Team:EPF-Lausanne/LOVTAP Results
From 2009.igem.org
Contents |
Results
Characterization of Read-Out n°1
Our first read-out system consists of the Trp promoter followed by the RFP gene. In E. coli, the Trp promoter is situated in front of the Trp operon, which contains the genes necessary for the tryptophane biosynthetic pathway. In brief, genes placed after the Trp promoter should be repressed in the presence of tryptophane.
To characterize this biobrick, we cultured the cells in a medium without tryptophane: M9 medium to which we added all other amino acids and thiamine. You can find the protocol for the M9 medium here. The purpose of the experiment was to see the difference of RFP expression depending on whether we added tryptophane to the medium or not.
Protocol
- Cells containing the read-out system were cultured overnight in M9 medium.
- On the next day, re-inoculated 0.75 mL of cell culture into 25 mL of fresh medium in an Erlenmeyer flask. Incubated for 2 hours at 37°C.
- Normalized the OD of all flasks to 0.06 by adding the appropriate amount of fresh medium.
- Prepared the plate for the measurements with the qPCR machine, 50 ul per well. For the tryptophane, we used a 4.25 g/L solution. For ATC, it was a 500 ng/mL solution in 50% EtOH, and for IPTG, we always added it so as to have a final concentration of 1mM. The conditions we tested were 1/2 Trp, 1 Trp and 3/2 Trp, 1/2 corresponding to 1 ul.
- The plate was then loaded into the qPCR machine, which took measurements of red fluorescence every 3 minutes, giving the graphs you can see below.
Please note that there was a time lag between when we started loading the wells on the plate and when the measurements began, so the induction might already have started.
Expected Results
In this experiment, we would expect to see a slight increase in fluorescence for the cells without tryptophane (simply due to cell growth), compared to a decrease in fluorescence for the samples in which we added tryptophane, since tryptophane would enhance the activity of the tryptophane repressor, which in turn would repress the RFP gene via the Trp promoter. Also to be noted, the RFP decay shouldn't be too abrupt, considering that the RFP protein used doesn't contain a degradation tag.
Graphs & Figures
Analysis & Conclusions
We can see that there is a decrease in RFP expression when Trp is added to the medium versus a stable expression for the -Trp condition, which corresponds to what we expected. From this we can therefore conclude that our first read-out system is functional, confirming that the sequence for the Trp promoter seems to be correct.
Characterization of Read-Out n°2
The read-out system n°2 is a double-repressor system: there is a Trp promoter in front of the TetR gene, followed by the Tet promoter in front of the RFP gene. This means that it should react both to tryptophane and tetracycline, an allosteric blocker of TetR (for our experiments we used anhydrous tetracycline, ATC).
The experiment this time was done both in LB and M9 media.
Protocol
- Cells were cultured overnight in LB medium at 37°C.
- Re-inoculated 1 mL of cell culture into 100 mL of fresh medium in Erlenmeyer flasks. Normalized the OD to 0.1 by adding fresh medium. Left to incubate at 37°C for about 2h30.
- Normalized the OD to 0.2 by adding fresh medium.
- In 2 mL Eppendorf tubes, gently mixed 1.8 mL of cell culture with 0.2 mL of Trp solution, ATC solution or cell culture (negative control). The solutions we used for Trp and ATC had respective concentrations of 8 mg/mL and 500 ng/mL (ATC solution in 50% EtOH).
- Loaded 30 ul into each well of the plate for the qPCR machine, with quadruplicates for each condition. The qPCR machine took a measurement of the fluorescence every 3 minutes: this was the data used to obtain the graph you can see further below.
Expected Results
For the negative control (cell culture only), we should see a basal level of RFP expression due to the leakage of the system; this level should remain more or less stable throughout the experiment. On the other hand, both the adding of Trp and ATC should result in an increase in fluorescence. For Trp, its presence in the medium should activate the expression of the Trp repressor, which binds to the Trp promoter, in turn repressing the TetR gene, and so preventing TetR from repressing the RFP gene. Similarly, ATC allosterically blocks TetR, and therefore also prevents it from repressing the RFP gene. In short, Trp and ATC should have a positive effect on RFP expression, thus leading to an increase in fluorescence.
Graphs & Figures
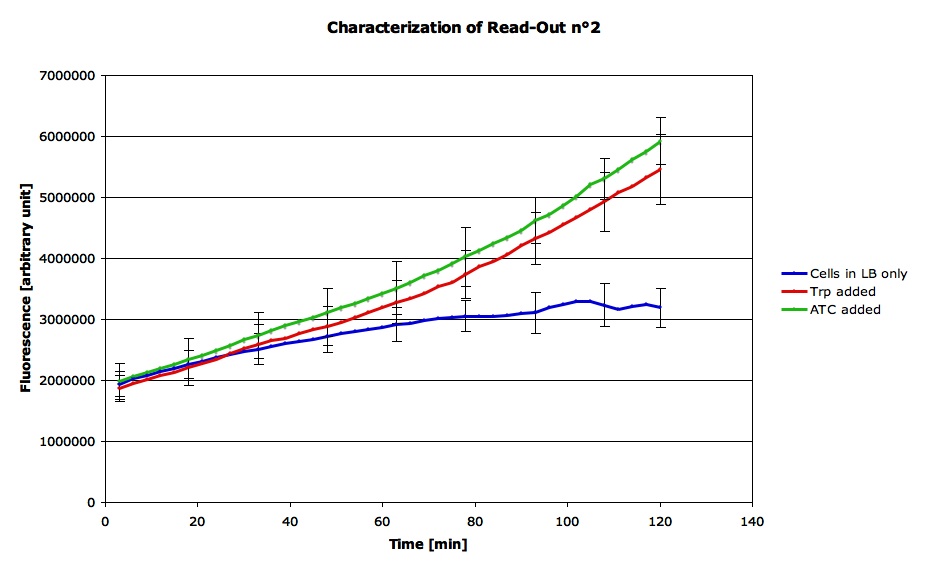
This graph shows the average curve for each condition (tryptophane added, ATC added, or nothing):
Analysis & Conclusions
In the graph, you clearly see that after a certain delay, an increase in fluorescence can be noted for the samples where Trp or ATC was added, while the fluorescence of the cells cultured in LB alone remains at a significantly lower level (note the error bars). This confirms that the read-out system reacts as expected, with an increase in fluorescence in response to both ATC and Trp. We can therefore assume that both the Trp promoter and the Tet promoter are functional and that we can use this part in order to characterize the functioning of the LovTap protein.
Characterization of the entire system
After having successfully characterize both of our read-out systems, we did further experiments in order to evaluate the functioning of our entire genetic circuit, which includes the LovTap biobrick (go to the Strategy page for more information). For convenience, we chose to work with the read-out system n°2: indeed, since the RFP doesn't contain a degradation tag, we assumed that the read-out n°1 would react more slowly, as it involved waiting for the RFP to be degraded before we could really see a response. In comparison, the read-out n°2 could give us a good indication of the kinetics of the response.
The genetic circuit here being more complex, we conducted several different experiments which are described below.
Experiment n°1: Single point measurement
Protocol
Now that we have characterized the read out systems, we were able to see if the LovTAP protein would show the same pattern of response when illuminated as when we added Trp to the medium.
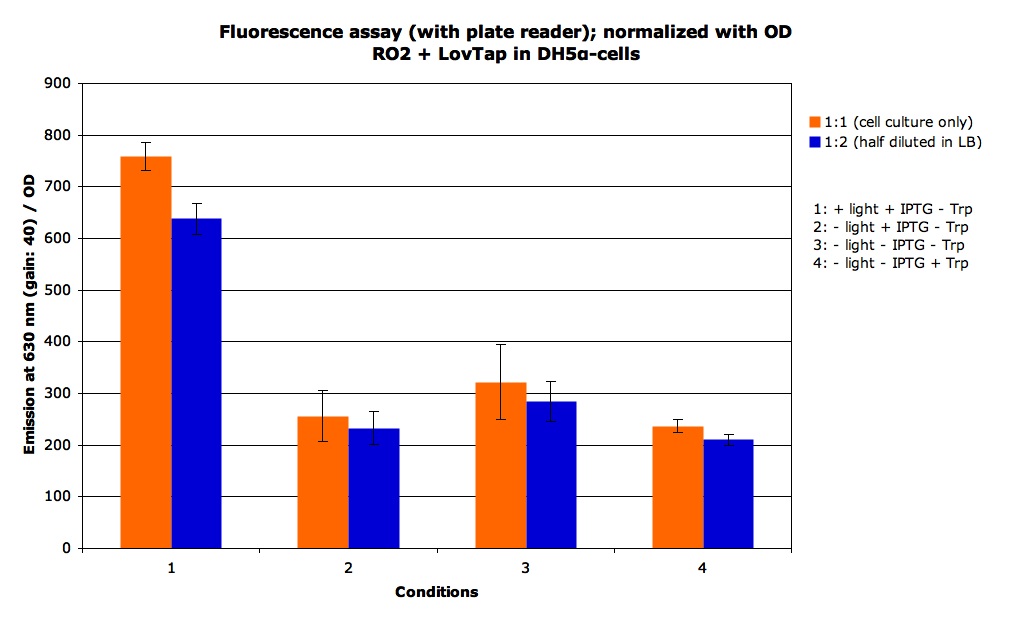
We some static measurement of fluorescence. For the + light condition we exposed the cells to the light for 2 hours, we got the following results
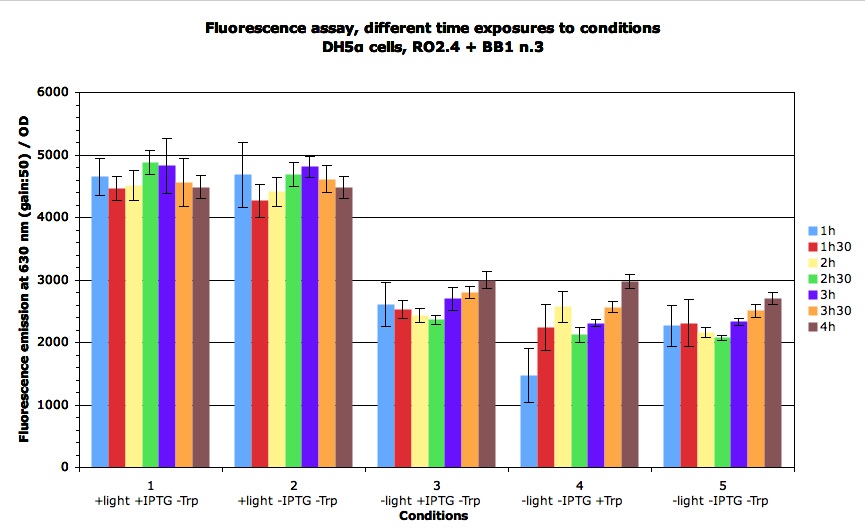
Then we wanted to see the time course of the induction : when is the RFP expressed, how etc.
It appears that this actually happens during the first hour.
Mutations
We used two different mutations in our project.
First we used some E. coli strains that had their Trp promoter mutated.
Then we used some strains we modified to test the results we had in the modeling session.
 "
"