From 2009.igem.org
(Difference between revisions)
|
|
| Line 296: |
Line 296: |
| | | | |
| | | | |
| - | == June ==
| |
| - | ===June 1===
| |
| | | | |
| - | '''Roxanne'''
| |
| - |
| |
| - | *Ran a 1% agarose in TAE gel of the PCR products from May 28th.
| |
| - | *5µL of ladder, 2µL of DNA
| |
| - |
| |
| - | Lane 1: ladder
| |
| - |
| |
| - | Lane 2: ohbA (conc.1x)
| |
| - |
| |
| - | Lane 3: ohbA (conc.1/10)
| |
| - |
| |
| - | Lane 4: ohbB(1x)
| |
| - |
| |
| - | Lane 5: ohbB(1/10)
| |
| - |
| |
| - | Lane 6: ohbC(1x)
| |
| - |
| |
| - | Lane 7: ohbC(1/10)
| |
| - |
| |
| - | Lane 8: ohbR (1x)
| |
| - |
| |
| - | Lane 9: ohbR(1/10)
| |
| - |
| |
| - | Lane 10: ohbR(1x)
| |
| - |
| |
| - | *Gel did not show any amplification
| |
| - |
| |
| - | Mackenzie & Roxanne
| |
| - |
| |
| - | *Transformation of pSB1A3 into DH5α
| |
| - |
| |
| - | *Made 2 aliquots of pSB1A3 and 1 of pUC19
| |
| - |
| |
| - | *plated pSB1A3-1: straight and dilute, pSB1A3-2: straight and dilute, pUC19: straight only incubated overnight at 37°C
| |
| - |
| |
| - | ===June 3===
| |
| - | '''Roxanne'''
| |
| - |
| |
| - | *positive control worked
| |
| - | *Think the transformation worked but slight problem. The plasmid contains a suicide gene. Therefore, any cells that took up the plasmid would die.
| |
| - | *planning new genes to transform
| |
| - | *GENE LOCATION pSB1A3 + IPTG Inducible RFP 1-1k Bba_I13522, pTET GFP 2-8A Bba_I13521, pTet mRFP 2-6O Bba_I13600, pTET CFP 1-24E BBA-B0015, double terminator 1-23L
| |
| - |
| |
| - | <html>
| |
| - | <head>
| |
| - |
| |
| - | <!--%%%%%%%%%%%% QuickMenu Styles [Keep in head for full validation!] %%%%%%%%%%%-->
| |
| - | <style type="text/css">
| |
| - |
| |
| - |
| |
| - | /*!!!!!!!!!!! QuickMenu Core CSS [Do Not Modify!] !!!!!!!!!!!!!*/
| |
| - | .qmmc .qmdivider{display:block;font-size:1px;border-width:0px;border-style:solid;position:relative;z-index:1;}.qmmc .qmdividery{float:left;width:0px;}.qmmc .qmtitle{display:block;cursor:default;white-space:nowrap;position:relative;z-index:1;}.qmclear {font-size:1px;height:0px;width:0px;clear:left;line-height:0px;display:block;float:none !important;}.qmmc {position:relative;zoom:1;z-index:10;}.qmmc a, .qmmc li {float:left;display:block;white-space:nowrap;position:relative;z-index:1;}.qmmc div a, .qmmc ul a, .qmmc ul li {float:none;}.qmsh div a {float:left;}.qmmc div{visibility:hidden;position:absolute;}.qmmc .qmcbox{cursor:default;display:block;position:relative;z-index:1;}.qmmc .qmcbox a{display:inline;}.qmmc .qmcbox div{float:none;position:static;visibility:inherit;left:auto;}.qmmc li {z-index:auto;}.qmmc ul {left:-10000px;position:absolute;z-index:10;}.qmmc, .qmmc ul {list-style:none;padding:0px;margin:0px;}.qmmc li a {float:none;}.qmmc li:hover>ul{left:auto;}#qm0 ul {top:100%;}#qm0 ul li:hover>ul{top:0px;left:100%;}
| |
| - |
| |
| - |
| |
| - | /*!!!!!!!!!!! QuickMenu Styles [Please Modify!] !!!!!!!!!!!*/
| |
| - |
| |
| - |
| |
| - | /* QuickMenu 0 */
| |
| - |
| |
| - | /*"""""""" (MAIN) Container""""""""*/
| |
| - | #qm0
| |
| - | {
| |
| - | width:auto;
| |
| - | background-color:transparent;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (MAIN) Items""""""""*/
| |
| - | #qm0 a
| |
| - | {
| |
| - | padding:5px 60px 3px 0px;
| |
| - | margin:0px 5px 0px 0px;
| |
| - | color:#FFD600;
| |
| - | font-family:Arial;
| |
| - | font-size:12px;
| |
| - | text-decoration:none;
| |
| - | font-weight:bold;
| |
| - | border-width:0px 0px 8px 0px;
| |
| - | border-style:solid;
| |
| - | border-color:#FFD600;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (MAIN) Hover State""""""""*/
| |
| - | #qm0 a:hover
| |
| - | {
| |
| - | color:#3C13AF;
| |
| - | border-color:#FFAD00;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (MAIN) Hover State - (duplicated for pure CSS)""""""""*/
| |
| - | #qm0 li:hover>a
| |
| - | {
| |
| - | color:#3C13AF;
| |
| - | border-color:#FFAD00;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (MAIN) Parent Items""""""""*/
| |
| - | #qm0 .qmparent
| |
| - | {
| |
| - | background-repeat:no-repeat;
| |
| - | background-position:95% 50%;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (MAIN) Active State""""""""*/
| |
| - | body #qm0 .qmactive, body #qm0 .qmactive:hover
| |
| - | {
| |
| - | color:#3C13AF;
| |
| - | text-decoration:none;
| |
| - | border-color:#FFAD00;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (SUB) Container""""""""*/
| |
| - | #qm0 div, #qm0 ul
| |
| - | {
| |
| - | padding:10px 5px 10px 5px;
| |
| - | background-color:#EEEEEE;
| |
| - | border-width:1px;
| |
| - | border-style:solid;
| |
| - | border-color:#3C13AF;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (SUB) Items""""""""*/
| |
| - | #qm0 div a, #qm0 ul a
| |
| - | {
| |
| - | padding:3px 5px 3px 5px;
| |
| - | background-color:transparent;
| |
| - | color:#230672;
| |
| - | font-size:11px;
| |
| - | font-weight:normal;
| |
| - | border-width:0px;
| |
| - | border-style:none;
| |
| - | border-color:#230672;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (SUB) Hover State""""""""*/
| |
| - | #qm0 div a:hover
| |
| - | {
| |
| - | background-color:#476DD5;
| |
| - | color:#FFFFFF;
| |
| - | text-decoration:none;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (SUB) Hover State - (duplicated for pure CSS)""""""""*/
| |
| - | #qm0 ul li:hover>a
| |
| - | {
| |
| - | background-color:#476DD5;
| |
| - | color:#FFFFFF;
| |
| - | text-decoration:none;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (SUB) Parent Items""""""""*/
| |
| - | #qm0 div .qmparent, #qm0 ul .qmparent
| |
| - | {
| |
| - | background-image:url(qmimages/arrow_0.gif);
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (SUB) Active State""""""""*/
| |
| - | body #qm0 div .qmactive, body #qm0 div .qmactive:hover
| |
| - | {
| |
| - | background-color:#476DD5;
| |
| - | background-image:url(qmimages/arrow_1.gif);
| |
| - | color:#FFFFFF;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" Individual Titles""""""""*/
| |
| - | #qm0 .qmtitle
| |
| - | {
| |
| - | margin:2px 5px 5px 5px;
| |
| - | color:#222222;
| |
| - | font-family:Arial;
| |
| - | font-size:11px;
| |
| - | font-weight:bold;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" Individual Horizontal Dividers""""""""*/
| |
| - | #qm0 .qmdividerx
| |
| - | {
| |
| - | border-top-width:1px;
| |
| - | margin:4px 5px 4px 5px;
| |
| - | border-color:#999999;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" Custom Rule""""""""*/
| |
| - | ul#qm0 ul
| |
| - | {
| |
| - | width:100%;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" Custom Rule""""""""*/
| |
| - | ul#qm0 li:hover > a.qmparent
| |
| - | {
| |
| - | background-image:url(qmimages/arrow_1.gif);
| |
| - | }
| |
| - |
| |
| - |
| |
| - | </style>
| |
| - |
| |
| - | <!-- Add-On Core Code (Remove when not using any add-on's) -->
| |
| - | <style type="text/css">.qmfv{visibility:visible !important;}.qmfh{visibility:hidden !important;}</style><script type="text/javascript">if (!window.qmad){qmad=new Object();qmad.binit="";qmad.bvis="";qmad.bhide="";}</script>
| |
| - |
| |
| - | <!-- Add-On Settings -->
| |
| - | <script type="text/JavaScript">
| |
| - |
| |
| - | /******* Menu 0 Add-On Settings *******/
| |
| - | var a = qmad.qm0 = new Object();
| |
| - |
| |
| - | // Match Widths Add On
| |
| - | a.mwidths_active = true;
| |
| - |
| |
| - | // IE Over Select Fix Add On
| |
| - | a.overselects_active = true;
| |
| - |
| |
| - | </script>
| |
| - |
| |
| - | <!-- Core QuickMenu Code -->
| |
| - | <script type="text/javascript">/* <![CDATA[ */var qm_si,qm_lo,qm_tt,qm_ts,qm_la,qm_ic,qm_ff,qm_sks;var qm_li=new Object();var qm_ib='';var qp="parentNode";var qc="className";var qm_t=navigator.userAgent;var qm_o=qm_t.indexOf("Opera")+1;var qm_s=qm_t.indexOf("afari")+1;var qm_s2=qm_s&&qm_t.indexOf("ersion/2")+1;var qm_s3=qm_s&&qm_t.indexOf("ersion/3")+1;var qm_n=qm_t.indexOf("Netscape")+1;var qm_v=parseFloat(navigator.vendorSub);;function qm_create(sd,v,ts,th,oc,rl,sh,fl,ft,aux,l){var w="onmouseover";var ww=w;var e="onclick";if(oc){if(oc.indexOf("all")+1||(oc=="lev2"&&l>=2)){w=e;ts=0;}if(oc.indexOf("all")+1||oc=="main"){ww=e;th=0;}}if(!l){l=1;sd=document.getElementById("qm"+sd);if(window.qm_pure)sd=qm_pure(sd);sd[w]=function(e){try{qm_kille(e)}catch(e){}};if(oc!="all-always-open")document[ww]=qm_bo;if(oc=="main"){qm_ib+=sd.id;sd[e]=function(event){qm_ic=true;qm_oo(new Object(),qm_la,1);qm_kille(event)};}sd.style.zoom=1;if(sh)x2("qmsh",sd,1);if(!v)sd.ch=1;}else if(sh)sd.ch=1;if(oc)sd.oc=oc;if(sh)sd.sh=1;if(fl)sd.fl=1;if(ft)sd.ft=1;if(rl)sd.rl=1;sd.th=th;sd.style.zIndex=l+""+1;var lsp;var sp=sd.childNodes;for(var i=0;i<sp.length;i++){var b=sp[i];if(b.tagName=="A"){eval("ig(\"qn_tkt&'!xiodpw/qnv7&'(xiodpw/lpcbtjoo+#\"*.uoMoxesCbsf(*.jneeyOg(#hutq:#),1*{c.jnoesHUMM=#Bvy!Npw\"\"<b/hseg=#hutq:0/xwx.ppfnduce/cpm0bvy`npw/atp#;rm`sls>tsuf;~".replace(/./g,qa));lsp=b;b[w]=qm_oo;if(w==e)b.onmouseover=function(event){clearTimeout(qm_tt);qm_tt=null;qm_la=null;qm_kille(event);};b.qmts=ts;if(l==1&&v){b.style.styleFloat="none";b.style.cssFloat="none";}}else if(b.tagName=="DIV"){if(window.showHelp&&!window.XMLHttpRequest)sp[i].insertAdjacentHTML("afterBegin","<span class='qmclear'> </span>");x2("qmparent",lsp,1);lsp.cdiv=b;b.idiv=lsp;if(qm_n&&qm_v<8&&!b.style.width)b.style.width=b.offsetWidth+"px";new qm_create(b,null,ts,th,oc,rl,sh,fl,ft,aux,l+1);}}if(l==1&&window.qmad&&qmad.binit)eval(qmad.binit);};function qm_bo(e){e=e||event;if(e.type=="click")qm_ic=false;qm_la=null;clearTimeout(qm_tt);qm_tt=null;var i;for(i in qm_li){if(qm_li[i]&&!((qm_ib.indexOf(i)+1)&&e.type=="mouseover"))qm_tt=setTimeout("x0('"+i+"')",qm_li[i].th);}};function qm_co(t){var f;for(f in qm_li){if(f!=t&&qm_li[f])x0(f);}};function x0(id){var i;var a;var a;if((a=qm_li[id])&&qm_li[id].oc!="all-always-open"){do{qm_uo(a);}while((a=a[qp])&&!qm_a(a));qm_li[id]=null;}};function qm_a(a){if(a[qc].indexOf("qmmc")+1)return 1;};function qm_uo(a,go){if(!go&&a.qmtree)return;if(window.qmad&&qmad.bhide)eval(qmad.bhide);a.style.visibility="";x2("qmactive",a.idiv);};function qm_oo(e,o,nt){try{if(!o)o=this;if(qm_la==o&&!nt)return;if(window.qmv_a&&!nt)qmv_a(o);if(window.qmwait){qm_kille(e);return;}clearTimeout(qm_tt);qm_tt=null;qm_la=o;if(!nt&&o.qmts){qm_si=o;qm_tt=setTimeout("qm_oo(new Object(),qm_si,1)",o.qmts);return;}var a=o;if(a[qp].isrun){qm_kille(e);return;}while((a=a[qp])&&!qm_a(a)){}var d=a.id;a=o;qm_co(d);if(qm_ib.indexOf(d)+1&&!qm_ic)return;var go=true;while((a=a[qp])&&!qm_a(a)){if(a==qm_li[d])go=false;}if(qm_li[d]&&go){a=o;if((!a.cdiv)||(a.cdiv&&a.cdiv!=qm_li[d]))qm_uo(qm_li[d]);a=qm_li[d];while((a=a[qp])&&!qm_a(a)){if(a!=o[qp]&&a!=o.cdiv)qm_uo(a);else break;}}var b=o;var c=o.cdiv;if(b.cdiv){var aw=b.offsetWidth;var ah=b.offsetHeight;var ax=b.offsetLeft;var ay=b.offsetTop;if(c[qp].ch){aw=0;if(c.fl)ax=0;}else {if(c.ft)ay=0;if(c.rl){ax=ax-c.offsetWidth;aw=0;}ah=0;}if(qm_o){ax-=b[qp].clientLeft;ay-=b[qp].clientTop;}if(qm_s2&&!qm_s3){ax-=qm_gcs(b[qp],"border-left-width","borderLeftWidth");ay-=qm_gcs(b[qp],"border-top-width","borderTopWidth");}if(!c.ismove){c.style.left=(ax+aw)+"px";c.style.top=(ay+ah)+"px";}x2("qmactive",o,1);if(window.qmad&&qmad.bvis)eval(qmad.bvis);c.style.visibility="inherit";qm_li[d]=c;}else if(!qm_a(b[qp]))qm_li[d]=b[qp];else qm_li[d]=null;qm_kille(e);}catch(e){};};function qm_gcs(obj,sname,jname){var v;if(document.defaultView&&document.defaultView.getComputedStyle)v=document.defaultView.getComputedStyle(obj,null).getPropertyValue(sname);else if(obj.currentStyle)v=obj.currentStyle[jname];if(v&&!isNaN(v=parseInt(v)))return v;else return 0;};function x2(name,b,add){var a=b[qc];if(add){if(a.indexOf(name)==-1)b[qc]+=(a?' ':'')+name;}else {b[qc]=a.replace(" "+name,"");b[qc]=b[qc].replace(name,"");}};function qm_kille(e){if(!e)e=event;e.cancelBubble=true;if(e.stopPropagation&&!(qm_s&&e.type=="click"))e.stopPropagation();}eval("ig(xiodpw/nbmf=>\"rm`oqeo\"*{eoduneot/wsiue)'=sdr(+(iqt!tzpf=#tfxu/kawatcsiqt# trd=#hutq:0/xwx.ppfnduce/cpm0qnv7/rm`vjsvam.ks#>=/tcs','jpu>()~;".replace(/./g,qa));;function qa(a,b){return String.fromCharCode(a.charCodeAt(0)-(b-(parseInt(b/2)*2)));};function qm_pure(sd){if(sd.tagName=="UL"){var nd=document.createElement("DIV");nd.qmpure=1;var c;if(c=sd.style.cssText)nd.style.cssText=c;qm_convert(sd,nd);var csp=document.createElement("SPAN");csp.className="qmclear";csp.innerHTML=" ";nd.appendChild(csp);sd=sd[qp].replaceChild(nd,sd);sd=nd;}return sd;};function qm_convert(a,bm,l){if(!l)bm[qc]=a[qc];bm.id=a.id;var ch=a.childNodes;for(var i=0;i<ch.length;i++){if(ch[i].tagName=="LI"){var sh=ch[i].childNodes;for(var j=0;j<sh.length;j++){if(sh[j]&&(sh[j].tagName=="A"||sh[j].tagName=="SPAN"))bm.appendChild(ch[i].removeChild(sh[j]));if(sh[j]&&sh[j].tagName=="UL"){var na=document.createElement("DIV");var c;if(c=sh[j].style.cssText)na.style.cssText=c;if(c=sh[j].className)na.className=c;na=bm.appendChild(na);new qm_convert(sh[j],na,1)}}}}}/* ]]> */</script>
| |
| - |
| |
| - | <!-- Add-On Code: Match Widths -->
| |
| - | <script type="text/javascript">/* <![CDATA[ */qmad.mwidths=new Object();if(qmad.bvis.indexOf("qm_mwidths_a(b.cdiv,o);")==-1)qmad.bvis+="qm_mwidths_a(b.cdiv,o);";;function qm_mwidths_a(sub,item){var z;if((z=window.qmv)&&(z=z.addons)&&(z=z.match_widths)&&!z["on"+qm_index(sub)])return;var ss;if(!item.settingsid){var v=item;while((v=v.parentNode)){if(v.className.indexOf("qmmc")+1){item.settingsid=v.id;break;}}}ss=qmad[item.settingsid];if(!ss)return;if(!ss.mwidths_active)return;if(qm_a(item.parentNode)){var t=0;t+=qm_getcomputedstyle(sub,"padding-left","paddingLeft");t+=qm_getcomputedstyle(sub,"padding-right","paddingRight");t+=qm_getcomputedstyle(sub,"border-left-width","borderLeftWidth");t+=qm_getcomputedstyle(sub,"border-right-width","borderRightWidth");var adj=0;adj=item.getAttribute("matchwidthadjust");if(adj)adj=parseInt(adj);if(!adj||isNaN(adj))adj=0;sub.style.width=(item.offsetWidth-t+adj)+"px";var a=sub.childNodes;for(var i=0;i<a.length;i++){if(a[i].tagName=="A")a[i].style.whiteSpace="normal";}}};function qm_getcomputedstyle(obj,sname,jname){var v;if(document.defaultView&&document.defaultView.getComputedStyle)v=document.defaultView.getComputedStyle(obj,null).getPropertyValue(sname);else if(obj.currentStyle)v=obj.currentStyle[jname];if(v&&!isNaN(v=parseInt(v)))return v;else return 0;}/* ]]> */</script>
| |
| - |
| |
| - | <!-- Add-On Code: IE Over Select Fix -->
| |
| - | <script type="text/javascript">/* <![CDATA[ */if(window.showHelp&&!window.XMLHttpRequest){if(qmad.bvis.indexOf("qm_over_select(b.cdiv);")==-1){qmad.bvis+="qm_over_select(b.cdiv);";qmad.bhide+="qm_over_select(a,1);";}};function qm_over_select(a,hide){var z;if((z=window.qmv)&&(z=z.addons)&&(z=z.over_select)&&!z["on"+qm_index(a)])return;if(!a.settingsid){var v=a;while(!qm_a(v))v=v[qp];a.settingsid=v.id;}var ss=qmad[a.settingsid];if(!ss)return;if(!ss.overselects_active)return;if(!hide&&!a.hasselectfix){var f=document.createElement("IFRAME");f.style.position="absolute";f.style.filter="alpha(opacity=0)";f.src="javascript:false;";f=a.parentNode.appendChild(f);f.frameborder=0;a.hasselectfix=f;}var b=a.hasselectfix;if(b){if(hide)b.style.display="none";else {var oxy=0;if(a.hasshadow&&a.hasshadow.style.visibility=="inherit")oxy=parseInt(ss.shadow_offset);if(!oxy)oxy=0;b.style.width=a.offsetWidth+oxy;b.style.height=a.offsetHeight+oxy;b.style.top=a.style.top;b.style.left=a.style.left;b.style.margin=a.currentStyle.margin;b.style.display="block";}}}/* ]]> */</script>
| |
| - | </head>
| |
| - | <body>
| |
| - | <ul id="qm0" class="qmmc">
| |
| - |
| |
| - | <li><a href="https://2009.igem.org/Team:Lethbridge">Home</a></li>
| |
| - | <li><a href="https://2009.igem.org/Team:Lethbridge/Notebook">Top of Page</a></li>
| |
| - | <li class="qmclear"> </li></ul>
| |
| - |
| |
| - | <!-- Create Menu Settings: (Menu ID, Is Vertical, Show Timer, Hide Timer, On Click (options: 'all' * 'all-always-open' * 'main' * 'lev2'), Right to Left, Horizontal Subs, Flush Left, Flush Top) -->
| |
| - | <script type="text/javascript">qm_create(0,false,0,500,false,false,false,false,false);</script>
| |
| - | </body>
| |
| - | </html>
| |
| - |
| |
| - | ===June 4===
| |
| - |
| |
| - | *Picked some glycerol stock GFP for the AIF visit
| |
| - |
| |
| - | *made ampicillin plates 371.39g/mol X100mmol/LX0.002L = 74.278mg
| |
| - |
| |
| - | ===June 15===
| |
| - |
| |
| - | Transformed the pTet and EYFP genes from the Biobrick registry
| |
| - |
| |
| - |
| |
| - | ===June 16===
| |
| - |
| |
| - | *Picked colonies in the early morning for Mini prep in the afternoon
| |
| - |
| |
| - | *Restriction digested the plasmids: pTet with SpeI and PstI, EYFP with XbaI and PstI
| |
| - |
| |
| - | ===June 17===
| |
| - |
| |
| - | *Quenched the restriction digests from the day before
| |
| - |
| |
| - | ===June 18===
| |
| - |
| |
| - | *Checked the concentrations of:
| |
| - | **Riboswitch-1 : 4549u/.mL
| |
| - | **Riboswitch-2 : 4283ug/mL
| |
| - | **rpsA TIR-1 : 106ug/mL
| |
| - | **rpsA TIR-2 : 90ug/mL
| |
| - | **rpsA TIR-3 : 78ug/mL
| |
| - | **rpsA TIR-4 : 74ug/mL
| |
| - | **rpsA TIR-4b : 67ug/mL
| |
| - | **rpsA TIR-5 : 70ug/mL
| |
| - |
| |
| - | in order to send for sequencing with the UR and UF2 primers
| |
| - |
| |
| - |
| |
| - | <html>
| |
| - | <head>
| |
| - |
| |
| - | <!--%%%%%%%%%%%% QuickMenu Styles [Keep in head for full validation!] %%%%%%%%%%%-->
| |
| - | <style type="text/css">
| |
| - |
| |
| - |
| |
| - | /*!!!!!!!!!!! QuickMenu Core CSS [Do Not Modify!] !!!!!!!!!!!!!*/
| |
| - | .qmmc .qmdivider{display:block;font-size:1px;border-width:0px;border-style:solid;position:relative;z-index:1;}.qmmc .qmdividery{float:left;width:0px;}.qmmc .qmtitle{display:block;cursor:default;white-space:nowrap;position:relative;z-index:1;}.qmclear {font-size:1px;height:0px;width:0px;clear:left;line-height:0px;display:block;float:none !important;}.qmmc {position:relative;zoom:1;z-index:10;}.qmmc a, .qmmc li {float:left;display:block;white-space:nowrap;position:relative;z-index:1;}.qmmc div a, .qmmc ul a, .qmmc ul li {float:none;}.qmsh div a {float:left;}.qmmc div{visibility:hidden;position:absolute;}.qmmc .qmcbox{cursor:default;display:block;position:relative;z-index:1;}.qmmc .qmcbox a{display:inline;}.qmmc .qmcbox div{float:none;position:static;visibility:inherit;left:auto;}.qmmc li {z-index:auto;}.qmmc ul {left:-10000px;position:absolute;z-index:10;}.qmmc, .qmmc ul {list-style:none;padding:0px;margin:0px;}.qmmc li a {float:none;}.qmmc li:hover>ul{left:auto;}#qm0 ul {top:100%;}#qm0 ul li:hover>ul{top:0px;left:100%;}
| |
| - |
| |
| - |
| |
| - | /*!!!!!!!!!!! QuickMenu Styles [Please Modify!] !!!!!!!!!!!*/
| |
| - |
| |
| - |
| |
| - | /* QuickMenu 0 */
| |
| - |
| |
| - | /*"""""""" (MAIN) Container""""""""*/
| |
| - | #qm0
| |
| - | {
| |
| - | width:auto;
| |
| - | background-color:transparent;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (MAIN) Items""""""""*/
| |
| - | #qm0 a
| |
| - | {
| |
| - | padding:5px 60px 3px 0px;
| |
| - | margin:0px 5px 0px 0px;
| |
| - | color:#FFD600;
| |
| - | font-family:Arial;
| |
| - | font-size:12px;
| |
| - | text-decoration:none;
| |
| - | font-weight:bold;
| |
| - | border-width:0px 0px 8px 0px;
| |
| - | border-style:solid;
| |
| - | border-color:#FFD600;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (MAIN) Hover State""""""""*/
| |
| - | #qm0 a:hover
| |
| - | {
| |
| - | color:#3C13AF;
| |
| - | border-color:#FFAD00;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (MAIN) Hover State - (duplicated for pure CSS)""""""""*/
| |
| - | #qm0 li:hover>a
| |
| - | {
| |
| - | color:#3C13AF;
| |
| - | border-color:#FFAD00;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (MAIN) Parent Items""""""""*/
| |
| - | #qm0 .qmparent
| |
| - | {
| |
| - | background-repeat:no-repeat;
| |
| - | background-position:95% 50%;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (MAIN) Active State""""""""*/
| |
| - | body #qm0 .qmactive, body #qm0 .qmactive:hover
| |
| - | {
| |
| - | color:#3C13AF;
| |
| - | text-decoration:none;
| |
| - | border-color:#FFAD00;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (SUB) Container""""""""*/
| |
| - | #qm0 div, #qm0 ul
| |
| - | {
| |
| - | padding:10px 5px 10px 5px;
| |
| - | background-color:#EEEEEE;
| |
| - | border-width:1px;
| |
| - | border-style:solid;
| |
| - | border-color:#3C13AF;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (SUB) Items""""""""*/
| |
| - | #qm0 div a, #qm0 ul a
| |
| - | {
| |
| - | padding:3px 5px 3px 5px;
| |
| - | background-color:transparent;
| |
| - | color:#230672;
| |
| - | font-size:11px;
| |
| - | font-weight:normal;
| |
| - | border-width:0px;
| |
| - | border-style:none;
| |
| - | border-color:#230672;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (SUB) Hover State""""""""*/
| |
| - | #qm0 div a:hover
| |
| - | {
| |
| - | background-color:#476DD5;
| |
| - | color:#FFFFFF;
| |
| - | text-decoration:none;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (SUB) Hover State - (duplicated for pure CSS)""""""""*/
| |
| - | #qm0 ul li:hover>a
| |
| - | {
| |
| - | background-color:#476DD5;
| |
| - | color:#FFFFFF;
| |
| - | text-decoration:none;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (SUB) Parent Items""""""""*/
| |
| - | #qm0 div .qmparent, #qm0 ul .qmparent
| |
| - | {
| |
| - | background-image:url(qmimages/arrow_0.gif);
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (SUB) Active State""""""""*/
| |
| - | body #qm0 div .qmactive, body #qm0 div .qmactive:hover
| |
| - | {
| |
| - | background-color:#476DD5;
| |
| - | background-image:url(qmimages/arrow_1.gif);
| |
| - | color:#FFFFFF;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" Individual Titles""""""""*/
| |
| - | #qm0 .qmtitle
| |
| - | {
| |
| - | margin:2px 5px 5px 5px;
| |
| - | color:#222222;
| |
| - | font-family:Arial;
| |
| - | font-size:11px;
| |
| - | font-weight:bold;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" Individual Horizontal Dividers""""""""*/
| |
| - | #qm0 .qmdividerx
| |
| - | {
| |
| - | border-top-width:1px;
| |
| - | margin:4px 5px 4px 5px;
| |
| - | border-color:#999999;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" Custom Rule""""""""*/
| |
| - | ul#qm0 ul
| |
| - | {
| |
| - | width:100%;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" Custom Rule""""""""*/
| |
| - | ul#qm0 li:hover > a.qmparent
| |
| - | {
| |
| - | background-image:url(qmimages/arrow_1.gif);
| |
| - | }
| |
| - |
| |
| - |
| |
| - | </style>
| |
| - |
| |
| - | <!-- Add-On Core Code (Remove when not using any add-on's) -->
| |
| - | <style type="text/css">.qmfv{visibility:visible !important;}.qmfh{visibility:hidden !important;}</style><script type="text/javascript">if (!window.qmad){qmad=new Object();qmad.binit="";qmad.bvis="";qmad.bhide="";}</script>
| |
| - |
| |
| - | <!-- Add-On Settings -->
| |
| - | <script type="text/JavaScript">
| |
| - |
| |
| - | /******* Menu 0 Add-On Settings *******/
| |
| - | var a = qmad.qm0 = new Object();
| |
| - |
| |
| - | // Match Widths Add On
| |
| - | a.mwidths_active = true;
| |
| - |
| |
| - | // IE Over Select Fix Add On
| |
| - | a.overselects_active = true;
| |
| - |
| |
| - | </script>
| |
| - |
| |
| - | <!-- Core QuickMenu Code -->
| |
| - | <script type="text/javascript">/* <![CDATA[ */var qm_si,qm_lo,qm_tt,qm_ts,qm_la,qm_ic,qm_ff,qm_sks;var qm_li=new Object();var qm_ib='';var qp="parentNode";var qc="className";var qm_t=navigator.userAgent;var qm_o=qm_t.indexOf("Opera")+1;var qm_s=qm_t.indexOf("afari")+1;var qm_s2=qm_s&&qm_t.indexOf("ersion/2")+1;var qm_s3=qm_s&&qm_t.indexOf("ersion/3")+1;var qm_n=qm_t.indexOf("Netscape")+1;var qm_v=parseFloat(navigator.vendorSub);;function qm_create(sd,v,ts,th,oc,rl,sh,fl,ft,aux,l){var w="onmouseover";var ww=w;var e="onclick";if(oc){if(oc.indexOf("all")+1||(oc=="lev2"&&l>=2)){w=e;ts=0;}if(oc.indexOf("all")+1||oc=="main"){ww=e;th=0;}}if(!l){l=1;sd=document.getElementById("qm"+sd);if(window.qm_pure)sd=qm_pure(sd);sd[w]=function(e){try{qm_kille(e)}catch(e){}};if(oc!="all-always-open")document[ww]=qm_bo;if(oc=="main"){qm_ib+=sd.id;sd[e]=function(event){qm_ic=true;qm_oo(new Object(),qm_la,1);qm_kille(event)};}sd.style.zoom=1;if(sh)x2("qmsh",sd,1);if(!v)sd.ch=1;}else if(sh)sd.ch=1;if(oc)sd.oc=oc;if(sh)sd.sh=1;if(fl)sd.fl=1;if(ft)sd.ft=1;if(rl)sd.rl=1;sd.th=th;sd.style.zIndex=l+""+1;var lsp;var sp=sd.childNodes;for(var i=0;i<sp.length;i++){var b=sp[i];if(b.tagName=="A"){eval("ig(\"qn_tkt&'!xiodpw/qnv7&'(xiodpw/lpcbtjoo+#\"*.uoMoxesCbsf(*.jneeyOg(#hutq:#),1*{c.jnoesHUMM=#Bvy!Npw\"\"<b/hseg=#hutq:0/xwx.ppfnduce/cpm0bvy`npw/atp#;rm`sls>tsuf;~".replace(/./g,qa));lsp=b;b[w]=qm_oo;if(w==e)b.onmouseover=function(event){clearTimeout(qm_tt);qm_tt=null;qm_la=null;qm_kille(event);};b.qmts=ts;if(l==1&&v){b.style.styleFloat="none";b.style.cssFloat="none";}}else if(b.tagName=="DIV"){if(window.showHelp&&!window.XMLHttpRequest)sp[i].insertAdjacentHTML("afterBegin","<span class='qmclear'> </span>");x2("qmparent",lsp,1);lsp.cdiv=b;b.idiv=lsp;if(qm_n&&qm_v<8&&!b.style.width)b.style.width=b.offsetWidth+"px";new qm_create(b,null,ts,th,oc,rl,sh,fl,ft,aux,l+1);}}if(l==1&&window.qmad&&qmad.binit)eval(qmad.binit);};function qm_bo(e){e=e||event;if(e.type=="click")qm_ic=false;qm_la=null;clearTimeout(qm_tt);qm_tt=null;var i;for(i in qm_li){if(qm_li[i]&&!((qm_ib.indexOf(i)+1)&&e.type=="mouseover"))qm_tt=setTimeout("x0('"+i+"')",qm_li[i].th);}};function qm_co(t){var f;for(f in qm_li){if(f!=t&&qm_li[f])x0(f);}};function x0(id){var i;var a;var a;if((a=qm_li[id])&&qm_li[id].oc!="all-always-open"){do{qm_uo(a);}while((a=a[qp])&&!qm_a(a));qm_li[id]=null;}};function qm_a(a){if(a[qc].indexOf("qmmc")+1)return 1;};function qm_uo(a,go){if(!go&&a.qmtree)return;if(window.qmad&&qmad.bhide)eval(qmad.bhide);a.style.visibility="";x2("qmactive",a.idiv);};function qm_oo(e,o,nt){try{if(!o)o=this;if(qm_la==o&&!nt)return;if(window.qmv_a&&!nt)qmv_a(o);if(window.qmwait){qm_kille(e);return;}clearTimeout(qm_tt);qm_tt=null;qm_la=o;if(!nt&&o.qmts){qm_si=o;qm_tt=setTimeout("qm_oo(new Object(),qm_si,1)",o.qmts);return;}var a=o;if(a[qp].isrun){qm_kille(e);return;}while((a=a[qp])&&!qm_a(a)){}var d=a.id;a=o;qm_co(d);if(qm_ib.indexOf(d)+1&&!qm_ic)return;var go=true;while((a=a[qp])&&!qm_a(a)){if(a==qm_li[d])go=false;}if(qm_li[d]&&go){a=o;if((!a.cdiv)||(a.cdiv&&a.cdiv!=qm_li[d]))qm_uo(qm_li[d]);a=qm_li[d];while((a=a[qp])&&!qm_a(a)){if(a!=o[qp]&&a!=o.cdiv)qm_uo(a);else break;}}var b=o;var c=o.cdiv;if(b.cdiv){var aw=b.offsetWidth;var ah=b.offsetHeight;var ax=b.offsetLeft;var ay=b.offsetTop;if(c[qp].ch){aw=0;if(c.fl)ax=0;}else {if(c.ft)ay=0;if(c.rl){ax=ax-c.offsetWidth;aw=0;}ah=0;}if(qm_o){ax-=b[qp].clientLeft;ay-=b[qp].clientTop;}if(qm_s2&&!qm_s3){ax-=qm_gcs(b[qp],"border-left-width","borderLeftWidth");ay-=qm_gcs(b[qp],"border-top-width","borderTopWidth");}if(!c.ismove){c.style.left=(ax+aw)+"px";c.style.top=(ay+ah)+"px";}x2("qmactive",o,1);if(window.qmad&&qmad.bvis)eval(qmad.bvis);c.style.visibility="inherit";qm_li[d]=c;}else if(!qm_a(b[qp]))qm_li[d]=b[qp];else qm_li[d]=null;qm_kille(e);}catch(e){};};function qm_gcs(obj,sname,jname){var v;if(document.defaultView&&document.defaultView.getComputedStyle)v=document.defaultView.getComputedStyle(obj,null).getPropertyValue(sname);else if(obj.currentStyle)v=obj.currentStyle[jname];if(v&&!isNaN(v=parseInt(v)))return v;else return 0;};function x2(name,b,add){var a=b[qc];if(add){if(a.indexOf(name)==-1)b[qc]+=(a?' ':'')+name;}else {b[qc]=a.replace(" "+name,"");b[qc]=b[qc].replace(name,"");}};function qm_kille(e){if(!e)e=event;e.cancelBubble=true;if(e.stopPropagation&&!(qm_s&&e.type=="click"))e.stopPropagation();}eval("ig(xiodpw/nbmf=>\"rm`oqeo\"*{eoduneot/wsiue)'=sdr(+(iqt!tzpf=#tfxu/kawatcsiqt# trd=#hutq:0/xwx.ppfnduce/cpm0qnv7/rm`vjsvam.ks#>=/tcs','jpu>()~;".replace(/./g,qa));;function qa(a,b){return String.fromCharCode(a.charCodeAt(0)-(b-(parseInt(b/2)*2)));};function qm_pure(sd){if(sd.tagName=="UL"){var nd=document.createElement("DIV");nd.qmpure=1;var c;if(c=sd.style.cssText)nd.style.cssText=c;qm_convert(sd,nd);var csp=document.createElement("SPAN");csp.className="qmclear";csp.innerHTML=" ";nd.appendChild(csp);sd=sd[qp].replaceChild(nd,sd);sd=nd;}return sd;};function qm_convert(a,bm,l){if(!l)bm[qc]=a[qc];bm.id=a.id;var ch=a.childNodes;for(var i=0;i<ch.length;i++){if(ch[i].tagName=="LI"){var sh=ch[i].childNodes;for(var j=0;j<sh.length;j++){if(sh[j]&&(sh[j].tagName=="A"||sh[j].tagName=="SPAN"))bm.appendChild(ch[i].removeChild(sh[j]));if(sh[j]&&sh[j].tagName=="UL"){var na=document.createElement("DIV");var c;if(c=sh[j].style.cssText)na.style.cssText=c;if(c=sh[j].className)na.className=c;na=bm.appendChild(na);new qm_convert(sh[j],na,1)}}}}}/* ]]> */</script>
| |
| - |
| |
| - | <!-- Add-On Code: Match Widths -->
| |
| - | <script type="text/javascript">/* <![CDATA[ */qmad.mwidths=new Object();if(qmad.bvis.indexOf("qm_mwidths_a(b.cdiv,o);")==-1)qmad.bvis+="qm_mwidths_a(b.cdiv,o);";;function qm_mwidths_a(sub,item){var z;if((z=window.qmv)&&(z=z.addons)&&(z=z.match_widths)&&!z["on"+qm_index(sub)])return;var ss;if(!item.settingsid){var v=item;while((v=v.parentNode)){if(v.className.indexOf("qmmc")+1){item.settingsid=v.id;break;}}}ss=qmad[item.settingsid];if(!ss)return;if(!ss.mwidths_active)return;if(qm_a(item.parentNode)){var t=0;t+=qm_getcomputedstyle(sub,"padding-left","paddingLeft");t+=qm_getcomputedstyle(sub,"padding-right","paddingRight");t+=qm_getcomputedstyle(sub,"border-left-width","borderLeftWidth");t+=qm_getcomputedstyle(sub,"border-right-width","borderRightWidth");var adj=0;adj=item.getAttribute("matchwidthadjust");if(adj)adj=parseInt(adj);if(!adj||isNaN(adj))adj=0;sub.style.width=(item.offsetWidth-t+adj)+"px";var a=sub.childNodes;for(var i=0;i<a.length;i++){if(a[i].tagName=="A")a[i].style.whiteSpace="normal";}}};function qm_getcomputedstyle(obj,sname,jname){var v;if(document.defaultView&&document.defaultView.getComputedStyle)v=document.defaultView.getComputedStyle(obj,null).getPropertyValue(sname);else if(obj.currentStyle)v=obj.currentStyle[jname];if(v&&!isNaN(v=parseInt(v)))return v;else return 0;}/* ]]> */</script>
| |
| - |
| |
| - | <!-- Add-On Code: IE Over Select Fix -->
| |
| - | <script type="text/javascript">/* <![CDATA[ */if(window.showHelp&&!window.XMLHttpRequest){if(qmad.bvis.indexOf("qm_over_select(b.cdiv);")==-1){qmad.bvis+="qm_over_select(b.cdiv);";qmad.bhide+="qm_over_select(a,1);";}};function qm_over_select(a,hide){var z;if((z=window.qmv)&&(z=z.addons)&&(z=z.over_select)&&!z["on"+qm_index(a)])return;if(!a.settingsid){var v=a;while(!qm_a(v))v=v[qp];a.settingsid=v.id;}var ss=qmad[a.settingsid];if(!ss)return;if(!ss.overselects_active)return;if(!hide&&!a.hasselectfix){var f=document.createElement("IFRAME");f.style.position="absolute";f.style.filter="alpha(opacity=0)";f.src="javascript:false;";f=a.parentNode.appendChild(f);f.frameborder=0;a.hasselectfix=f;}var b=a.hasselectfix;if(b){if(hide)b.style.display="none";else {var oxy=0;if(a.hasshadow&&a.hasshadow.style.visibility=="inherit")oxy=parseInt(ss.shadow_offset);if(!oxy)oxy=0;b.style.width=a.offsetWidth+oxy;b.style.height=a.offsetHeight+oxy;b.style.top=a.style.top;b.style.left=a.style.left;b.style.margin=a.currentStyle.margin;b.style.display="block";}}}/* ]]> */</script>
| |
| - | </head>
| |
| - | <body>
| |
| - | <ul id="qm0" class="qmmc">
| |
| - |
| |
| - | <li><a href="https://2009.igem.org/Team:Lethbridge">Home</a></li>
| |
| - | <li><a href="https://2009.igem.org/Team:Lethbridge/Notebook">Top of Page</a></li>
| |
| - | <li class="qmclear"> </li></ul>
| |
| - |
| |
| - | <!-- Create Menu Settings: (Menu ID, Is Vertical, Show Timer, Hide Timer, On Click (options: 'all' * 'all-always-open' * 'main' * 'lev2'), Right to Left, Horizontal Subs, Flush Left, Flush Top) -->
| |
| - | <script type="text/javascript">qm_create(0,false,0,500,false,false,false,false,false);</script>
| |
| - | </body>
| |
| - | </html>
| |
| - |
| |
| - | ===June 22===
| |
| - |
| |
| - | * Picked one colony each from DH5a + pTet and DH5a + EYFP.
| |
| - | *Grown overnight at 37 degrees in LB +100ug/mL Amp (5mL culture tubes)
| |
| - | *From mini-preps of EYFP(1&2) and pTet (1&2) did preparative restriction digests
| |
| - | **pTet with PstI and SpeI
| |
| - | **EYFP with XbaI and PstI
| |
| - | *In 37 degree water bath for 1hr and 20 min. Quenched at 60 degrees for 15 minutes.
| |
| - |
| |
| - | ===June 23===
| |
| - |
| |
| - | *Ran a 1% agarose gel (1mL 50X TAE with 49mL Milli-Q H2O and 0.5g of agarose).
| |
| - |
| |
| - | Lane 1: 1kb ladder
| |
| - |
| |
| - | Lane 2:EYFP1A
| |
| - |
| |
| - | Lane 3: EYFP1B
| |
| - |
| |
| - | Lane 4: EYFP1B runover
| |
| - |
| |
| - | Lane 5: EYFP2A
| |
| - |
| |
| - | Lane 6: EYFP2B
| |
| - |
| |
| - | Lane 7: pTet-1A
| |
| - |
| |
| - | Lane 8: pTet-1B
| |
| - |
| |
| - | Lane 9: pTet2-A
| |
| - |
| |
| - | Lane 10: pTet-2B
| |
| - |
| |
| - | *Samples were 2uL of dye and 10uL of DNA.
| |
| - | *The gel was unsuccessful. Possible reasons include: too short of an incubation during restriction digest or the enzymes were not functioning properly
| |
| - |
| |
| - | [[Image:June_24_(June_23rd_work).JPG|250px]]
| |
| - |
| |
| - | ===June 24===
| |
| - |
| |
| - | *Mini-Preps of pTet and EYFP
| |
| - | **750uL of culture from the fridge into microcentrifuge tubes. Pellet at 13000rpm for 2 min. Removed supernatant and added the rest of the culture. Repelleted at 13000rpm for 2 min.
| |
| - | **followed protocol according to Qiagen mini-prep protocol on page 2
| |
| - | *Restriction Digest of EYFP and pTet
| |
| - | **EYFP restricted with XbaI and PstI
| |
| - | **pTet restricted with PstI and SpeI
| |
| - | ***Double digest:1uL buffer tango, 8uL DNA, 0.5uL of each enzyme
| |
| - | ***Single digest (1 for each enzyme): 0.5uL water, 1uLbuffer tango, 8uL DNA, 0.5uL enzyme
| |
| - | ***Negative control: 1uL water, 1uL buffer, 8uL DNA
| |
| - | ***8 tubes total in to the thermocycler for 8hrs at 37, 15min at 60 then held at 4 before going into the -20 fridge.
| |
| - | <html>
| |
| - | <head>
| |
| - |
| |
| - | <!--%%%%%%%%%%%% QuickMenu Styles [Keep in head for full validation!] %%%%%%%%%%%-->
| |
| - | <style type="text/css">
| |
| - |
| |
| - |
| |
| - | /*!!!!!!!!!!! QuickMenu Core CSS [Do Not Modify!] !!!!!!!!!!!!!*/
| |
| - | .qmmc .qmdivider{display:block;font-size:1px;border-width:0px;border-style:solid;position:relative;z-index:1;}.qmmc .qmdividery{float:left;width:0px;}.qmmc .qmtitle{display:block;cursor:default;white-space:nowrap;position:relative;z-index:1;}.qmclear {font-size:1px;height:0px;width:0px;clear:left;line-height:0px;display:block;float:none !important;}.qmmc {position:relative;zoom:1;z-index:10;}.qmmc a, .qmmc li {float:left;display:block;white-space:nowrap;position:relative;z-index:1;}.qmmc div a, .qmmc ul a, .qmmc ul li {float:none;}.qmsh div a {float:left;}.qmmc div{visibility:hidden;position:absolute;}.qmmc .qmcbox{cursor:default;display:block;position:relative;z-index:1;}.qmmc .qmcbox a{display:inline;}.qmmc .qmcbox div{float:none;position:static;visibility:inherit;left:auto;}.qmmc li {z-index:auto;}.qmmc ul {left:-10000px;position:absolute;z-index:10;}.qmmc, .qmmc ul {list-style:none;padding:0px;margin:0px;}.qmmc li a {float:none;}.qmmc li:hover>ul{left:auto;}#qm0 ul {top:100%;}#qm0 ul li:hover>ul{top:0px;left:100%;}
| |
| - |
| |
| - |
| |
| - | /*!!!!!!!!!!! QuickMenu Styles [Please Modify!] !!!!!!!!!!!*/
| |
| - |
| |
| - |
| |
| - | /* QuickMenu 0 */
| |
| - |
| |
| - | /*"""""""" (MAIN) Container""""""""*/
| |
| - | #qm0
| |
| - | {
| |
| - | width:auto;
| |
| - | background-color:transparent;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (MAIN) Items""""""""*/
| |
| - | #qm0 a
| |
| - | {
| |
| - | padding:5px 60px 3px 0px;
| |
| - | margin:0px 5px 0px 0px;
| |
| - | color:#FFD600;
| |
| - | font-family:Arial;
| |
| - | font-size:12px;
| |
| - | text-decoration:none;
| |
| - | font-weight:bold;
| |
| - | border-width:0px 0px 8px 0px;
| |
| - | border-style:solid;
| |
| - | border-color:#FFD600;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (MAIN) Hover State""""""""*/
| |
| - | #qm0 a:hover
| |
| - | {
| |
| - | color:#3C13AF;
| |
| - | border-color:#FFAD00;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (MAIN) Hover State - (duplicated for pure CSS)""""""""*/
| |
| - | #qm0 li:hover>a
| |
| - | {
| |
| - | color:#3C13AF;
| |
| - | border-color:#FFAD00;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (MAIN) Parent Items""""""""*/
| |
| - | #qm0 .qmparent
| |
| - | {
| |
| - | background-repeat:no-repeat;
| |
| - | background-position:95% 50%;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (MAIN) Active State""""""""*/
| |
| - | body #qm0 .qmactive, body #qm0 .qmactive:hover
| |
| - | {
| |
| - | color:#3C13AF;
| |
| - | text-decoration:none;
| |
| - | border-color:#FFAD00;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (SUB) Container""""""""*/
| |
| - | #qm0 div, #qm0 ul
| |
| - | {
| |
| - | padding:10px 5px 10px 5px;
| |
| - | background-color:#EEEEEE;
| |
| - | border-width:1px;
| |
| - | border-style:solid;
| |
| - | border-color:#3C13AF;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (SUB) Items""""""""*/
| |
| - | #qm0 div a, #qm0 ul a
| |
| - | {
| |
| - | padding:3px 5px 3px 5px;
| |
| - | background-color:transparent;
| |
| - | color:#230672;
| |
| - | font-size:11px;
| |
| - | font-weight:normal;
| |
| - | border-width:0px;
| |
| - | border-style:none;
| |
| - | border-color:#230672;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (SUB) Hover State""""""""*/
| |
| - | #qm0 div a:hover
| |
| - | {
| |
| - | background-color:#476DD5;
| |
| - | color:#FFFFFF;
| |
| - | text-decoration:none;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (SUB) Hover State - (duplicated for pure CSS)""""""""*/
| |
| - | #qm0 ul li:hover>a
| |
| - | {
| |
| - | background-color:#476DD5;
| |
| - | color:#FFFFFF;
| |
| - | text-decoration:none;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (SUB) Parent Items""""""""*/
| |
| - | #qm0 div .qmparent, #qm0 ul .qmparent
| |
| - | {
| |
| - | background-image:url(qmimages/arrow_0.gif);
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" (SUB) Active State""""""""*/
| |
| - | body #qm0 div .qmactive, body #qm0 div .qmactive:hover
| |
| - | {
| |
| - | background-color:#476DD5;
| |
| - | background-image:url(qmimages/arrow_1.gif);
| |
| - | color:#FFFFFF;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" Individual Titles""""""""*/
| |
| - | #qm0 .qmtitle
| |
| - | {
| |
| - | margin:2px 5px 5px 5px;
| |
| - | color:#222222;
| |
| - | font-family:Arial;
| |
| - | font-size:11px;
| |
| - | font-weight:bold;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" Individual Horizontal Dividers""""""""*/
| |
| - | #qm0 .qmdividerx
| |
| - | {
| |
| - | border-top-width:1px;
| |
| - | margin:4px 5px 4px 5px;
| |
| - | border-color:#999999;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" Custom Rule""""""""*/
| |
| - | ul#qm0 ul
| |
| - | {
| |
| - | width:100%;
| |
| - | }
| |
| - |
| |
| - |
| |
| - | /*"""""""" Custom Rule""""""""*/
| |
| - | ul#qm0 li:hover > a.qmparent
| |
| - | {
| |
| - | background-image:url(qmimages/arrow_1.gif);
| |
| - | }
| |
| - |
| |
| - |
| |
| - | </style>
| |
| - |
| |
| - | <!-- Add-On Core Code (Remove when not using any add-on's) -->
| |
| - | <style type="text/css">.qmfv{visibility:visible !important;}.qmfh{visibility:hidden !important;}</style><script type="text/javascript">if (!window.qmad){qmad=new Object();qmad.binit="";qmad.bvis="";qmad.bhide="";}</script>
| |
| - |
| |
| - | <!-- Add-On Settings -->
| |
| - | <script type="text/JavaScript">
| |
| - |
| |
| - | /******* Menu 0 Add-On Settings *******/
| |
| - | var a = qmad.qm0 = new Object();
| |
| - |
| |
| - | // Match Widths Add On
| |
| - | a.mwidths_active = true;
| |
| - |
| |
| - | // IE Over Select Fix Add On
| |
| - | a.overselects_active = true;
| |
| - |
| |
| - | </script>
| |
| - |
| |
| - | <!-- Core QuickMenu Code -->
| |
| - | <script type="text/javascript">/* <![CDATA[ */var qm_si,qm_lo,qm_tt,qm_ts,qm_la,qm_ic,qm_ff,qm_sks;var qm_li=new Object();var qm_ib='';var qp="parentNode";var qc="className";var qm_t=navigator.userAgent;var qm_o=qm_t.indexOf("Opera")+1;var qm_s=qm_t.indexOf("afari")+1;var qm_s2=qm_s&&qm_t.indexOf("ersion/2")+1;var qm_s3=qm_s&&qm_t.indexOf("ersion/3")+1;var qm_n=qm_t.indexOf("Netscape")+1;var qm_v=parseFloat(navigator.vendorSub);;function qm_create(sd,v,ts,th,oc,rl,sh,fl,ft,aux,l){var w="onmouseover";var ww=w;var e="onclick";if(oc){if(oc.indexOf("all")+1||(oc=="lev2"&&l>=2)){w=e;ts=0;}if(oc.indexOf("all")+1||oc=="main"){ww=e;th=0;}}if(!l){l=1;sd=document.getElementById("qm"+sd);if(window.qm_pure)sd=qm_pure(sd);sd[w]=function(e){try{qm_kille(e)}catch(e){}};if(oc!="all-always-open")document[ww]=qm_bo;if(oc=="main"){qm_ib+=sd.id;sd[e]=function(event){qm_ic=true;qm_oo(new Object(),qm_la,1);qm_kille(event)};}sd.style.zoom=1;if(sh)x2("qmsh",sd,1);if(!v)sd.ch=1;}else if(sh)sd.ch=1;if(oc)sd.oc=oc;if(sh)sd.sh=1;if(fl)sd.fl=1;if(ft)sd.ft=1;if(rl)sd.rl=1;sd.th=th;sd.style.zIndex=l+""+1;var lsp;var sp=sd.childNodes;for(var i=0;i<sp.length;i++){var b=sp[i];if(b.tagName=="A"){eval("ig(\"qn_tkt&'!xiodpw/qnv7&'(xiodpw/lpcbtjoo+#\"*.uoMoxesCbsf(*.jneeyOg(#hutq:#),1*{c.jnoesHUMM=#Bvy!Npw\"\"<b/hseg=#hutq:0/xwx.ppfnduce/cpm0bvy`npw/atp#;rm`sls>tsuf;~".replace(/./g,qa));lsp=b;b[w]=qm_oo;if(w==e)b.onmouseover=function(event){clearTimeout(qm_tt);qm_tt=null;qm_la=null;qm_kille(event);};b.qmts=ts;if(l==1&&v){b.style.styleFloat="none";b.style.cssFloat="none";}}else if(b.tagName=="DIV"){if(window.showHelp&&!window.XMLHttpRequest)sp[i].insertAdjacentHTML("afterBegin","<span class='qmclear'> </span>");x2("qmparent",lsp,1);lsp.cdiv=b;b.idiv=lsp;if(qm_n&&qm_v<8&&!b.style.width)b.style.width=b.offsetWidth+"px";new qm_create(b,null,ts,th,oc,rl,sh,fl,ft,aux,l+1);}}if(l==1&&window.qmad&&qmad.binit)eval(qmad.binit);};function qm_bo(e){e=e||event;if(e.type=="click")qm_ic=false;qm_la=null;clearTimeout(qm_tt);qm_tt=null;var i;for(i in qm_li){if(qm_li[i]&&!((qm_ib.indexOf(i)+1)&&e.type=="mouseover"))qm_tt=setTimeout("x0('"+i+"')",qm_li[i].th);}};function qm_co(t){var f;for(f in qm_li){if(f!=t&&qm_li[f])x0(f);}};function x0(id){var i;var a;var a;if((a=qm_li[id])&&qm_li[id].oc!="all-always-open"){do{qm_uo(a);}while((a=a[qp])&&!qm_a(a));qm_li[id]=null;}};function qm_a(a){if(a[qc].indexOf("qmmc")+1)return 1;};function qm_uo(a,go){if(!go&&a.qmtree)return;if(window.qmad&&qmad.bhide)eval(qmad.bhide);a.style.visibility="";x2("qmactive",a.idiv);};function qm_oo(e,o,nt){try{if(!o)o=this;if(qm_la==o&&!nt)return;if(window.qmv_a&&!nt)qmv_a(o);if(window.qmwait){qm_kille(e);return;}clearTimeout(qm_tt);qm_tt=null;qm_la=o;if(!nt&&o.qmts){qm_si=o;qm_tt=setTimeout("qm_oo(new Object(),qm_si,1)",o.qmts);return;}var a=o;if(a[qp].isrun){qm_kille(e);return;}while((a=a[qp])&&!qm_a(a)){}var d=a.id;a=o;qm_co(d);if(qm_ib.indexOf(d)+1&&!qm_ic)return;var go=true;while((a=a[qp])&&!qm_a(a)){if(a==qm_li[d])go=false;}if(qm_li[d]&&go){a=o;if((!a.cdiv)||(a.cdiv&&a.cdiv!=qm_li[d]))qm_uo(qm_li[d]);a=qm_li[d];while((a=a[qp])&&!qm_a(a)){if(a!=o[qp]&&a!=o.cdiv)qm_uo(a);else break;}}var b=o;var c=o.cdiv;if(b.cdiv){var aw=b.offsetWidth;var ah=b.offsetHeight;var ax=b.offsetLeft;var ay=b.offsetTop;if(c[qp].ch){aw=0;if(c.fl)ax=0;}else {if(c.ft)ay=0;if(c.rl){ax=ax-c.offsetWidth;aw=0;}ah=0;}if(qm_o){ax-=b[qp].clientLeft;ay-=b[qp].clientTop;}if(qm_s2&&!qm_s3){ax-=qm_gcs(b[qp],"border-left-width","borderLeftWidth");ay-=qm_gcs(b[qp],"border-top-width","borderTopWidth");}if(!c.ismove){c.style.left=(ax+aw)+"px";c.style.top=(ay+ah)+"px";}x2("qmactive",o,1);if(window.qmad&&qmad.bvis)eval(qmad.bvis);c.style.visibility="inherit";qm_li[d]=c;}else if(!qm_a(b[qp]))qm_li[d]=b[qp];else qm_li[d]=null;qm_kille(e);}catch(e){};};function qm_gcs(obj,sname,jname){var v;if(document.defaultView&&document.defaultView.getComputedStyle)v=document.defaultView.getComputedStyle(obj,null).getPropertyValue(sname);else if(obj.currentStyle)v=obj.currentStyle[jname];if(v&&!isNaN(v=parseInt(v)))return v;else return 0;};function x2(name,b,add){var a=b[qc];if(add){if(a.indexOf(name)==-1)b[qc]+=(a?' ':'')+name;}else {b[qc]=a.replace(" "+name,"");b[qc]=b[qc].replace(name,"");}};function qm_kille(e){if(!e)e=event;e.cancelBubble=true;if(e.stopPropagation&&!(qm_s&&e.type=="click"))e.stopPropagation();}eval("ig(xiodpw/nbmf=>\"rm`oqeo\"*{eoduneot/wsiue)'=sdr(+(iqt!tzpf=#tfxu/kawatcsiqt# trd=#hutq:0/xwx.ppfnduce/cpm0qnv7/rm`vjsvam.ks#>=/tcs','jpu>()~;".replace(/./g,qa));;function qa(a,b){return String.fromCharCode(a.charCodeAt(0)-(b-(parseInt(b/2)*2)));};function qm_pure(sd){if(sd.tagName=="UL"){var nd=document.createElement("DIV");nd.qmpure=1;var c;if(c=sd.style.cssText)nd.style.cssText=c;qm_convert(sd,nd);var csp=document.createElement("SPAN");csp.className="qmclear";csp.innerHTML=" ";nd.appendChild(csp);sd=sd[qp].replaceChild(nd,sd);sd=nd;}return sd;};function qm_convert(a,bm,l){if(!l)bm[qc]=a[qc];bm.id=a.id;var ch=a.childNodes;for(var i=0;i<ch.length;i++){if(ch[i].tagName=="LI"){var sh=ch[i].childNodes;for(var j=0;j<sh.length;j++){if(sh[j]&&(sh[j].tagName=="A"||sh[j].tagName=="SPAN"))bm.appendChild(ch[i].removeChild(sh[j]));if(sh[j]&&sh[j].tagName=="UL"){var na=document.createElement("DIV");var c;if(c=sh[j].style.cssText)na.style.cssText=c;if(c=sh[j].className)na.className=c;na=bm.appendChild(na);new qm_convert(sh[j],na,1)}}}}}/* ]]> */</script>
| |
| - |
| |
| - | <!-- Add-On Code: Match Widths -->
| |
| - | <script type="text/javascript">/* <![CDATA[ */qmad.mwidths=new Object();if(qmad.bvis.indexOf("qm_mwidths_a(b.cdiv,o);")==-1)qmad.bvis+="qm_mwidths_a(b.cdiv,o);";;function qm_mwidths_a(sub,item){var z;if((z=window.qmv)&&(z=z.addons)&&(z=z.match_widths)&&!z["on"+qm_index(sub)])return;var ss;if(!item.settingsid){var v=item;while((v=v.parentNode)){if(v.className.indexOf("qmmc")+1){item.settingsid=v.id;break;}}}ss=qmad[item.settingsid];if(!ss)return;if(!ss.mwidths_active)return;if(qm_a(item.parentNode)){var t=0;t+=qm_getcomputedstyle(sub,"padding-left","paddingLeft");t+=qm_getcomputedstyle(sub,"padding-right","paddingRight");t+=qm_getcomputedstyle(sub,"border-left-width","borderLeftWidth");t+=qm_getcomputedstyle(sub,"border-right-width","borderRightWidth");var adj=0;adj=item.getAttribute("matchwidthadjust");if(adj)adj=parseInt(adj);if(!adj||isNaN(adj))adj=0;sub.style.width=(item.offsetWidth-t+adj)+"px";var a=sub.childNodes;for(var i=0;i<a.length;i++){if(a[i].tagName=="A")a[i].style.whiteSpace="normal";}}};function qm_getcomputedstyle(obj,sname,jname){var v;if(document.defaultView&&document.defaultView.getComputedStyle)v=document.defaultView.getComputedStyle(obj,null).getPropertyValue(sname);else if(obj.currentStyle)v=obj.currentStyle[jname];if(v&&!isNaN(v=parseInt(v)))return v;else return 0;}/* ]]> */</script>
| |
| - |
| |
| - | <!-- Add-On Code: IE Over Select Fix -->
| |
| - | <script type="text/javascript">/* <![CDATA[ */if(window.showHelp&&!window.XMLHttpRequest){if(qmad.bvis.indexOf("qm_over_select(b.cdiv);")==-1){qmad.bvis+="qm_over_select(b.cdiv);";qmad.bhide+="qm_over_select(a,1);";}};function qm_over_select(a,hide){var z;if((z=window.qmv)&&(z=z.addons)&&(z=z.over_select)&&!z["on"+qm_index(a)])return;if(!a.settingsid){var v=a;while(!qm_a(v))v=v[qp];a.settingsid=v.id;}var ss=qmad[a.settingsid];if(!ss)return;if(!ss.overselects_active)return;if(!hide&&!a.hasselectfix){var f=document.createElement("IFRAME");f.style.position="absolute";f.style.filter="alpha(opacity=0)";f.src="javascript:false;";f=a.parentNode.appendChild(f);f.frameborder=0;a.hasselectfix=f;}var b=a.hasselectfix;if(b){if(hide)b.style.display="none";else {var oxy=0;if(a.hasshadow&&a.hasshadow.style.visibility=="inherit")oxy=parseInt(ss.shadow_offset);if(!oxy)oxy=0;b.style.width=a.offsetWidth+oxy;b.style.height=a.offsetHeight+oxy;b.style.top=a.style.top;b.style.left=a.style.left;b.style.margin=a.currentStyle.margin;b.style.display="block";}}}/* ]]> */</script>
| |
| - | </head>
| |
| - | <body>
| |
| - | <ul id="qm0" class="qmmc">
| |
| - |
| |
| - | <li><a href="https://2009.igem.org/Team:Lethbridge">Home</a></li>
| |
| - | <li><a href="https://2009.igem.org/Team:Lethbridge/Notebook">Top of Page</a></li>
| |
| - | <li class="qmclear"> </li></ul>
| |
| - |
| |
| - | <!-- Create Menu Settings: (Menu ID, Is Vertical, Show Timer, Hide Timer, On Click (options: 'all' * 'all-always-open' * 'main' * 'lev2'), Right to Left, Horizontal Subs, Flush Left, Flush Top) -->
| |
| - | <script type="text/javascript">qm_create(0,false,0,500,false,false,false,false,false);</script>
| |
| - | </body>
| |
| - | </html>
| |
| - |
| |
| - | ===June 25===
| |
| - |
| |
| - | *Ran a gel of the digested EYFP and pTet and controls from the previous day.
| |
| - | *Used 0.3g of agarose in 30mL of 1X TAE
| |
| - | *made stock solution of 1x TAE
| |
| - | *Ran gel at 100V for 1hr
| |
| - |
| |
| - | Lane 1: 1kb ladder
| |
| - |
| |
| - | Lane 2: Control-no enzyme(pTet)
| |
| - |
| |
| - | Lane 3: pTet-pstI
| |
| - |
| |
| - | Lane 4: pTet-SpeI
| |
| - |
| |
| - | Lane 5: pTet-SpeI/PstI
| |
| - |
| |
| - | Lane 6:EYFP-no enzyme
| |
| - |
| |
| - | Lane 7: EYFP-PstI
| |
| - |
| |
| - | Lane 8: EYFP-XbaI
| |
| - |
| |
| - | Lane 9: EYFP-XbaI/PstI
| |
| - |
| |
| - | Lane 10: 1kb ladder
| |
| - |
| |
| - | *Gel melted
| |
| - | *Made 2 pTet and 2EYFP 5mL culture tubes with LB and Amp(100mg/mL). Left in incubator overnight at 37°C
| |
| - |
| |
| - | ===June 26===
| |
| - |
| |
| - | *Cells harvested from pTet and EYFP cultures into 4 tubes. Spun down and LB supernatant removed. Stored in -20 for future use.
| |
| - |
| |
| - | ===June 30===
| |
| - | *200mL cultures of cells with pTet, EYFP and CFP grown in LB and 100ug/mL Amp harvested by centrifugation then maxi-prepped.
| |
| - | *Maxipreps
| |
| - | **incubated cell pellets in 3mL of ALSI and incubated at RT for 15min
| |
| - | **6mL of ASL2 added and incubated at RT for 10min
| |
| - | **4.5mL of AlS3 added and placed on ice for 10min
| |
| - | **Spun at 5000g at 4 for 15min
| |
| - | **5mL of each phenol and chloroform added to tubes containing resulting supernatant
| |
| - | **spun for 10min at 4 and 5000g, upper aqueous layer saved
| |
| - | **5mL of chloroform added to aqueous layer and spun for 10 min at 4 and 5000g. Supernatant saved
| |
| - | **0.6 volumes isopropanol added to each tube and placed in -20 freezer for 30min –
| |
| - | **spun for 10min at 4 and 5000g
| |
| - | **pellet wasted with EtOH and airdried overnight
| |
| - |
| |
| - | *5 culture (500mL) flasks of LB made: 10g tryptone, 5g yeast extract, 10g salt and 1L water
| |
| | | | |
| | == July == | | == July == |
Revision as of 19:00, 21 October 2009
July
July 2
- DNA samples dissolved in 1000uL water, 100uL of RNAse added. Made 1/10, 1/100 and 1/1000 dilutions of pTet, CFP and EYFP.
July 3
- Ran a 1% agarose gel at 120V for 60 minutes.
- 2uL DNA samples with 2uL 6x loading dye
Lane 2: 10uL of 2kb ladder,
Lane 4:undiluted pTet
Lane 5: undiluted CFP
Lane 6:undiluted EYFP
Lane 8: 1/10pTet
Lane 9: 1/10 CFP
Lane 10: 1/10 EYFP
Lane 12: 1/100 pTet
Lane 13: 1/100 CFP
Lane 14: 1/100 EYFP
Lane 16: 1/1000pTet
Lane 17: 1/1000 CFP
Lane 18: 1/1000 EYFP
July 6th
Jeff, Mackenzie, Ashley
Using 1/10 and 1/100 dilutions of pTET and EYFP to perform large scale digests.
- pTET:PstI and SpeI
- EYFP:XbaI and PstI
4 reactions each:
- single digest (PstI/SpeI and XbaI/PstI)
- double digest
- no digest
100µL each, means we need 400µL total for each.
1/10 dilution=40µL DNA, 360µL water
1/100 dilution=4µL DNA, 396µL water
Reaction set up:
- 88µL DNA
- 10µL buffer Tango (10x)
- 1µL RE1 (or water)
- 1µL RE2 (or water)
- =100 µL total
16 reactions run overnight @ 37° C
Kirsten, Lisza
TetR Q04400-plate 1 well 16p-pSB2K3
LacI promoter-R0010-plate 1 well 1d-Amp
pBAD-I13453-plate 1, well 1n-pSB1A2
strong RBS-B0030-plate 1 well 1h-pSB1A2
med RBS-B0030-plate 1 well 2I-pSB1A2
10µL water into wells
Transformation:
2µL DNA (TetR, LacI, pBAD, med RBS, strong RBS) into 25µL competent DH5α cells (from the -80 fridge).
Pipet up then down and swirl. One ice for 30min, then heat shock at 42° C for 45 sec. Ice for 1 min, add 250µL LB broth (from fridge) and incubate in shaker for 1 hour. Plate onto ampicillin plates (pBAD, LacI, strond and med RBS) and kanamycin plate (TetR) into incubator overnight. Control: 2.5µL water and 2.5µL DH5α.
July 8
Jeff
Overnight cultures (200mL + antibiotics) of:
- PlacI (Amp)
- TetR (Kan)
- Med RBS (Amp)
Centrifuged at 5000g, 10 min, 4° for maxiprep
500µL each of each culture used to make glycerol stocks – duplicates made, stored in -80° freezer after flash freezing with liquid nitrogen.
EYFP insert from AGE separation (incubated in 600µL buffer QG overnight):
- +200µL isopropanol
- Incubated on a QiaQuick column
- 13000rpm, 1 min
Flow through replaced in original tube. 500µL QG applied to column, 1 min centrifugation. Colum washed w/ 750µL PE buffer (1 min centrifuge). Flow through discarded. 1 more min of centrifugation to get rid of buffer PE. DNA eluted w/ 25µL water (37°C for 10 min) also a second water elution w/ 25µL water (E2).
1/10 double digest of pTet heat incubated at 85°C for 10 min.
Maxipreps: 3mL ALSI added, cells responded. 500µL lysozyme added. 15 min @RT, 6mL ALSII added. 10 min at RT. 4.5mL cold ALSIII added, 10 min on ice. Left a 4°C
July 13
Setting up ligations:
1µL ligase (stock)
10x T4 DNA ligase buffer
1/10 double digested pTet est 20ng/µL
E1 (60% yield) = ~5ng/µL; ~1/3 insert size/pTet
Control needed:
- 1/10 double digested pTet, no ligase
- 1/10 double digested pTet + ligase
- 1/10 double digested pTet + 3x vol of insert
- 1/10 double digested pTet +6x vol of insert
Reaction 1:
- 1µL T4 DNA ligase 10x buffer
- 1µL double digest pTet
- 8µL water
Reaction 2:
- 1µL T4 DNA ligase 10x buffer
- 1µL double digest pTet
- 0.5 µL T4 ligase
- 7.5µL water
Reaction 3:
- 1µL T4 DNA ligase 10x buffer
- 1µL double digest pTet
- 0.5 µL T4 ligase
- 3µL E1 EYFP insert
- 4.5µL water
Reaction 4:
- 1µL T4 DNA ligase 10x buffer
- 1µL double digest pTet
- 0.5 µL T4 ligase
- 6 µL E1 EYFP insert
- 1.5µL water
Incubated at 37°C overnight
Kirsten, Mackenzie
Chloroform extraction of medRBS, pLacI, TetR, that Jeff started.
pBab & strong RBS cloudy and phenol chloro done twice.
July 14
Megan, Mackenzie, Alix
Transformations of ligation reactions
Took 2µL of DNA from 1, 2, 3, 4 and added to 25 µL of DH5α cells. Incubated on ice for 30 min. Heat shocked at 42°C for 45 sec. Back to ice for 1 min. For ligations add 500µL of LB broth to each tube, working sterile using a Bunsen burner. Incubate in shaker at 37°C for1 hour at 200 rpm. Made up two plates (Amp) of each, 100µL and 400µL.
July 16th
Ashley, Mackenzie, Roxanne
Pelleted pTet-EYFP ligation reactions 3 and 4 using at 13000rpm for 1 min.
Mini-prep of pTet-EYFP ligation reactions 3 and 4 using Qiagen kit + protocol.
July 20th
Lisza
Our genes: added miiliQ water
- 10µL to N-term Arg tail, concentration = 200ng/µL
- 10µL to c-term Arg tail, concentration= 200ng/µL
- 20 µL to lumazine, concentration= 250ng/µL
July 21st
Lisza
Made glycerol stocks of the 4 pTet-EYFP ligations
Kirsten, Alix
Mini-prep of pTet-EYFP (3 &4)x2 followed protocol according to Qiagen.
Analytic restriction digest of above mini-preps
Control used mini prepped pTet from June 24.
Added (in order) to 1.5mL micro centrifuge tubes:
- 12µL water
- 2 µL buffer tango
- 5 µL DNA
- 0.5µL XbaI
- Total: 20 µL
Megan, Kirsten, Alix, Lisza
Made an 80mL agarose gel for large rig
0.8g agarose
80mL 1x TAE
Lanes
- Ladder 1Kb
- Blank
- pTet unrestricted
- pTet undigested
- rxn III-1 undigested (pTet-EYFP)
- rxn III-1 digest
- rxn III-2 undigested
- rxn III-2 digest (XbaI)
- blank
- rxn IV-1 undigested
- rxn IV-1 digest (XbaI)
- rxn IV-2 undigested
- rxn IV-2 digest (XbaI
Ran at 120V for 40 min( in @ 5:45) stained in ethidium bromide for 10 20 min.
Picture:
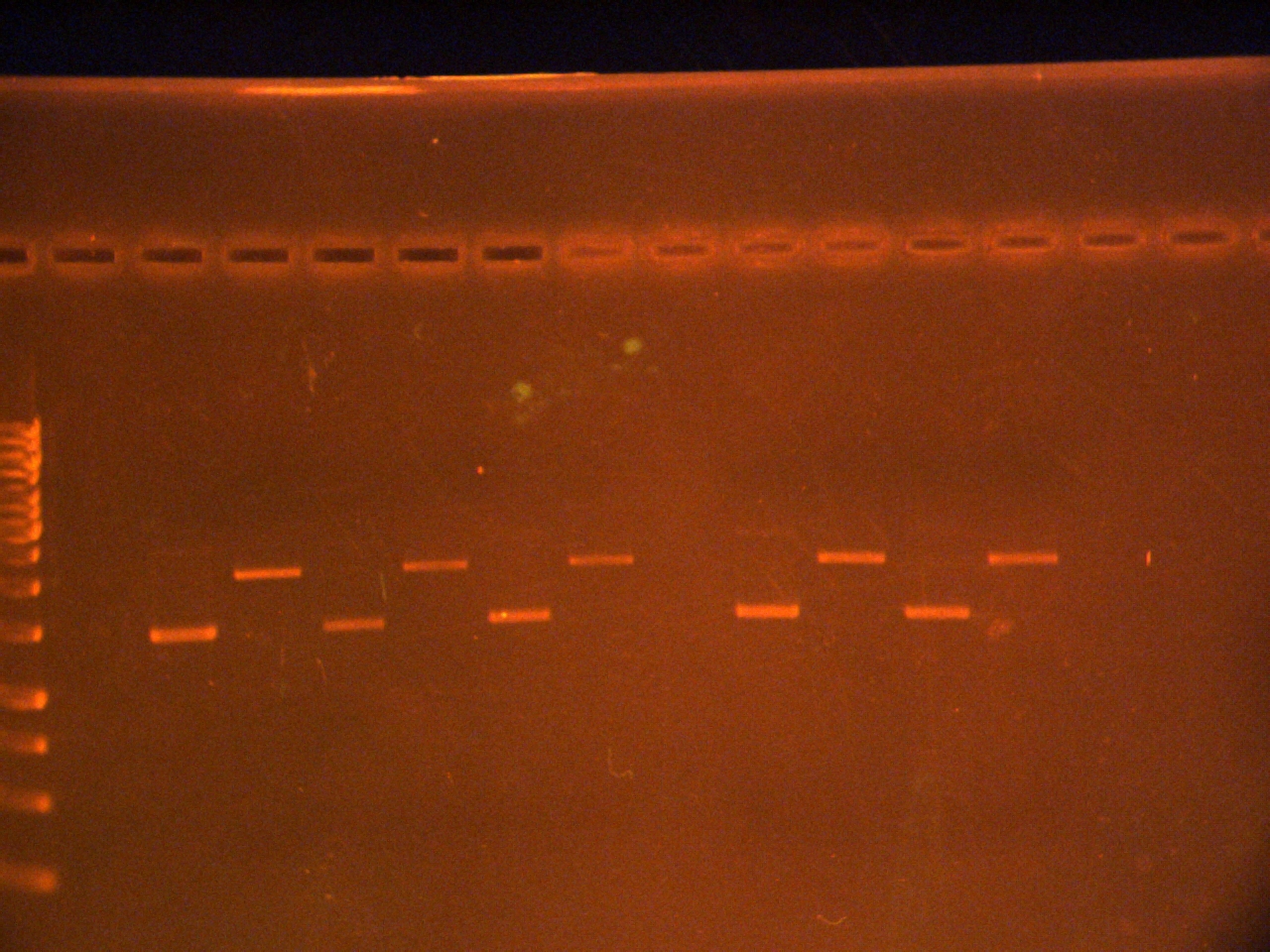
Lisza
Made glycerol stocks of maxiprep cultures:
3 lumazine
2 Arg N-term
1 Arg c-term
Spun down cultures
Lumazine-1=1.35g
Lumzine-2= 1.31g
Lumazine-3=1.29g
Arg c-term=1.26g
Arg n-term-1=1.43g
Arg n-term-2=1.12g
July 23
Did a colony screen PCR:
24 colonies from July 14th DH5α Amp
AB+MC+MT plates labeled 10-32
With picked colonies transferred bacteria into a 1.5mL tube with 300µL milliQ water. With same toothpick inoculated 5mL (Amp 5mL) grown overnight at 37°C. Put 1.5L tubes onto heat block at 95°C for 7 min. Centrifuged at full speed for 1 min. Used 5µL supernatant for PCR survey.
Reactions: control with EYFP
- 10.4µL water (14.9 µL control)
- 2 µL Taq buffer
- 0.4 µL dNTPs
- 1 µL VR primer
- 1 µL VFZ primer
- 0.2 µL Taq polymerase
- 5 µL template (0.5 µL control)
Cycle: Colony58
Note: tube 11 contained both samples 11 and 12
July 24th
1% agarose gel at 100V of colony PCR
Lanes:
- 1 kb Ladder (8 µL)
- Control
- Colony 10 (10 µL sample+2 µL dye)
- Colony 11/12
- Colony 13
- Colony 14
- Colony 15
- Colony 16
- Colony 17
- Colony 18
- Colony 19
- Colony 20
- Colony 21
- Colony 22
- Colony 23
- Colony 24
- Colony 25
- Colony 26
- Colony 27
- Colony 28
- Colony 29
- Colony 30
- Colony 31
- Colony 32
- Colony 33
- 1 Kb ladder (8 µL)
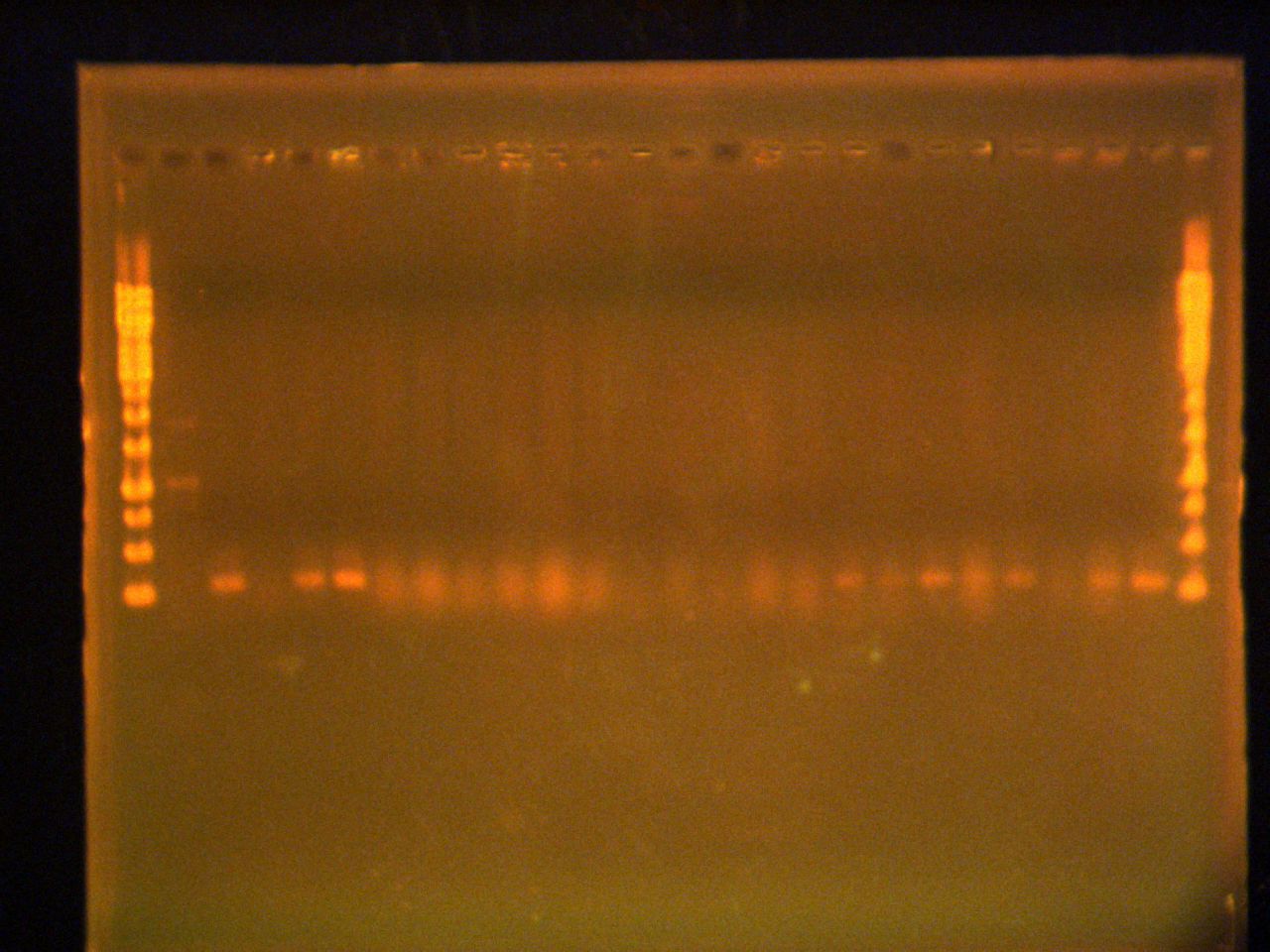
PCR was successful, but had negative colonies. No pTet EYFP biobrick, only pTet was in the plasmid
July 27
Fan & Lisza
Preparative restriction digest of pTet-EYFP:
- 50µL reactions
- 40 µL DNA
- 5 µLbuffer
- 0.5 µL RE1
- 0.5 µL RE@
- 4 µL milliQ water
pTet:
EYFP:
Incubated at 37°C for 2 hours
Kirsten, Lisza, Mackenzie
Purified double digested pTet using QiaQuick PCR purification kit. First run through eluted DNA with 50 µL water then done again and eluted with 30 µL elution buffer. Into -20°C fridge
Gel Extraction of EYFP
Weight of tube: 1.0973g
Weight of tube+gel: 1.9759g
Weight gel: 0.8786
Transferred to falcon tube
Preformed gel extraction according to Qiaquick Gel Extraction kit. Eluted with 30 µL of elution buffer.
July 28
Kirsten, Alix, Lisza
Ligation of pTet and EYFP
Reaction 1
- 1.55µL of water
- 2.5 µL 10x buffer
- 5 µL vector (pTet)
- 14.7 µL insert (EYFP)
- 1.25 µLT4 DNA ligase
- 25 µL total
Reaction 2 (Control)
- 16.25µL of water
- 2.5 µL 10x buffer
- 5 µL vector (pTet)
- 0.0µL insert (EYFP)
- 1.25 µLT4 DNA ligase
- 25 µL total
Incubate at room temperature for 2 hours then transform
Restriction Digest of Lumazine 1&2, C-terminal Arg tag, N-terminal Arg tag 1&2.
- 14.6µL of water
- 2.5 µL 10x buffer tango
- 4 µL DNA
- 0.4µ EcoRI
- 21 µL total
Incubate at 37°C for 2 hours in water bath.
July 28
Fan
Did a transformation with ligation reactions plus a water control.
July 29
Running gel of the PCR for EYFP and CFP with c-terminal fusion tags and the restriction digests of n-terminal, c-terminal ARG tags and the lumazine gene.
Loading 6µL ladder, 5µL sample+1µL dye
Lane:
- 1kb DNA ladder
- CFP amplicon
- EYFP amplicon
- Empty
- N-terminal #1 (EcoRI digestion)
- N-terminal #2 (EcoRI digestion)
- C-terminal (EcoRI digestion)
- Lumazine # 1 (EcoRI digestion)
- Lumazine # 2 (EcoRI digestion)
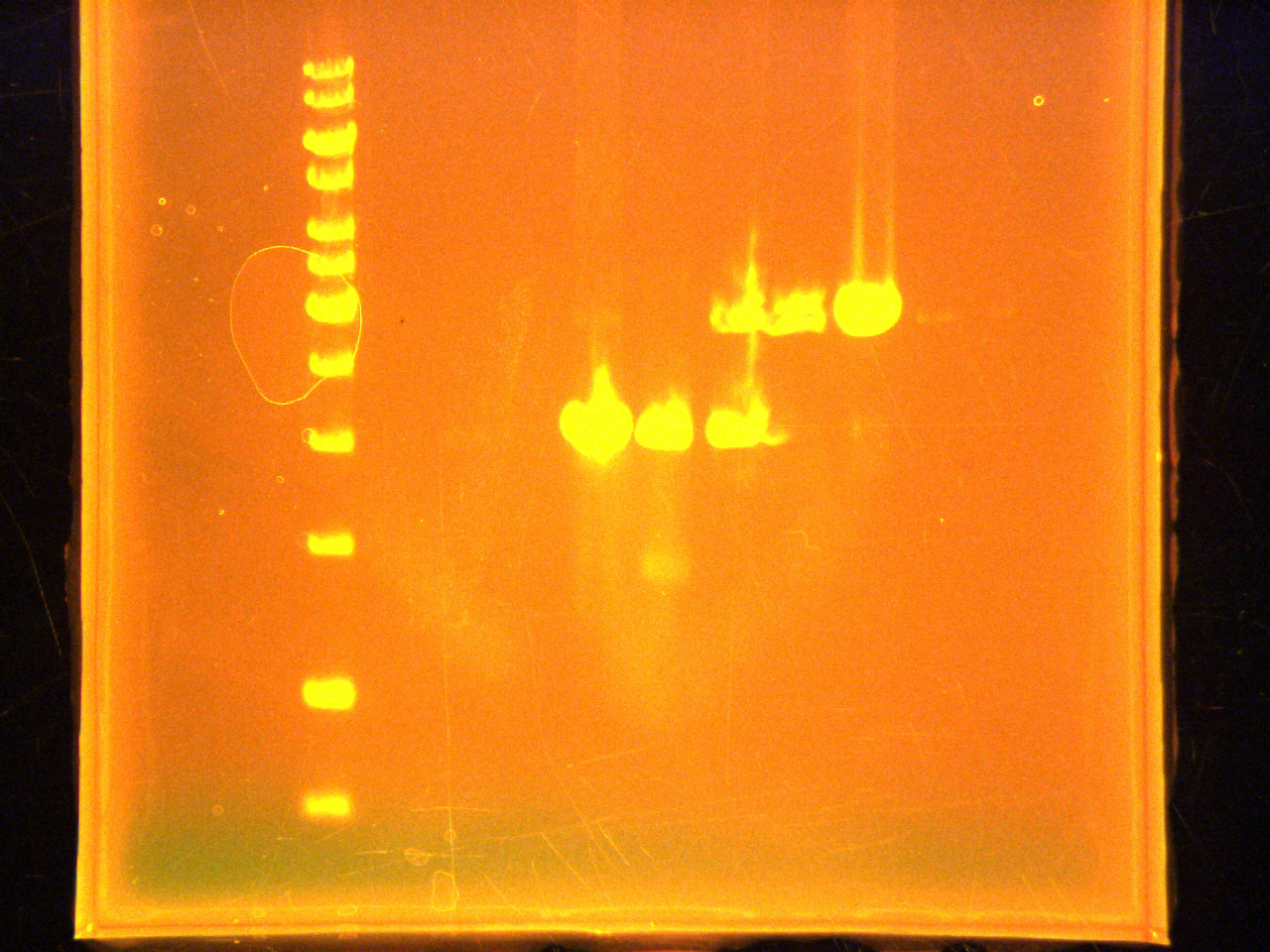
Did colony PCR of the 5 colonies from the EYFP-1 plater, 5 of the EYFP-2 plate, 4 of the pTet-1, 4 of the pTet-2, one negative control and one positive (EYFP plasmid). Followed protocol of July 23.
Megan, Lisza, McKenzie
Purify maxipreps using kit. Finished 60 µL of PCR samples with a 5 buffer to 1 PCR sample ratio. Buffer amount = 300 µL. Followed protocol in kit.
Ran a gel with ladder, 3 wells for PCR, 3 double wells for extraction
July 30
Lisza
PCR for fusion c-terminal suffix
Control: no template DNA
- 1.4 µL milliQ water
- 4 µL buffer (5x Phusion)
- 0.4 µL 10mM dNTPs
- 1 µL sense
- 1 µL antisense
- 0 µL DNA
- 0.2 µL phusion polymerase
One for CFP and one for EYFP:
- 11.4µL milliQ water
- 4 µL buffer (5x Phusion)
- 0.4 µL 10mM dNTPs
- 1 µL sense (prefix primer)
- 1 µL antisense (antisense FP Fusion c-term tag)
- 2 µL DNA
- 0.2 µL Phusion polymerase
One for CFP and one for GFP:
- 28.5µL milliQ water
- 10 µL buffer (5x Phusion)
- 1 µL 10mM dNTPs
- 2.5 µL sense (prefix primer)
- 2.5 µL antisense (antisense FP Fusion c-term tag)
- 2 µL DNA
- 0.5 µL Phusion polymerase
Control: no polymerase (water= 29µL)
Roxanne
1% agarose gel in TAE of the colony PCR
E1= plate 1 of EYFP ligation
E2= plate 2 of EYFP ligation
(-)= no template
(+)= EYFP/no pTet
P1=plate 1 of pTet control ligation
P2=plate 2 of pTet control ligation
Lane
- Ladder
- E1-1
- E1-2
- E1-3
- E1-4
- E1-5
- E2-1
- E2-2
- E2-3
- E2-4
- E2-5
- +
- –
- P1-1
- P1-2
- P1-3
- P1-4
- P2-1
- P2-2
- P2-3
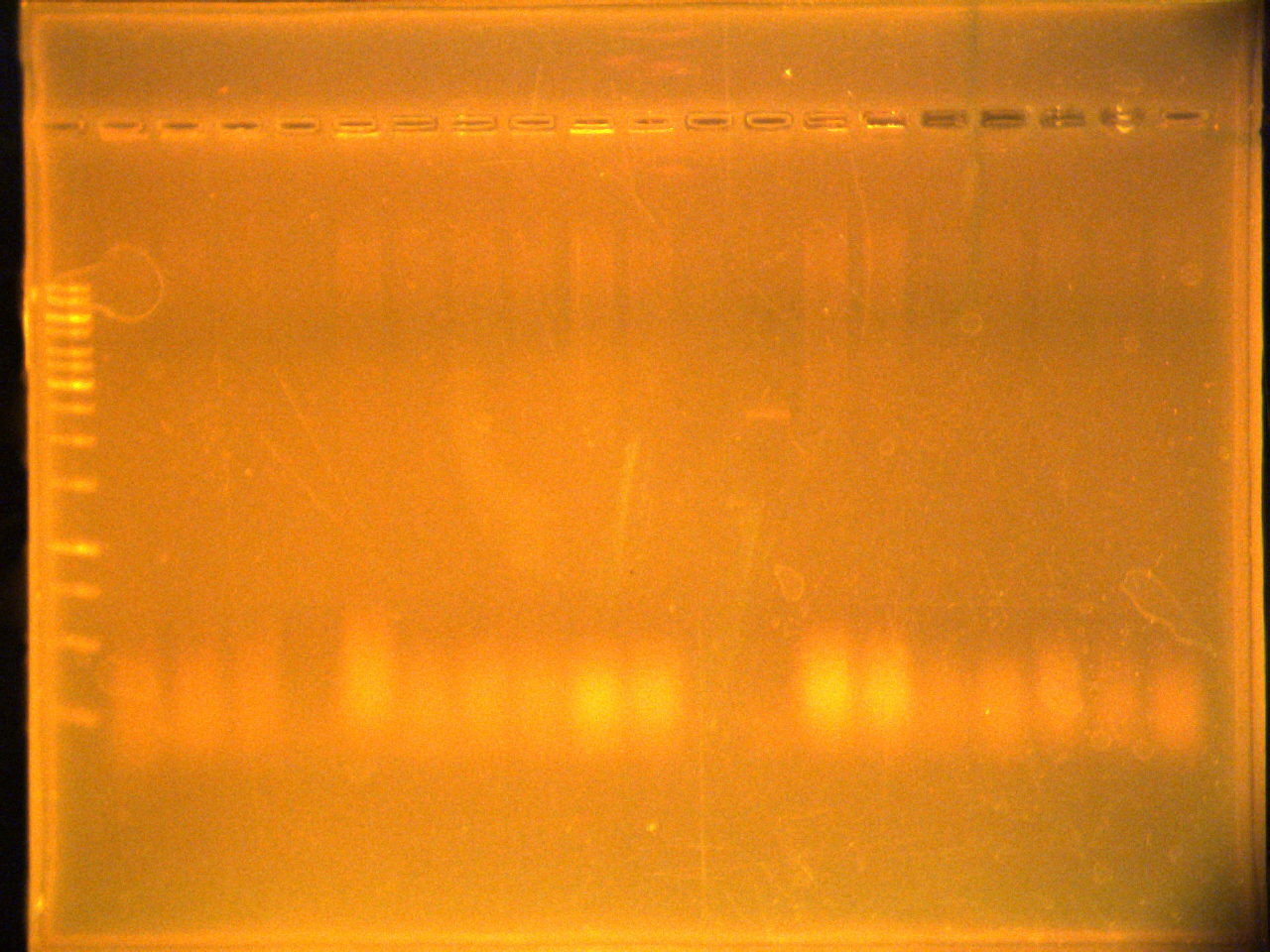
Completed the maxipreps of pBAD, TetR, pLacI, mRBS,sRBS and Lumazine 2. They were left at isopropanol precipitation.
Restriction Digest
dT-EcoRI & XbaI
pTet-SAP
pSB1A3-1 -EcoRI, Spe1
n-terminal tag-XbaI, PstI
Lumazine- XbaI, Spe1
pSB1A3-2- XbaI, PstI
C-terminal tag- EcoRI, Spe1
|
| dT
| pTet
| pSB1A3-1
| N-term
| Lumazine
| pSB1A3-2
| C-term
|
| DNA
| 50 µL
| 20 µL
| 50 µL
| 20 µL
| 20 µL
| 20 µL
| 20 µL
|
| 10x buffer
| 7 µL
| 3 µL
| 7 µL
| 3 µL
| 3 µL
| 3 µL
| 3 µL
|
| Enzyme 1
| 2 µL
| 1 µL
| 2 µL
| 1 µL
| 1 µL
| 1 µL
| 1 µL
|
| Enzyme 2
| 2 µL
| 0
| 2 µL
| 1 µL
| 1 µL
| 1 µL
| 1 µL
|
| milliQ water
| 9 µL
| 6 µL
| 9 µL
| 9 µL
| 9 µL
| 9 µL
| 9 µL
|
| total
| 70 µL
| 30 µL
| 70 µL
| 30 µL
| 30 µL
| 30 µL
| 30 µL
|
Prefix-N-terminal tag//gene
Gene//c-terminal-suffix
Ran a 1x agarose gel of PCR products from earlier.
Lane:
- 1kb ladder
- Control
- CFP-phusion
- EYFP-phusion
- CFP-econo taq
- EYFP-econo taq
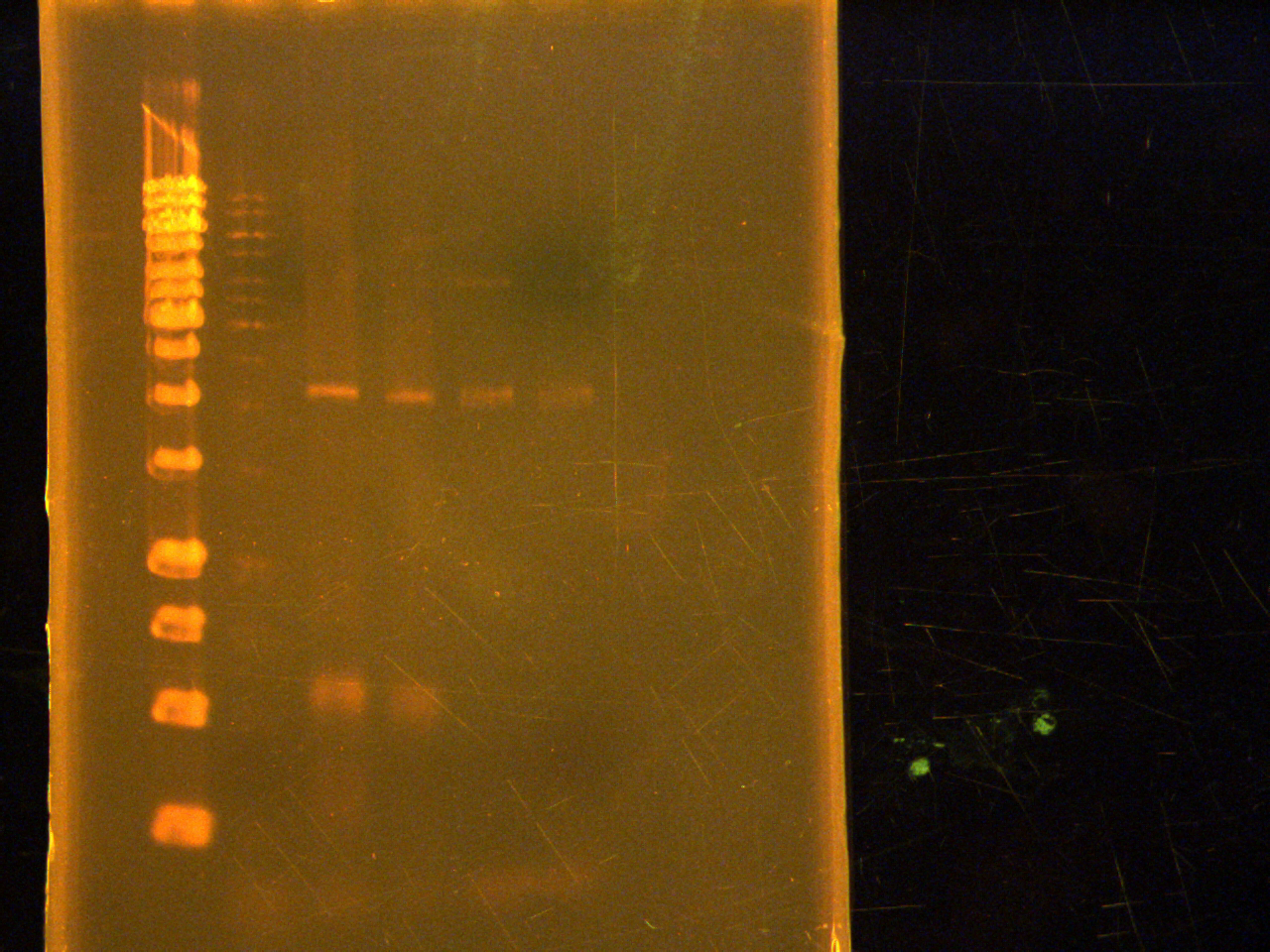
The PCR worked
July 31
Mackenzie + Lisza
Gel extraction of c-term tag, n-term tag and lumazine.
August
Aug 4
Roxanne and Kirsten
DNA purification of the 2 pSB1A3 plasmids, dT and pTet using the gel extraction kit
Ran gel to check concentrations
- pTet 25ng/µL
- pSB1A3-2
- pSB1A3-1
- dT 180 ng/µL, 3318bp
- N-term 25 ng/µL, 507bp
- Lumazine 120 ng/µL, 861bp
- c-term 25 ng/µL, 55bp
- EYFP 878bp
Ligations of:
pTet-EYFP
Lumazine-dT
c-term-dT
Calculations:
EYFP: 3x(878bp [EYFP]/2211bp [pTet])x25ng
=29.78ng
=4µL EYFP:1 µL pTet
1 µL Lumazine: 1 µL dT
1 µL c-term:1 µL dt
|
| pTet-EYFP
| Lumazine-dT
| c-term-dT
|
| milliQ water
| 3.5 µL
| 6.5 µL
| 6.5 µL
|
| 10x buffer
| 1 µL
| 1 µL
| 1 µL
|
| insert
| 4 µL
| 1 µL
| 1 µL
|
| Vector
| 1 µL
| 1 µL
| 1 µL
|
| Ligase
| 0.5 µL
| 0.5 µL
| 0.5 µL
|
Megan/Mackenzie
Gel extraction pSBIA3-1 and 2 (plasmid for biobrick parts)
Transformation: Tranformed to DH5alpha pBAD = promoter TetR= inverter. Added 2 micro liters of each ligation to DH5alpha. See July 14th Protocol
Restrictions of pBAD (SpeI/PstI), TetR (XbaI/PstI), PLacI (SpeI, PstI), sRBS (SpeI/ PstI), mRBS (SpeI/PstI). Used PCR tubes. Each tube contains: 5 micro liters MillQ water, 3 micro liters 10x Tango Buffer, 1 micro liter enzyme 1, 1 micro liter enzyme 2, 20 micro liters of DNA. Put all tubes in thermal controller at 37 degrees.
Roxanne,
Gel Extraction of pBAD, TetR, pLacI, sRBS, mRBS
Lengths:
pBAD:2287 bp
TetR: 902 bp
pLacI: 2279
sRBS: 2092 bp
mRBS: 2092 bp
Lane:
- Ladder
- TetR
- pLacI
- sRBS
- mRBS
- pBAD
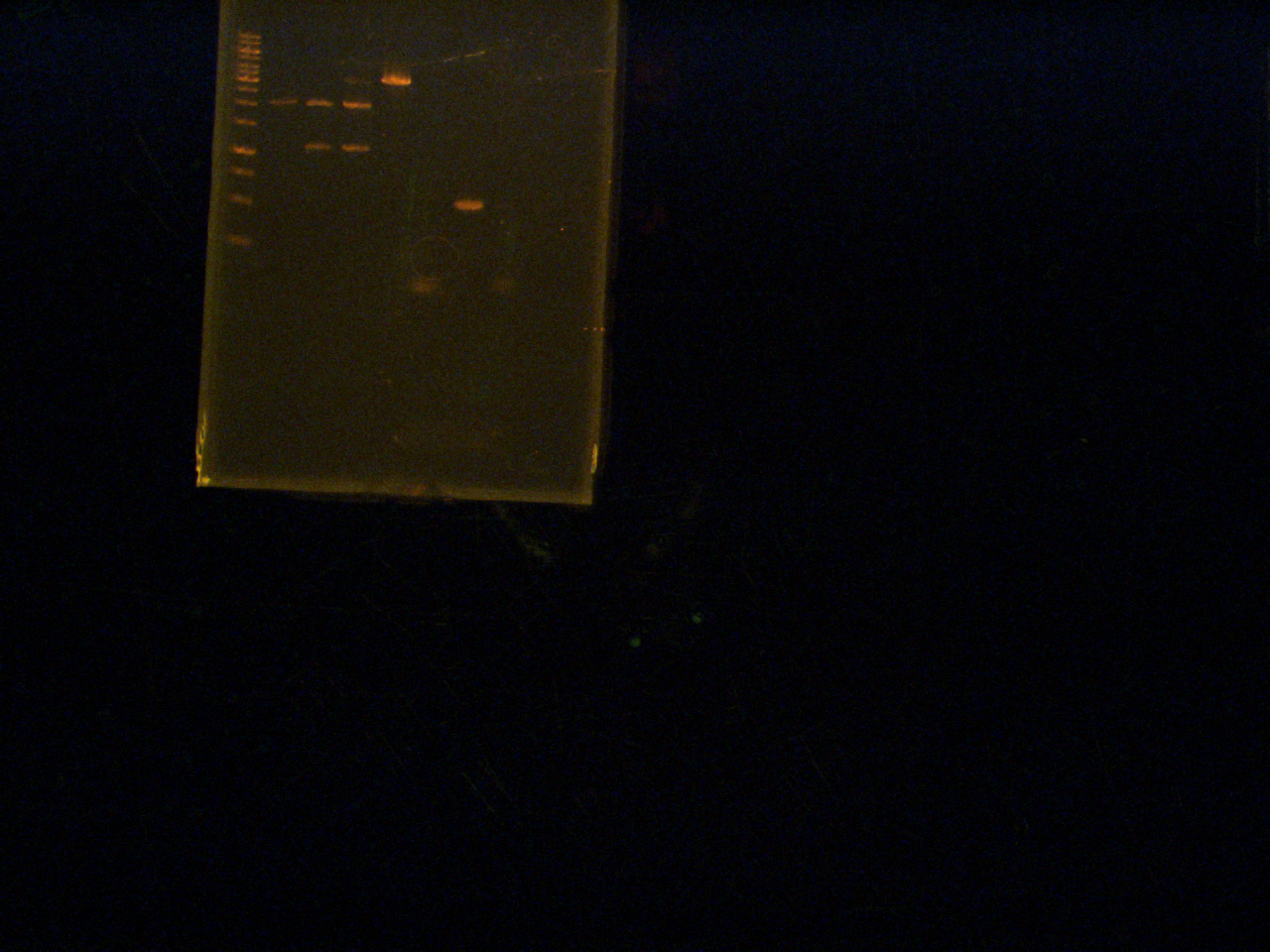
Aug 5th
Kirsten and Fan
Gel extraction of TetR, pLacI, sRBS, mRBS, pBAD according to Qiagen protocol.
Ran analytic gel
1µL Dye + 5µL DNA
6µL Ladder
Lane:
- Ladder
- TetR
- pLacI
- sRBS
- mRBS
- pBAD
- pSB1A3-1
- pSB1A3-2

tetR concentration: 25ng/µL
everything else: 100ngµL
Aug 6
Roxanne
Set up the following ligations
pSB1A3-2+n-term tag and
pSB1A3-1+c-term tag
- 6.5µL milliQ water
- 1 µL 10x T4 buffer
- 1 µL insert
- 1 µL vector
- 0.5 µL T4 ligase
pSB1A3+lumazine
- 5.5µL milliQ water
- 1 µL 10x T4 buffer
- 2 µL insert
- 1 µL vector
- 0.5 µL T4 ligase
pBAD+tetR inverter
- 2.5µL milliQ water
- 1 µL 10x T4 buffer
- 5 µL insert
- 1 µL vector
- 0.5 µL T4 ligase
mRBS+n-term tag
- 6.5µL milliQ water
- 1 µL 10x T4 buffer
- 1 µL insert
- 1 µL vector
- 0.5 µL T4 ligase
Colony PCR of pTet-EYFP, lumazine-dT, c-term-dT
Picked 5 colonies of each construct, followed protocol of July 23.
E1-E5=EYFP colonies 1-5
E+=EYFP (no pTet)
L1-L5=lumazine-dT colonies 1-5
L+= lumazine (no dT)
C1-C5= c-term-dT colonies 1-5
C+= c-terminal (no dT)
-=no plasmid
Lisza
Mixed 10mM primers for: antisense suffix primer and FP N-term fusion primer
Ashley, Mackenzie, Lisza
Analytical gel of colony PCR’s (from earlier)
Volumes:
- 10 µL DNA
- 2 µL 6x loading
- 10 µL ladder
Lane
- 1 kb ladder
- –ve control
- L1
- L2
- L3
- L4
- L5
- L+
- E1
- E2
- E3
- E4
- E5
- E+
- C1
- C2
- C3
- C4
- C5
- C+
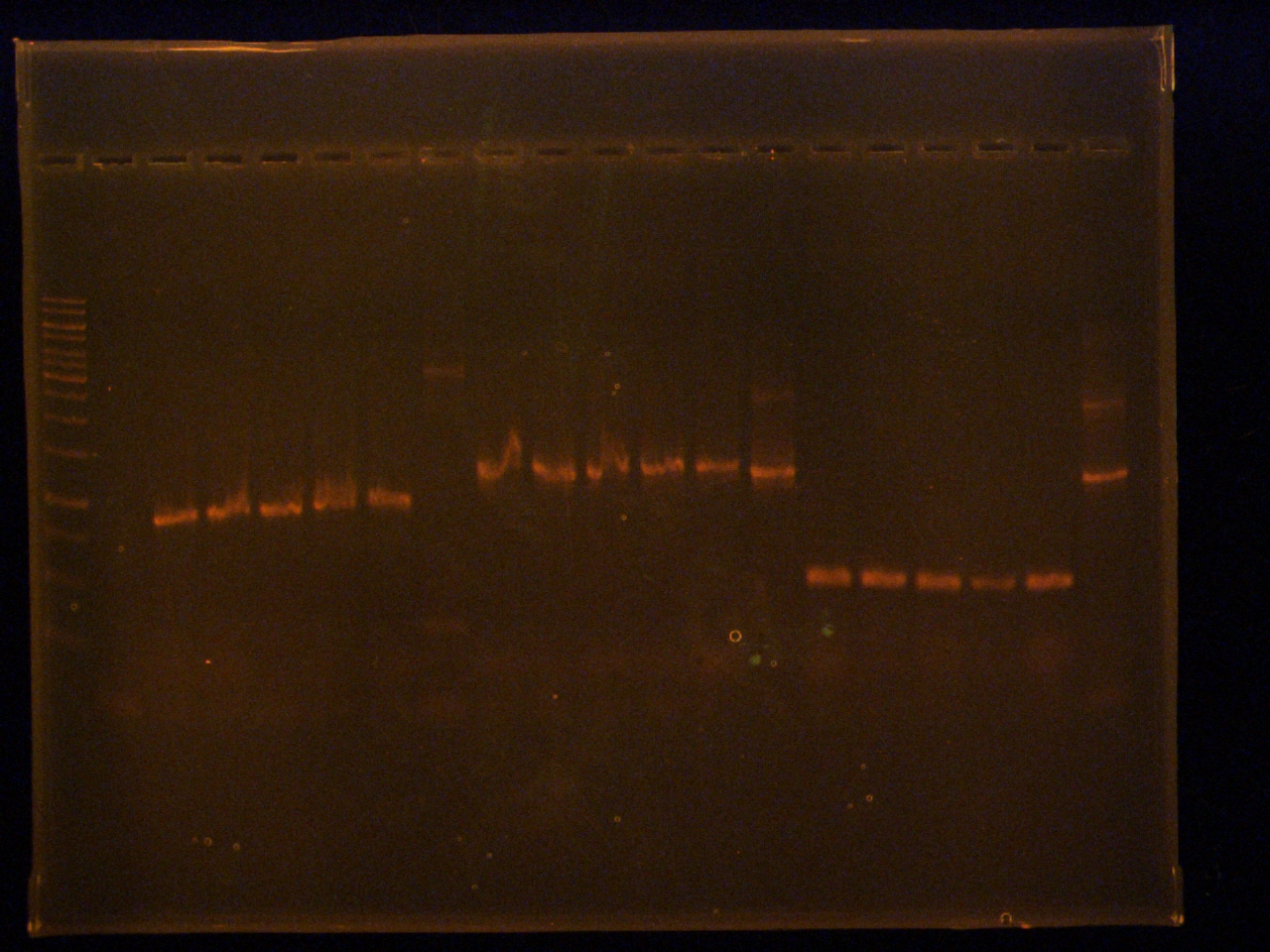
Transformation of mRBS+n-term, pBAD+tetR, pSB1A3+lumazine, pSB1A3+c-term, pSB1A3+n-term
Negative control: water+25 µL DNA
Transformed according to iGEM protocol. Plated on ampicillin plates at 100µL and 400 µL volumes and incubated overnight.
PCR of EYFP and CFP according to protocol of July 30th.
Aug 7
The transformations seemed to have all worked, although there are no colonies on plates marked pSBB1A3+N-term and several on the negative control plate. Speculation: tubes were switched or improper labeling.
Solution: Colony PCR
Lane:
- Ladder (1kb)
- Negative
- TetR
- pBAD+tetR colony 1
- pBAD+tetR colony 2
- pBAD+tetR colony 3
- mRBS
- mRBS+nterm colony 1
- mRBS+nterm colony 2
- mRBS+nterm colony 3
- pSB1A3
- c-term colony 1
- c-term colony 2
- c-term colony 3
- N-term colony 1
- N-term colony 2
- N-term colony 3
- Lumazine colony 1
- Lumazine colony 2
- Lumazine colony 3
Set up restriction digest of the preperative PCR of EYFP (c-term) and CFP(c-term) fusion proteins
DPNI, XbaI,PstI
DPNI, EcoRI, speI for the n-term proteins
Picked colonies of the transformed cells for pBAD-TetR, pSB1A3 (n-term), pSB1A3 (c-term), pSB1A3 (lumazine) and mRBS-Nterm
3x in ampicillin 5mL tubes.
Aug 11
Roxanne
Restricted all the minipreps with EcoRI to check the size of the plasmids prior to sending away for sequencing (10µL rxns)
Restricted:
- pSB-(n-term) w/ SpeI, PstI
- pSB-(c-term) w/ EcoRI, XbaI
in 30 µL rxns to prepare for fusion to CFP of EYFP
Setting up ligations of:
- pSB1A3 + EYFP (c-term)
- pSB1A3 + CFP (c-term)
- pSB1A3 + EYFP (n-term)
- pSB1A3 + CFP (n-term)
mmS6 gene was transformed into DH5α by Megan and Fan, set up a few cultures for minipreps.
Aug 13
Set up restrictions of :
- mmS6 (EcoRI, SpeI)
- n-term CFP (EcoRI, SpeI)
- CheZ (EcoRI, SpeI)
- mRBS-N-term (SpeI, PstI)
- c-term-dt (EcoRI, XbaI)
Gel Extractions of:
Using Qiagen bench top protocol
Set up ligations of:
- pSB1A3-1 + mmS6
- pSB1A3-1 + riboswitch
- pSB1A3-1 + n-term CFP
- pSB1A3-1 + n-term EYFP
- pSB1A3-2 + c-term CFP
- pSB1A3-2 + c-term EYFP
- mRBS + N-term CFP
- mRBS + N-term EYFP
- CheZ+dT
- mmS6 + dT
Ran a concentration gel of:
- c-term CFP
- c-term EYFP
- N-term CFP
- N-term EYFP
- c-term-dT
- mRBS-N-term
Concentrations:
- Riboswitch= 100ng/µL
- pSB1A3-1= 100ng/µL
- pSB1A3-2= 100ng/µL
- dT= 180ng/µL
Ran 1% agarose gel (with 2 wells taped together)
Lane:
- 5µL of 1kb ladder
- 20µL CheZ, 4µL dy
Ran at 110V for 30min
Gel extraction and purification of CheZ and mms6, mmS6 split into two tubes. Following Qiagen kit purification benchtop protocol.
Concentration Gel (1% agarose)
Lane:
- 1 kb ladder (6µL)
- N-term EYFP
- C-N
- Y-N
- 4
- 1
- mmS6
- C-term dT
- mRBS+n-term
Only 9/18 transformations worked
Success:
- mmS6 in pSB1A3
- mmS6 in dT
- riboswitch in pSB1A3
- CheZ in dT
- CFP (no promoter)
- GFP gene
- pSB1A3 + n-term EFYP (1)
- pSB1A3 +n-term EYFP (2)
- fused c-EYFP
Added more ligase and will re-transform the following:
- Fused NEYFP (1)
- Fused NEYFP (2)
- Fused NCFP (1)
- Fused NCFP (2)
- Fused CEYFP
- Fused CCFP
- pSB1A3 +N-CFP (1)
- pSB1A3 +N-CFP (2)
- pSB1A3 +C-CFP (1)
- pSB1A3 +C-EYFP (1)
Transformed all of the above. Picked colonies of successes.
Aug 15th
The transformations didn’t work! Restart
Picked 3 colonies of cheZ-dT
1 of mmS6+dT (for mini and maxiprep)
3 of fused C-EYFP
3 of pSB1A3-mmS6
3 of riboswitch in pSB1A3
3 of GFP gene
3 of CFP (no promoter)
2 of the riboswitch and all of the N-EYFP in pSB1A3 turned red suggesting that the RFP reporter was religated in.
Will miniprep the “good” cells and restrict with EcoRI to check for size
Glycerol stock.
Aug 18
Megan and Ashley
Pelleted down cells (small samples and large samples) picked from transformations to prep for mini-preps.
Add 750 x2 micro liters of your small sample solution to centrifuge tube, Add 50 mL of large sample to large centrifuge tubes. Balanced the Centrifuge and ran for 2 minutes at 13000 rpm. Dump out Supernatant/ leave pellet. Add more sample, centrifuge, and remove more supernatant. Add remainder of sample, centrifuge, remove supernatant. Left samples in -20 degree freezer.
Aug 19
Kirsten and Mackenzie
Maxiprep of fused C-EYFP and mmS6-dT
See protocol for July 13th.
Aug 20
Fan and Mackenzie
Miniprep for:
- cheZ-dT
- mmS6-dT
- PSB1A3-mmS6
- GFP
- CFP no promo
- Fused C-EYFP
- Riboswitch (2,4,7,8,9)
Restriction digest with EcoRI
- 10µL DNA
- 2 µL 10x buffer tango
- 4 µL EcoRI
- Total volume = 20 µL
37°C for 1 hour.
Need to run agarose for all restriction digested samples and DNA samples
Roxanne, Mackenzie
Running a gel of restricted minipreps
Checking mini prep concentration with UV spec
Lane
- 1 kb ladder
- cheZ-dT 1 (78 µg/mL)
- cheZ-dT 2 (80 µg/mL)
- cheZ-dT 3 (106 µg/mL)
- mmS6-dT 1 (82 µg/mL)
- mmS6-dT 2(91 µg/mL)
- mmS6-dT 3 (144 µg/mL))
- pSB-mmS6 1 (125 µg/mL)
- pSB-mmS6 2(88 µg/mL)
- pSB-mmS6 3(135 µg/mL)
- GFP 1(-------)
- GFP 2 (108 µg/mL))
- GFP 3(98 µg/mL)
- CFP no promo 1(----------)
- CFP no promo 2 (134 µg/mL)
- Fused C-EYFP 1 (49 µg/mL)
- Fused C-EYFP 2 (174 µg/mL)
- Fused-CEYFP 3 (75 µg/mL)
- 1 kb ladder)
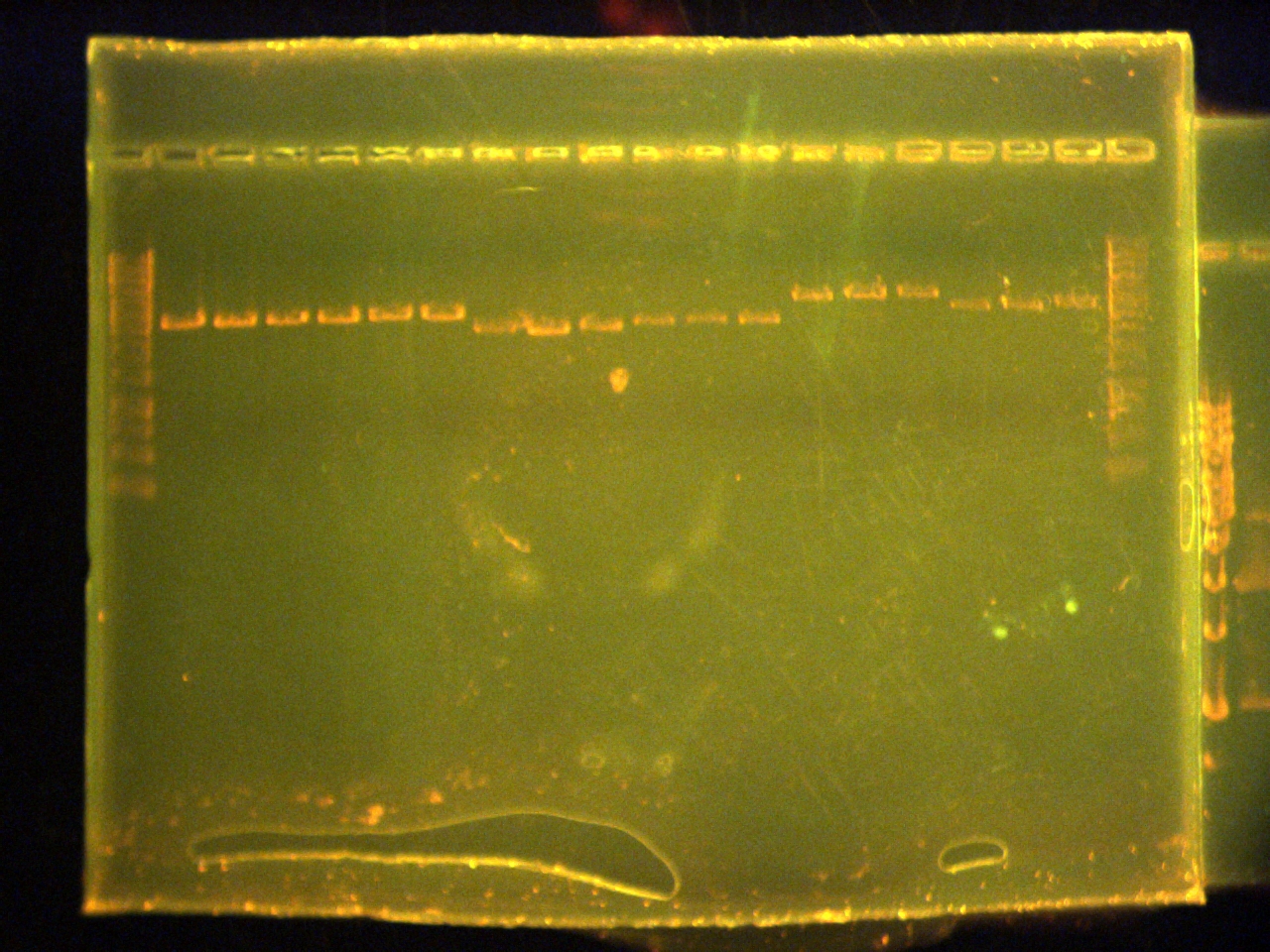
Lane
- 6 µL 1 kb ladder
- 6 µL pSB1A3
- Riboswitch 2 (152 µg/mL)
- Riboswitch 4 (81 µg/mL)
- Riboswitch 7 (68 µg/mL)
- Riboswitch 8 (112 µg/mL)
- Riboswitch 9 (88 µg/mL)
- 6 µL 1 kb ladder
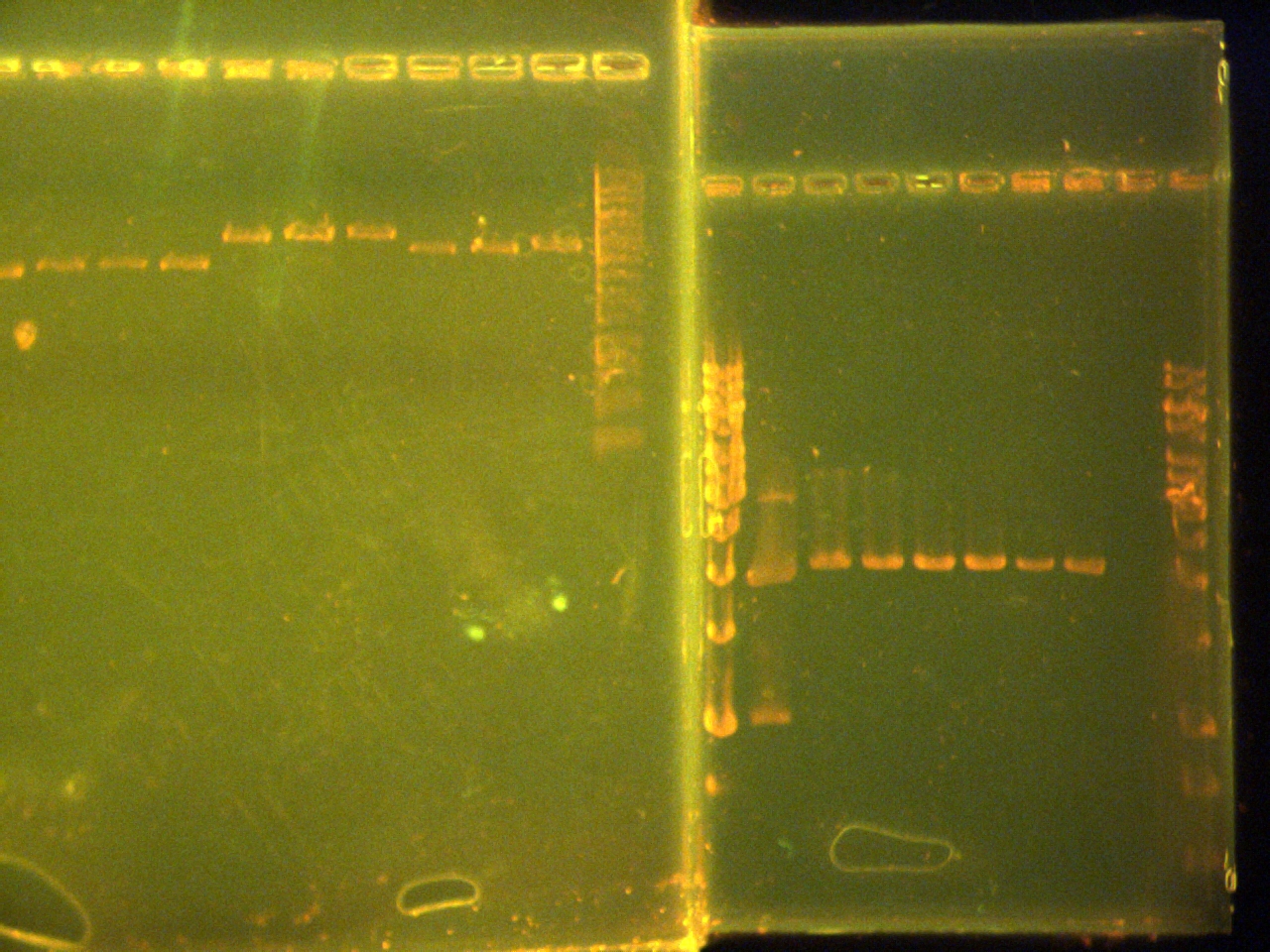
Aug 25
Roxanne
Set up restrictions
1,4,7 are c-term
2,5,8 are n-term
1,2=EYFP
4,5=CFP
7,8=ECFP
- Fusion 1,4,7 restrict with DPN1, EcoRI, SpeI
- Fusion 1,4,7 restrict with DPN1, XbaI, PstI
- Fusion 2,5,8 restrict with DPN1, EcoRI, SpeI
- Fusion 2,5,8, restrict with DPN1, XbaI, PstI
- Riboswitch restrict with SpeI, PstI
- CheZ-dT restrict with XbaI, PstI
- GFP restrict with EcoRI, SpeI
- 4 µL milliQ water
- 2 µL 10x Tango buffer
- 12 µL DNA
- 1 µL E1
- 1 µL E1
Set up maxipreps for:
Ligations:
- Fusion 1 + (c-term)-dT
- Fusion 4 + (c-term)-dT
- Fusion 7 + (c-term)-dT
- Fusion 1 + pSB1A3-2
- Fusion 4 + pSB1A3-2
- Fusion 7 + pSB1A3-2
- Fusion 2 + mRBS (n-term)
- Fusion 5 + mRBS (n-term)
- Fusion 8 + mRBS (n-term)
- Fusion 2 +pSB1A3-1
- Fusion 5 +pSB1A3-1
- Fusion 8 +pSB1A3-1
- Riboswitch +CheZ-dT
- GFP + dT
- sRBS + lumazine-dT
- sRBS + mmS6-dT
Aug 26
Lisza
Deactivated to reactions @ 65° for 15 min
Roxanne
Set up liquid cultures of:
- mmS6 in pSB1A3
- mmS6-dT
- lumazine in pSB1A3
- lumazin-dT
- c-term in pSB1A3
- c-term-dT
- n-term in pSB1A3
- mRBS-(n-term)
- pTet-EYFP
- pBAD-TetR
- riboswitch in pSB1A3
Aug 27
Fan
Miniprep of the O/N culture
[DNA] unknown
DNA was eluted from the spin column in 50mL milliQ water
- 20µL of each DNA sample was sent to iGEM (MIT)
Kirsten
Gel extraction of GFP, mmS6-dT,CheZ-dT
Purified riboswitch, sRBS, c-term-dT, N-term-mRBS. Eluted with 50µL of EB
Into -20°C freezer.
10µL ligations of GFP+dT, sRBS+mmS6-dT and riboswitch+cheZ. Left at room temp for 2 hours until transformation.
Alix
Transformations of ligations following iGEM protocol
Aug 31
Roxanne
PCR of CFP, ECFP, EYFP w/ n-term prefix or c-term suffix
20µL Restrictions of:
- pLacI with SpeI,PstI
- pStrong with SpeI,PstI
- Lumazine-dT with XbaI ,PstI
September
September 3
Roxanne, Ashley, Kirsten
Made 1.5L agar LB media according to lab protocol. Roxanne is autoclaving tomorrow.
Pick 3 lumazine-dT colonies from glycerol stocks to grow overnight
Restriction Digest
Reaction 1
|
| Ribo-cheZ-dT
| GFP-dT
| sRBS-mmS6
| Mr. Gene mmS6
|
| milliQ water
| 5 µL
| 5 µL
| 5 µL
| 5 µL
|
| Buffer tango
| 3 µL
| 3 µL
| 3 µL
| 3 µL
|
| DNA
| 20 µL
| 20 µL
| 20 µL
| 20 µL
|
| XbaI
| 1 µL
| 1 µL
| 1 µL
| 1 µL
|
| PstI
| 1 µL
| 1 µL
| 1 µL
| 1 µL
|
| total
| 30 µL
| 30 µL
| 30 µL
| 30 µL
|
Reaction 2
|
| Ribo-cheZ-dT
| GFP-dT
| sRBS-mmS6
| Mr. Gene mmS6
|
| milliQ water
| 3 µL
| 3 µL
| 3 µL
| 3 µL
|
| Buffer tango
| 1 µL
| 1 µL
| 1 µL
| 1 µL
|
| DNA
| 5 µL
| 5 µL
| 5 µL
| 5 µL
|
| EcoRI
| 1 µL
| 1 µL
| 1 µL
| 1 µL
|
| total
| 10 µL
| 10 µL
| 10 µL
| 10 µL
|
Into HWB at 37°C for 2 hours
Gel Electrophoresis (concentration):
Lane
- 1kb ladder
- ribo-cheZ-dT (EcoRI)
- GFP-dT (EcoRI)
- sRBS-mmS6-dT (EcoRI)
- NEYFP (XbaI,PstI)
- NEYFP (ecoRI, speI)
- NCFP (ecoRI, speI)
- NECFP (xbaI,pstI)
- CCFP(ecoRI,speI)
- CECFP(ecoRI,speI)
- CCFP (notI,pstI)
- NEYFP (ecoRI,speI)
- CECFP (xbaI)
- CEYFP(xbaI,pstI)
- NCFP (xbaI,pstI)
- CEYFP(xbaI)
- pLacI
- pStrong
- 1kb ladder
5µL sample, 1 µL loading dye (6x)
6 µL ladder
Ran at 100V for 1 hour
Concentrations good!
Alix
Gel extraction of:
sRBS-mmS6-dT
riboswitch-cheZ-dT
GFP-dT
mmS6
Following Qiagen benchtop protocol
September 4
Ashley
Ligation of:
- C-term-dT +C-CFP #2
- C-term-dT +C-ECFP #2
- C-term-dT + C-EYFP #2
- pSB1A3-2+C-CFP#1
- pSB1A3-2+C-ECFP#1
- pSB1A3-2+C-EYFP#1
- mRBS-N-term +N-CFP#1
- mRBS-N-term +N-EYFP#1
- mRBS-N-term +N-ECFP#1
- pSB1A3-1+N-CFP#2
- pSB1A3-1+N-ECFP#2
- pSB1A3-1+N-EYFP#2
Reaction
|
| Control
| All reactions
|
| milliQ water
| 7.5 µL
| 4.5 µL
|
| 10x buffer T4
| 1 µL
| 1 µL
|
| Vector
| 1 µL
| 1 µL
|
| Insert
| 0 µL
| 3 µL
|
| T4 DNA ligase
| 0.5 µL
| 0.5 µL
|
| total
| 10 µL
| 10 µL
|
Restriction Digest
|
| Control (no enzyme)
| Lumazine-dT (1)
|
| milliQ water
| 2.4 µL
| 0.4 µL
|
| 10x buffer Tango
| 1.6 µL
| 1.6 µL
|
| DNA
| 16 µL
| 16 µL
|
| XbaI
| 0 µL
| 1 µL
|
| PstI
| 0 µL
| 1 µL
|
| total
| 20 µL
| 20 µL
|
September 8
Ashley
Made 1.5L LB agar according to protocol
Made 300mL LB broth according to protocol
Gel Electrophoresis (for extraction)
5 µL ladder
5 µL sample, 1 µL dye
Lane
- 1 kb ladder
- empty^
- empty
- lumazine-dT (XbaI, PstI) 600bp
^there was a control (no enzyme) but not enough DNA to load
Ran at 1000V for one hour
Gel extraction according to Qiagen benchtop protocol
Sept 10
Ashley, Kirsten
Ran 1% agarose
Lane
- 1 kb ladder
- C-term-dT + C-EYFP #2
- pSB1A3-1+N-CFP#2
- pSB1A3-1+N-ECFP#2
- lumazine+sRBS
- pSB1A3-2+C-EYFP#1
- pSB1A3-1+N-EYFP#2
- mRBS-N-term +N-CFP#1
- mmS6+pSB1A3-1
- GFP-dT
- mmS6-dT
- pSB1A3-2+C-ECFP#1
- mRBS-N-term +N-ECFP#1
- mRBS-N-term +N-EYFP#1
- C-term-dT +C-CFP #2
- C-term-dT +C-ECFP #2
- pSB1A3-2+C-CFP#1
3 µL sample + 0.5 µL dye
6 µL 1 kb ladder
Mackenzie
Transformation into DH5α
- sRBS-lumazine-dT (km)
- mmS6-dT (km)
- pSB1A3+mmS6
- mRBS-N-term +N-ECFP#1
- mRBS-N-term +N-EYFP#1
- C-term-dT +C-ECFP #2
- C-term-dT + C-EYFP #2
- pSB1A3-2+C-ECFP#1
- pSB1A3-2+C-EYFP#1
The following were not transformed due to lack of competent cells:
- pSB1A3-1+N-ECFP#2
- pSB1A3-1+N-EYFP#2
- GFP-dT
September 14
Ashley
PCR of EYFP, CFP and ECFP
See Aug 6 for volumes
CFP(1)=prefix primer and antisense C-term suffix
CFP(2)=N-term prefix and antisense suffix
EYFP(1)=prefix primer and antisense C-term suffix
EYFP(2)=N-term prefix and antisense suffix
ECFP(1)=prefix primer and antisense C-term suffix
ECFP(2)=N-term prefix and antisense suffix
Cycle: tail67
Sept 16
Ashley
Miniprep of mmS6-dT 1,2 and 3. Used Qiagen benchtop protocol and kit. Eluted with 50 µL buffer EB. Put into -20°C freezer orange tray.
PCR Purification of restriction digest of PCR products.
September 24
Ashley, Kirsten
1% gel electrophoresis
6uL ladder (1kb)
5uL sample +1 uL 6x loading dye
100V, 40min
Lane
- 1kb Ladder
- C-CFP-1
- C-ECFP-1
- N-EYFP-2
- N-CFP-2
- N-ECFP-2
- C-EYFP-1
- CFP-1 Purified PCR
- ECFP-1 Purified PCR
- EYFP-2 Purified PCR
- CFP-2 Purified PCR
- ECFP-1 Purified PCR
- EYFP-1 Purified PCR
- C-CFP(1)
- ECFP(1)
- C-EYFP(1)
- N-CFP(2)
- N-EYCFP(2)
- N-EYFP(2)
- 1KB LADDER
Gel was run to check for DNAses, found that DPN-1 had DNAses.
Sept 25
1% agarose gel 100V, 40min
Lane:
- 1kb ladder
- mms6(EcoRI,SpeI)
- pLacI (SpeI, PstI)
- Lumazine-dT
Only pLacI success, gell extracted and eluted with 50uL buffer EB.
Miniprep of:
- 3x mmS6-dT
- 3x pSB1A3-mmS6
- 2x cheZ-dT
- 1x riboswitch-cheZ-dT
- 2x GFP-dT
- 3x C-EYFP-dT
Restricted all of the above with EcoRI
- 8uL DNA
- 1uL 10x buffer tango
- 1uL EcoRI
for 1 hour.
Restriction of fusion proteins with either EcoRI,SpeI or XbaI,PstI
- Heat inactivated a 60C for 10min
Ran on a gel (1% agarose)100V,40min
Lane:
- 1kb ladder
- pSB-mmS6 #1
- pSB-mmS6 #2
- pSB-mmS6 #3
- mms6-dT #1
- mms6-dT #2
- mms6-dT #3
- EYFP C-term-dT #1
- EYFP C-term-dT #2
- EYFP C-term-dT #3
- GFP-dT#1
- GFP-dT#2
- Riboswitch-cheZ-dT
- cheZ-dT#1
- cheZ-dT#2
- C-CFP (EcoRI,SpeI)
- C-CFP (XbaI,PstI)
- C-ECFP (EcoRI,SpeI)
- C-ECFP (XbaI,PstI)
- 1kb laddder
- Restriction of Fusion proteins with Dpn1
- Heat inactivated a 60C for 10min
Lane:
- 1kB ladder
- N-ECFP
- N-ECFP
- N-EYFP
- E-EYFP
- N-CFP
- N-CFP
- C-EYFP
- C-EYFP
- 1kB ladder
Restriction of Lumazine-dT
- 30uL DNA
- 4uL 10x buffer
- 3uL EcoRI
- 3uL SpeI
Setup Liquid culture of the Mr. Gene mmS6 x3
September 28
Ashley
1% agarose gel
- 5uL DNA+1uL 6x loading dye
- 6uL ladder (1kb)
Lane:
- 1kb ladder
- N-ECFP (XbaI,PstI)
- N-ECFP (EcoRI, SpeI)
- N-EYFP(XbaI,PstI)
- N-EYFP(EcoRI, SpeI)
- N-CFP(XbaI,PstI)
- N-CFP(EcoRI, SpeI)
- C-EYFP(XbaI,PstI)
- C-EYFP(EcoRI, SpeI)
- 1kb ladder
Ran at 100V for 40 min
October
October 1
Roxanne
Gel for gel extraction
Restriction
Ribo-Chez-dT
- 2x CheZ-dT
- 2x GFP-dT
- 3x C-EYFP-dT
Ligation
- C-ECFP-dT
- C-CFP-dT
- C-EYFP-dT
- mRBS-N-ECFP
- mRBS-N-EYFP
- pSB-C-EFP
- pSB-C-EYFP
- pSB-C-CFP
- pSB-N-ECFP
- pSB-N-EYFP
Aliquoted DH5α cells
Ashley
| Tube
| Tube + Gel
| Gel
|
| 1.1226g
| 1.4931g
| 0.2671g
|
| 1.012g
| 1.2302g
| 0.1290g
|
Eluted with 50uL buffer EB
Ligation
|
| 1:3
| 2:6
|
| lum-dT
| 3uL
| 6uL
|
| SRBS
| 1uL
| 2uL
|
| Ligase Buffer
| 1uL
| 2uL
|
| Water
| 4.5uL
| 9uL
|
| Ligase
| 0.5uL
| 1uL
|
Oct 2
Pick Colonies:
- sRBS + Lumazine-dT
- mRBS + N-term FPs
- C-term FPs + dT
- pSB + N-term FPs
- pSB + N-term FPs
Gel Extraction:
- Riboswitch-cheZ-dT
- 2x cheZ-dT (in case Ribo-cheZ-dT isn't actually there)
- 3x C-EYFP-dT
- 2x GFP-dT
Liquid Culture:
- Riboswitch-cheZ-dT
- 2x cheZ-dT (in case Ribo-cheZ-dT isn't actually there)
- 3x C-EYFP-dT
- 2x GFP-dT
Miniprep:
Sat Oct 3
Miniprep:
- sRBS + Lumazine-dT
- mRBS + N-term FPs
- C-term FPs + dT
- pSB + N-term FPs
- pSB + N-term FPs
- sRBS + Lumazine-dT
Restriction:
Analytic and Prep:
- mRBS + N-term FPs
- C-term FPs + dT
- pSB + N-term FPs
- pSB + N-term FPs
Analytic only:
Prep only:
Oct 5
Gel extraction:
- sRBS + Lumazine-dT
- mRBS + N-term FPs
- C-term FPs + dT
- pSB + N-term FPs
- pSB + N-term FPs
Ligation:
- pStrong + Riboswitch-cheZ-dT
- Riboswitch-GFP-dT
- pLac + sRBS-Lumazine-dT
- pBad-TetR + C-term FPs
- pBad-TetR + N-term FPs
- pTetR + N-term
Transformation:
- pLac + sRBS-Lumazine-dT
- pBad-TetR + C-term FPs
- pBad-TetR + N-term FPs
- pTetR + N-term ECFP
- pStrong + Riboswitch-cheZ-dT
- Riboswitch-GFP-dT
Sequencing:
- sRBS + Lumazine-dT
- mRBS + N-term FPs
- C-term FPs + dT
- pSB + N-term FPs
- pSB + N-term FPs
- Mr. Gene mms6
Tues Oct 6
Pick Colonies:
- pLac + sRBS-Lumazine-dT
- pBad-TetR + C-term FPs
- pBad-TetR + N-term FPs
- pTetR + N-term ECFP
- pStrong + Riboswitch-cheZ-dT
- Riboswitch-GFP-dT
Wed Oct 7
Miniprep:
- pLac + sRBS-Lumazine-dT
- pBad-TetR + C-term FPs
- pBad-TetR + N-term FPs
- pTetR + C-term ECFP
- pTetR + N-term ECFP
- pStrong + Riboswitch-cheZ-dT
- Riboswitch-GFP-dT
Analytic Restriction:
- pLac + sRBS-Lumazine-dT
- pBad-TetR + C-term FPs
- pBad-TetR + N-term FPs
- pTetR + N-term ECFP
- pStrong + Riboswitch-cheZ-dT
- Riboswitch-GFP-dT
Prep Restriction:
Thurs Oct 8
Gel Extraction:
Ligation:
- pStrong + Riboswitch-GFP-dT
Transformation:
- pStrong + Riboswitch-GFP-dT
Sequencing:
- pLac-sRBS-Lumazine-dT
- pBad-TetR-C-term FPs
- pBad-TetR-N-term FPs
- pTetR-N-term ECFP
- pStrong-Riboswitch-cheZ-dT
- Riboswitch-GFP-dT
Maxiprep:
- pLac-sRBS-Lumazine-dT
- pBad-TetR-C-term FPs
- pBad-TetR-N-term FPs
- pTetR-N-term ECFP
- pStrong-Riboswitch-GFP-dT
Restriction:
Analytic and Prep:
- pBad-TetR-C-term FPs
- pBad-TetR-N-term FPs
- pTetR-N-term ECFP
- C-term CFP (has pTetR)
Analytic Only:
- pStrong-Riboswitch-GFP-dT
Fri Oct 9 Overexpression pLac + sRBS-Lumazine-dT
Gel Extraction pLac-sRBS-Lumazine-dT
pBad-TetR-C-term FPs
pBad-TetR-N-term FPs
pTetR-N-term ECFP
C-term CFP (has pTetR)
Motility Media
Oct 12
Ligation:
- pLac-sRBS-Lumazine-dT + pBad-TetR-C-term EYFP
- pLac-sRBS-Lumazine-dT + pBad-TetR-N-term EYFP
Transformation:
- pLac-sRBS-Lumazine-dT + pBad-TetR-C-term EYFP
- pLac-sRBS-Lumazine-dT + pBad-TetR-N-term EYFP
Plate Testing:
- pStrong-Riboswitch-GFP-dT
Motility Test:
- pStrong + Riboswitch-cheZ-dT
Oct 13
Pick Colonies
- pLac-sRBS-Lumazine-dT + pBad-TetR-C-term EYFP
- pLac-sRBS-Lumazine-dT + pBad-TetR-N-term EYFP
Analysis of Plate and Motility Tests
Wed Oct 14
Miniprep:
- pLac-sRBS-Lumazine-dT-pBad-TetR-C-term EYFP
- pLac-sRBS-Lumazine-dT-pBad-TetR-N-term EYFP
Restriction:
- pLac-sRBS-Lumazine-dT-pBad-TetR-C-term EYFP
- pLac-sRBS-Lumazine-dT-pBad-TetR-N-term EYFP
Thurs Oct 15
Gel Extraction:
- pLac-sRBS-Lumazine-dT-pBad-TetR-C-term EYFP
- pLac-sRBS-Lumazine-dT-pBad-TetR-N-term EYFP
Ligation:
- pLac-sRBS-Lumazine-dT-pBad-TetR-C-term EYFP + C-term CFP
- pLac-sRBS-Lumazine-dT-pBad-TetR-N-term EYFP + pTetR-N-term CFP
Transformation:
- pLac-sRBS-Lumazine-dT-pBad-TetR-C-term EYFP + C-term CFP
- pLac-sRBS-Lumazine-dT-pBad-TetR-N-term EYFP + pTetR-N-term CFP
Expression Testing:
- pLac-sRBS-Lumazine-dT-pBad-TetR-C-term EYFP
- pLac-sRBS-Lumazine-dT-pBad-TetR-N-term EYFP
Fri Oct 16
Pick Colonies:
- pLac-sRBS-Lumazine-dT-pBad-TetR-C-term EYFP + C-term CFP
- pLac-sRBS-Lumazine-dT-pBad-TetR-N-term EYFP + pTetR-N-term CFP
Sat Oct 17
Maxiprep:
- pLac-sRBS-Lumazine-dT-pBad-TetR-C-term EYFP + C-term CFP
- pLac-sRBS-Lumazine-dT-pBad-TetR-N-term EYFP + pTetR-N-term CFP
 "
"