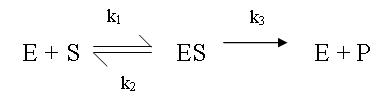Team:Imperial College London/Drylab/Enzyme/Analysis
From 2009.igem.org
(Difference between revisions)
| Line 38: | Line 38: | ||
*The Michaelis-Menten (MM) assumption is a simplifying assumption that is commonly made with enzymatic reactions. It says that since substrate binding is very fast compared to catalysis, the enzyme complex ES is always at quasi-steady state, ie: [[Image:ek2.jpg | 70px]], during the time of the experiment. | *The Michaelis-Menten (MM) assumption is a simplifying assumption that is commonly made with enzymatic reactions. It says that since substrate binding is very fast compared to catalysis, the enzyme complex ES is always at quasi-steady state, ie: [[Image:ek2.jpg | 70px]], during the time of the experiment. | ||
| - | *It is straightforward to prove that combining the MM assumption [[Image:ii09_ekeqn1.png | 350px]]with the conservation equation [E<sub>0</sub>]=[E]+[ES] yields a new form for the fourth equation | + | *It is straightforward to prove that combining the MM assumption [[Image:ii09_ekeqn1.png | 350px]]with the conservation equation [E<sub>0</sub>]=[E]+[ES] yields a new form for the fourth equation [[Image:ii09_ekeqnk4mm.png | 200px]] |
Revision as of 20:56, 9 October 2009

- Overview
- The model
- Simulations
Assumptions
In the standard model of enzymatic reaction, 3 sets of assumptions are made.
Enzymatic assumptions:
- The enzyme is specific only for the substrate and not for any other chemicals.
- Only one enzyme, our enzyme of interest is present and participating in the reaction.
- There is negligible formation of product without the enzyme.
- The rate of enzymatic activity remains constant over time because there is:
- no co-operativity of the system. Binding of substrate to one enzyme binding site doesn't influence the affinity or activity of an adjacent site.
- no allosteric regulations from either the product or the substrate.
- no product inhibition of the enzyme.
- The enzymatic reaction can be modelled by the following set of reactions (in particular the catalysed reaction is irreversible)
Degradation assumptions:
- All proteins are very stable and thus their degradation can be neglected over the course of the experiments
- If there is no creation of substrate during the experiment (which is the most common case), we thus have [S]0=[S]+[ES]+[P] at all time
- Likewise if there is no creation of enzyme during the experiment (which is the most common case), we have [E0] =[E]+[ES] at all time
Law of mass action assumptions:
- Free diffusion ; Free unrestricted thermodynamically driven random molecular motion
- The reagents are in thermal equilibrium at a constant absolute temperature
- The reagents are well mixed. [2]
Michaelis-Menten assumption:
- The Michaelis-Menten (MM) assumption is a simplifying assumption that is commonly made with enzymatic reactions. It says that since substrate binding is very fast compared to catalysis, the enzyme complex ES is always at quasi-steady state, ie:
 , during the time of the experiment.
, during the time of the experiment.
- It is straightforward to prove that combining the MM assumption
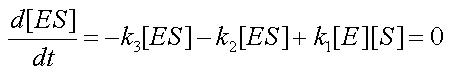 with the conservation equation [E0]=[E]+[ES] yields a new form for the fourth equation
with the conservation equation [E0]=[E]+[ES] yields a new form for the fourth equation 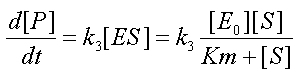
Model Predictions
- When substrate concentration is large, the enzyme concentration is the limiting factor, hence rate of reaction is directly proportional to [E].
- At very low [E], the reaction rate measured will be negligible as very low amounts of product will be produced.
- Increasing k1 and k3 values will increase the product synthesis rate.
The actual model...
To further explore the model, click here.
 "
"