is correlated with a fall in levels of the primary carbon source (glucose). Thus, by controlling the initial amount of glucose present in the system, we achieve a timer function for the start of <b>Module 2</b>. | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
<html> | <html> | ||
<center> | <center> | ||
<div class="highslide-gallery"> | <div class="highslide-gallery"> | ||
| - | <a href="https://static.igem.org/mediawiki/2009/ | + | <a href="https://static.igem.org/mediawiki/2009/5/5b/CAMPModule2.png" class="highslide" onclick="return hs.expand(this, config1)"> |
| - | <img src="https://static.igem.org/mediawiki/2009/ | + | <img src="https://static.igem.org/mediawiki/2009/5/5b/CAMPModule2.png" alt="" title="Click to enlarge" width="70%"/> |
</a> | </a> | ||
<div class="highslide-caption"> | <div class="highslide-caption"> | ||
| - | Module 2 | + | Module 2: Initiation |
</div> | </div> | ||
</div> | </div> | ||
</center> | </center> | ||
</html> | </html> | ||
| - | |||
| - | |||
| - | |||
| - | + | ==Results== | |
| + | ===Wet Lab=== | ||
<html> | <html> | ||
| - | < | + | <left> |
<div class="highslide-gallery"> | <div class="highslide-gallery"> | ||
| - | <a href="https://static.igem.org/mediawiki/2009/ | + | <a href="https://static.igem.org/mediawiki/2009/0/02/PcstAGFPConstruct.png" class="highslide" onclick="return hs.expand(this, config1)"> |
| - | <img src="https://static.igem.org/mediawiki/2009/ | + | <img src="https://static.igem.org/mediawiki/2009/0/02/PcstAGFPConstruct.png" alt="" title="Click to enlarge" width="45%"/> |
</a> | </a> | ||
<div class="highslide-caption"> | <div class="highslide-caption"> | ||
| - | Module 2: | + | Module 2: Autoinduction Testing Construct |
</div> | </div> | ||
</div> | </div> | ||
| - | </ | + | </left> |
</html> | </html> | ||
| - | =Results= | + | To characterise the properties of our autoinduction switch, we put the fluorescent protein GFP under the control of the PcstA promoter (<partinfo>BBa_K200018 </partinfo>) as shown above. <br> |
| + | <br> | ||
| + | [[Image:CRPmedia corrected fluor.jpg|400px|right]] | ||
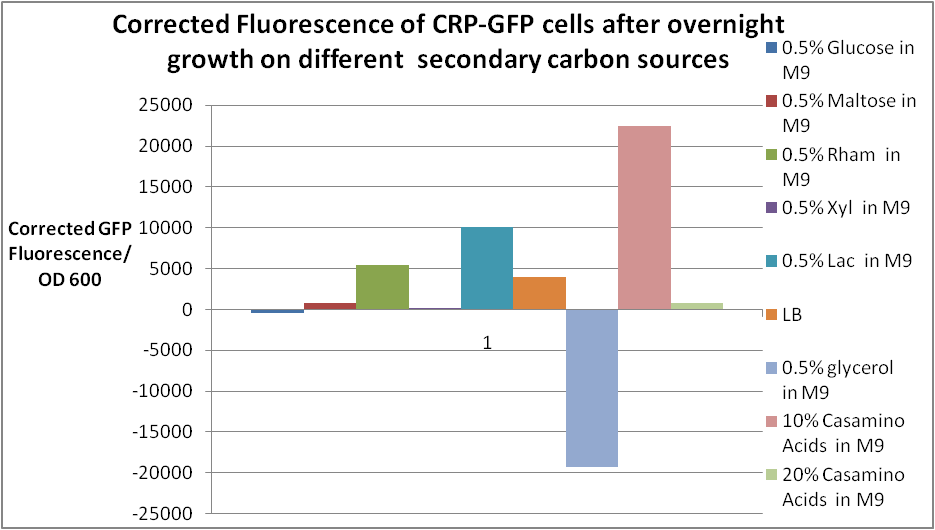
| + | Cells with BBa_K200018 were grown overnight in various different media, and the GFP fluorescence was measured. <br> | ||
| + | <br> | ||
| + | After overnight culture, the corrected fluorescence of glucose is almost negligible, showing that <b>glucose represses the PcstA promoter strongly</b>. <br> | ||
| + | <br> | ||
| + | For all the other secondary carbon sources, <b>10% Casamino Acids in M9 shows the highest corrected fluorescence</b> at 22000.<br> | ||
| + | <br> | ||
| + | <html><a href="https://2009.igem.org/Team:Imperial_College_London/Wetlab/Results#Autoinduction | ||
| + | "><img style="vertical-align:bottom;" width=50px align="left" src="http://i691.photobucket.com/albums/vv271/dk806/II09Learnmore.png"></a></html><b> About the wetlab results!</b> | ||
| + | <br> | ||
| + | <br> | ||
| + | <br> | ||
| + | |||
| + | ===Dry Lab=== | ||
| + | |||
| + | |||
| + | |||
[[Image:II09_diax_wolf.jpg | left]]We have modelled autoinduction by a number of models, and we hope that experimental results will confirm which model is most applicable to our system. | [[Image:II09_diax_wolf.jpg | left]]We have modelled autoinduction by a number of models, and we hope that experimental results will confirm which model is most applicable to our system. | ||
The first of our diauxic growth models is a cybernetic model developed by Kompala et al [1]. This model shows that glucose (S1) is used up before the secondary carbon source (S2). During this phase, the population (X) is in exponential grow until glucose (S1) runs out. This is followed by a stationary growth phase, and finally the population enters a second exponential growth phase as they start uptaking the secondary carbon source (S2). Another model was developed by Keasling [2] that takes into account the dynamics of the lactose operon. | The first of our diauxic growth models is a cybernetic model developed by Kompala et al [1]. This model shows that glucose (S1) is used up before the secondary carbon source (S2). During this phase, the population (X) is in exponential grow until glucose (S1) runs out. This is followed by a stationary growth phase, and finally the population enters a second exponential growth phase as they start uptaking the secondary carbon source (S2). Another model was developed by Keasling [2] that takes into account the dynamics of the lactose operon. | ||
| - | |||
<html><a href="https://2009.igem.org/Team:Imperial_College_London/Drylab/Autoinduction | <html><a href="https://2009.igem.org/Team:Imperial_College_London/Drylab/Autoinduction | ||
| - | "><img style="vertical-align:bottom;" width=50px align="left" src="http://i691.photobucket.com/albums/vv271/dk806/II09Learnmore.png"></a></html | + | "><img style="vertical-align:bottom;" width=50px align="left" src="http://i691.photobucket.com/albums/vv271/dk806/II09Learnmore.png"></a></html><b> About the models and simulations!</b> |
<br> | <br> | ||
<br> | <br> | ||
| + | |||
| + | <center> | ||
| + | |||
| + | |||
| + | </center> | ||
| + | |||
| + | ==Conclusion== | ||
| + | |||
| + | We have chosen a strategy of autoinduction to control the initiation of <b>Module 2</b>. Based on experimental data collected by our team, we were able to select 10% casamino acids as the most suitable secondary carbon source for maximal output from the PcstA promoter. | ||
| + | |||
| + | <center> | ||
| + | ===Project Tour=== | ||
| + | |||
<html><center> | <html><center> | ||
<a href="https://2009.igem.org/Team:Imperial_College_London/M1"><img width=150px src="http://i691.photobucket.com/albums/vv271/dk806/Module1L.jpg"></a> | <a href="https://2009.igem.org/Team:Imperial_College_London/M1"><img width=150px src="http://i691.photobucket.com/albums/vv271/dk806/Module1L.jpg"></a> | ||
| Line 115: | Line 131: | ||
</html> | </html> | ||
<br> | <br> | ||
| + | <hr> | ||
| + | ===Temporal Control Contents=== | ||
| + | |||
| + | <html><center><a href="https://2009.igem.org/Team:Imperial_College_London/Temporal_Control/Chemical_Induction"><img style="vertical-align:bottom;" width="20%" src="http://i691.photobucket.com/albums/vv271/dk806/II09_chemicalinduction.png"></a><a href="https://2009.igem.org/Team:Imperial_College_London/Temporal_Control/Autoinduction"><img style="vertical-align:bottom;" width="20%" src="http://i691.photobucket.com/albums/vv271/dk806/II09_Drylabmainimage1.png"></a><a href="https://2009.igem.org/Team:Imperial_College_London/Temporal_Control/Thermoinduction"><img style="vertical-align:bottom;" width="20%" src="http://i691.photobucket.com/albums/vv271/dk806/II09_Thermoinduction1.png"></a><a href="https://2009.igem.org/Team:Imperial_College_London/Wetlab/Results#Temporal_Control"><img style="vertical-align:bottom;"width="20%"src="http://i691.photobucket.com/albums/vv271/dk806/II09_Wetlabmainimage9.png"></a><a href="https://2009.igem.org/Team:Imperial_College_London/Drylab/Autoinduction"><img style="vertical-align:bottom;" width="20%" src="http://i691.photobucket.com/albums/vv271/dk806/II09_Drylabmainimage6.png"></a></center></html> | ||
| + | <html><table border="0" style="background-color:transparent;" width="100%"> | ||
| + | <tr><td width="0%"></td> | ||
| + | <td width="20%"><center><a href="https://2009.igem.org/Team:Imperial_College_London/Temporal_Control/Chemical_Induction"><b>Chemoinduction</b></a></center></td> | ||
| + | <td width="20%"><center><a href="https://2009.igem.org/Team:Imperial_College_London/Temporal_Control/Autoinduction"><b>Autoinduction</b></a></center></td> | ||
| + | <td width="20%"><center><a href="https://2009.igem.org/Team:Imperial_College_London/Temporal_Control/Thermoinduction"><b>Thermoinduction</b></a></center></td> | ||
| + | <td width="20%"><center><a href="https://2009.igem.org/Team:Imperial_College_London/Wetlab/Results#Temporal_Control"><b>Wet Lab</b></a></center></td> | ||
| + | <td width="20%"><center><a href="https://2009.igem.org/Team:Imperial_College_London/Drylab/Autoinduction"><b>Modelling</b></a></center></td></table></html> | ||
{{Imperial/09/TemplateBottom}} | {{Imperial/09/TemplateBottom}} | ||
Latest revision as of 03:52, 22 October 2009

Contents |
Module Integration - Autoinduction
Overview
The autoinduction process switches on Module 2 following the accumulation of a predetermined concentration of polypeptide. The term 'autoinduction' was chosen as the flipping of this switch is triggered by the chemical composition of the media in which our chassis is grown.
The 'on signal' orginates from inside the system: hence 'autoinduction'.
Rationale
Before embarking on the creation of an autoinduction switch, we reviewed a number of possible alternatives. A genetically encoded timer was initially considered however these present a considerable number of problems that were absent from an autoinduction switch.
| Genetic Timers | Autoinduction Switch |
|---|---|
| Existing genetic timers are unable to provide a long enough delay. | The length of our delay can be tuned by altering the composition of our media |
| They are liable to fall out of phase. | Since the whole population are exposed to the same media, a greater proportion will begin Module 2 in synchrony. |
| They require multiple genes. | Only a single promoter is required for our autoinduction switch. |
We believe that the advantages and modularity of our autoinduction switch is such that future iGEM teams will be able to deploy it in a broad range of senarios.
Initiation
Module 2 is triggered by a rise in levels of cyclic Adenosine Mono Phosphate (cAMP). This is because cAMP facilitates the dimerisation of the transcription factor CRP which induces transcription downstream of the PcstA promoter ([http://partsregistry.org/Part:BBa_K118011 BBa_K118011]). The rise in cAMP is correlated with a fall in levels of the primary carbon source (glucose). Thus, by controlling the initial amount of glucose present in the system, we achieve a timer function for the start of Module 2.
Results
Wet Lab
To characterise the properties of our autoinduction switch, we put the fluorescent protein GFP under the control of the PcstA promoter () as shown above.
Cells with BBa_K200018 were grown overnight in various different media, and the GFP fluorescence was measured.
After overnight culture, the corrected fluorescence of glucose is almost negligible, showing that glucose represses the PcstA promoter strongly.
For all the other secondary carbon sources, 10% Casamino Acids in M9 shows the highest corrected fluorescence at 22000.
 About the wetlab results!
About the wetlab results!
Dry Lab
We have modelled autoinduction by a number of models, and we hope that experimental results will confirm which model is most applicable to our system.The first of our diauxic growth models is a cybernetic model developed by Kompala et al [1]. This model shows that glucose (S1) is used up before the secondary carbon source (S2). During this phase, the population (X) is in exponential grow until glucose (S1) runs out. This is followed by a stationary growth phase, and finally the population enters a second exponential growth phase as they start uptaking the secondary carbon source (S2). Another model was developed by Keasling [2] that takes into account the dynamics of the lactose operon.
 About the models and simulations!
About the models and simulations!
Conclusion
We have chosen a strategy of autoinduction to control the initiation of Module 2. Based on experimental data collected by our team, we were able to select 10% casamino acids as the most suitable secondary carbon source for maximal output from the PcstA promoter.
Project Tour


Temporal Control Contents





 "
"