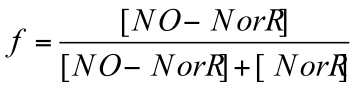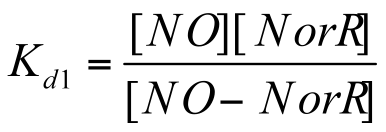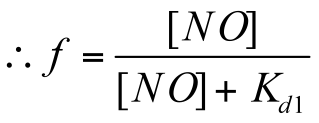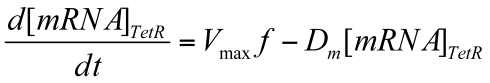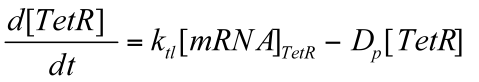Team:NTU-Singapore/Project/Prototype/Image
From 2009.igem.org
Imaging Device
The Imaging device is also one of our featured parts.
As we have elaborated earlier in the Prototype Design section, the device consists of mainly pTet regulated expression of IFP & HO-1, which in turn generate a unique fluorescence signal in the infrared spectrum!
We are extremely pleased to report that we have successfully converted this novel reporter protein into a standard Biobrick! Let us now see how exactly the device works.
Abstract
As part of the imaging capability we want for our system, we have eschewed traditional reporter proteins like GFP or YFP and have identified a novel reporter protein, infrared fluorescent protein (IFP). A recent work by Xiaokun et. al. (Tsien Lab, UCSD) has shown successful mammalian expression of infrared fluorescent protein engineered from a bacterial phytochrome. Xiaokun et. al. have also expressed the infrared fluorescent protein in E. coli strain TOP10.
IFP is preferred for our system over other FPs such as GFP or YFP because it has a unique excitation and emission maxima. The wavelength range of IFP fits so well with the wavelength range of typical in vivo optical imaging in deep tissues of animals i.e. between 650 and 900 nm for excitation and emission maxima respectively. This is because this range of wavelength minimizes the absorbance by haemoglobin, water and lipids, as well as light-scattering.
Therefore, IFP is particularly suitable for our ideal system as we desire to locate the atherosclerotic plaque site without invasive procedures.
Background
Our pLaqUe Out! system must be capable of highlighting the atherosclerotic plaque site upon detection by the preceding sensing mechanism of nitric oxide sensing promoter (pNO). The first thing that came to our mind was the variety of known fluorescent proteins (FPs) such as GFP, RFP, YFP, CFP. However, since our ultimate environment for our pLaqUe Out! system is in vivo, we envisioned an ideal system that can express a novel reporter protein with properties suitable for in vivo optical imaging. IFP was chosen as a potential candidate.
The gene encoding IFP used in our expression system is essentially the gene for IFP1.4. Xiaokun et. al. have shown that IFP1.4 was obtained from the gene encoding IFP1.0 by saturation, random mutagenesis, and DNA shuffling; and it was shown to be about four times brighter than IFP1.0, with a relative brightness of 383%.
Fig 1. - Normalized excitation (blue) and emission (red) spectra for Infrared Fluorescent Protein
Overall, IFP has an excitation and emission maxima of 684 nm & 708 nm respectively; compare this with the excitation and emission maxima for other fluorescent proteins, which have not exceeded 598 and 655 nm respectively.
Mechanism
Infrared fluorescent protein is a monomeric protein engineered from a bacterial phytochrome of the species Deinococcus radiodurans.
This bacteriophytochrome incorporates biliverdin IXα (BV) as the chromophore. Biliverdin IXα is in fact a natural product of heme catabolism by the enzyme heme oxygenase-1 (HO-1). BV is essential because in a mouse experiment conducted by Xiaokun et. al., BV has been shown to increase the infrared fluorescence to a maximal fivefold 1 hour after BV injection in the mouse with exogenously added BV.
In a phone call made to Dr. Xiaokun Shu, we have also been told that BV helps to stabilize the infrared fluorescent protein. Hence, it is essential that we incorporate HO-1 into our system to generate biliverdin IXα.
Sequence
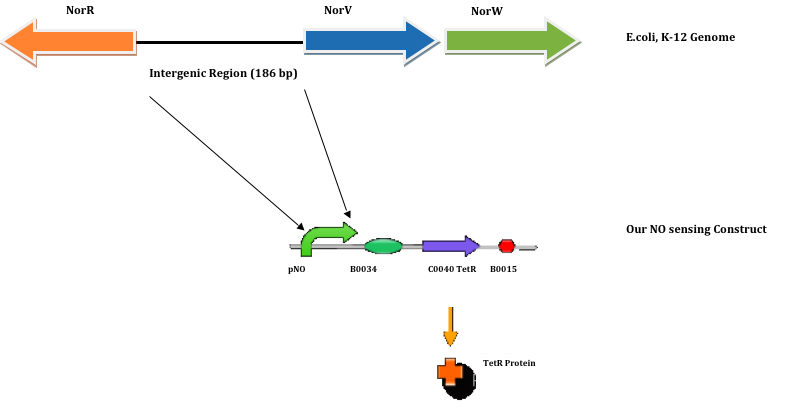
Using NCBI’s reference sequence NC_000913.2, the 186 bp NorR-NorVW intergenic region sequence is obtained using E.coli (strain: K-12; sub-strain: MG1655) as the search organism. The sequence is found in genome position 2830311 – 2830496. The NorR-NorVW intergenic sequence is as follows (5’ - 3’):
TCTTTGCCTCACTGTCAATTTGACTATAGATATTGTCATATCGACCATTTGATTGATAGTCATTTTGACT
ACTCATTAATGGGCATAATTTTATTTATAGAGTAAAAACAATCAGATAAAAAACTGGCACGCAATCTGCA
ATTAGCAAGACATCTTTTTAGAACACGCTGAATAAATTGAGGTTGC
The highlighted sequence is the sigma-54 promoter. The reason why we had included the entire intergenic region as our pNO promoter sequence is that there are 3 enhancer binding sites upstream of the sigma-54 promoter. These enhancer binding sites allow the transcription factor, NorR to bind to the DNA, thus leading to a localized increase in NorR concentration.
As such, the increase in localized NorR concentration facilitates the NorR to make contact with the sigma 54 – RNA polymerase.
Construct Design
In our system, Plaque Out!, we wish to regulate the production of TetR protein with respect to the concentration of NO.
The TetR protein would in turn regulate the production of Cholesterol esterase (CHE), Haem Oxyenase (HO-1), and Infra-red protein (IFP). Thus, our NO sensing construct is as follows:
Characteristic Equations
Ligand binding :
Using Hill equation to express the fraction of NO-bound NorR, we assume independent and non-cooperative binding of NO (i.e. Hill coefficient is taken to be 1).
Thus, fraction of NO-bound NorR is described as follows:
Since the dissociation constant Kd1 can be illustrated as:
Fraction of NO-bound NorR can be expressed as a function of available NO concentration, [NO]free
Transcription of TetR
Deterministic first order differential equation (ODE) is used to depict the NO-regulated transcription of TetR.
Where Vmax is max transcription rate & Dm is the degradation rate of mRNA.
Translation of TetR
Deterministic first order ODE is used again to represent the translation of TetR protein.
Where Ktl is the translation rate of mRNA & Dp is the degradation rate of TetR protein.
Modelling & Simulation
We make the following assumptions:
- Biological systems of transcription and translation are assumed to be linear and time-invariant
- Concentration of the transcription factor, NorR in E.coli is assumed to be in excess and constant. Hence, [NorR] is not taken into account in our model
- There is no time lag for NO diffusion from ext. environment into the cell
- Constant Degradation rates for mRNA as well as protein
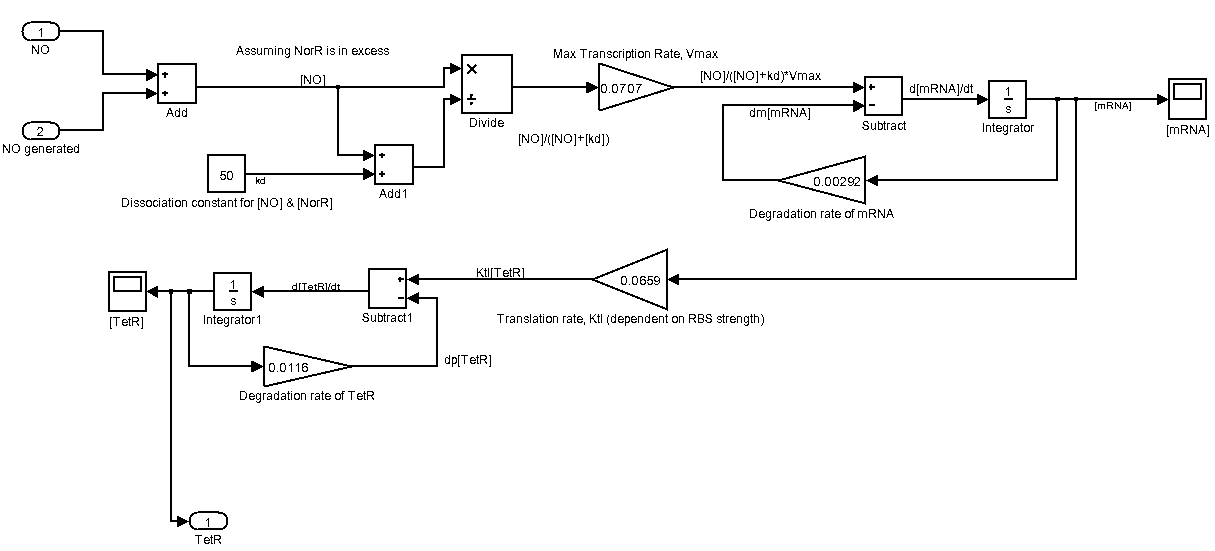
Instead of modelling with TetR, we are using GFP as our output. This is part of our effort in trying to predict the wetlab characterization of pNO.
The following is our Sensing device represented as a Simulink system. Please click on it for a larger view.
Device Construction
Device Characterization
Literature / References
Please proceed here to view our full list of references.
 "
"