Team:EPF-Lausanne/Results
From 2009.igem.org
Contents |
Fusion of the LOV domain and the trpR DNA-binding domain
The first step in our computational study of the LOV domain was to fuse the 2 domains of interest in VMD. We were then able to visualize the different proteins tried by Sosnick. The working protein, that we call LovTAP is the result of the fusion at PHE22 of trpR and can be seen on the next video. The general LOV domain is in yellow. Please note the chromophore called Flavin (FMN) in red in the center of LOV2. The trpR dna binding domain is in orange and DNA in gray.
Click here to see an example of code used in VMD for the fusion
We also modelized the other fusion tried by Sosnick.
- @MET11
- @ALA12
- @GLU13
- ...
- @PHE22
- ...
- @LEU25
I. Equilibration of dark state
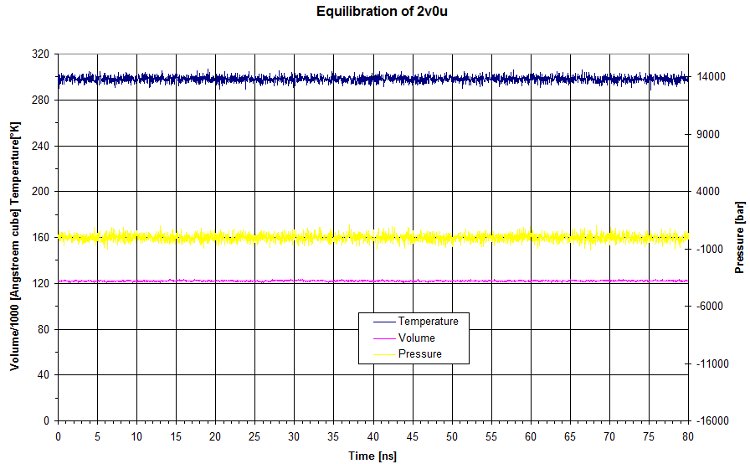
We run an equilibration of 80ns on the dark state (2v0u).
Here is a movie over the trajectory file.
Validation of the simulation
Here we look at the output to check input parameters.
The raw data for the equilibration match what we set for the NPT. Pressure and temperature are kept constant using namd dynamic. The volume is quite constant as well.
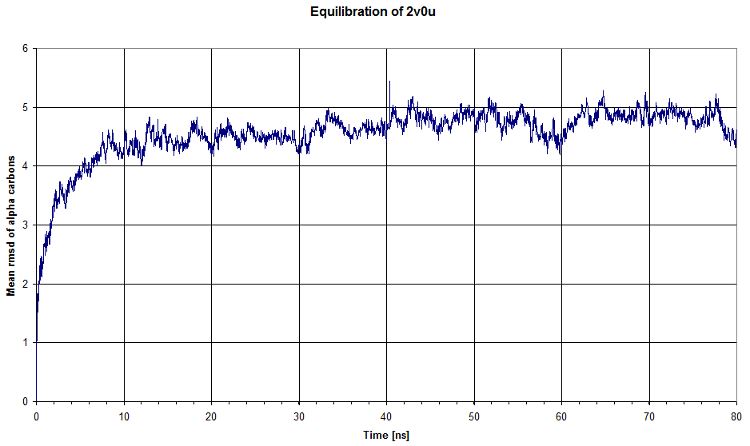
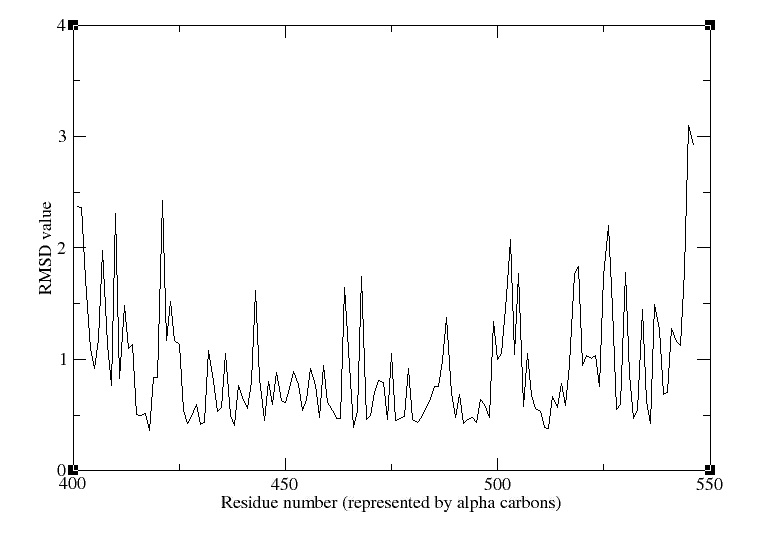
Then we computed the evolution of the rmsd compared to the first timestep of equilibration. We see that there is a plateau after ~40ns, which means that our system's energy is reaching a minimum. That's clearly what we expected.
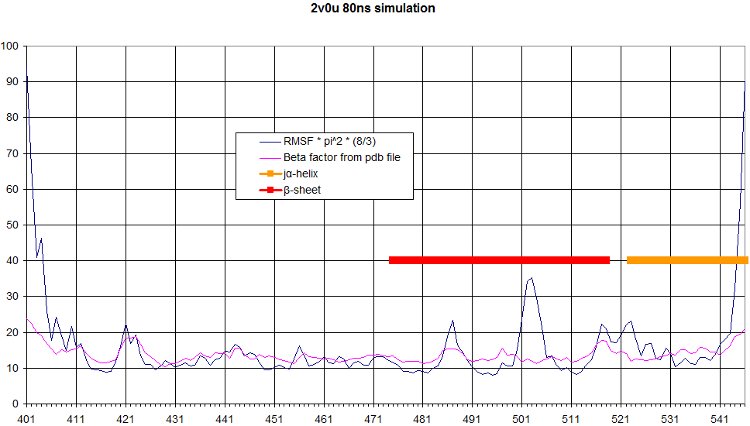
The comparison of the RMSF over the simulation to the beta factor measured during crystallography is a nice validation of our simulation. We get quite similar curves, with some differences at one end of the protein. We see in the movie that this part moves a lot.
Here we computed the oscillation of RMSF in function of residue number, and we highlight the interesting part of our protein, namely the beta sheet and the alpha helix.
Analysis of the simulation
We have organized our analysis on 2 main ideas:
- Find a structural change in the Jα helix based on the simulation using namd.
- Find residues showing different comportment in dark and light state
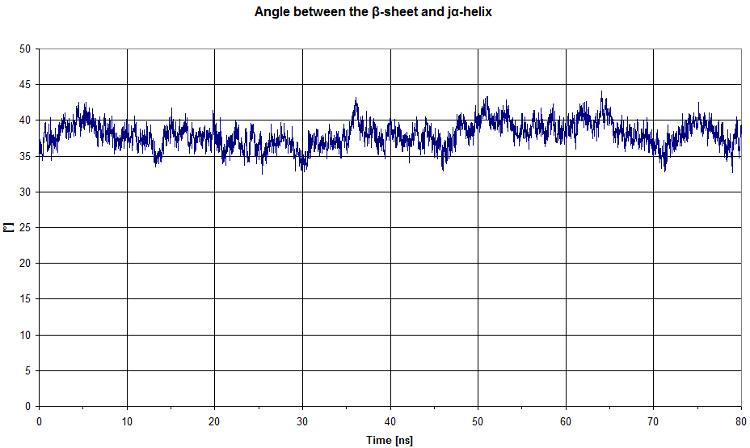
First, we start by looking at the angle between the beta sheet and the Jα helix.
Click here to see the code used in VMD to get angle data
We get a quite constant value. It will be more interesting to compare this graph to the light state.
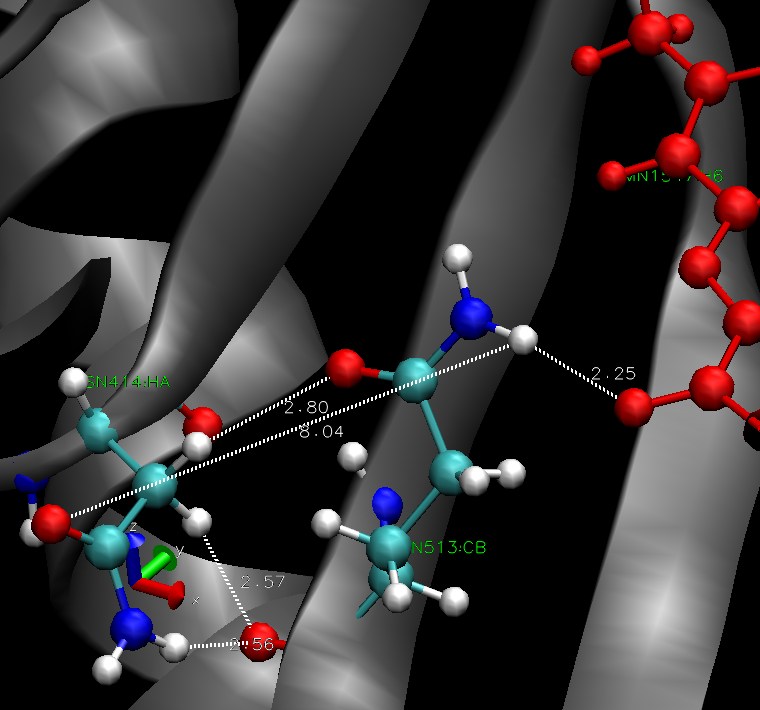
The Jα helix is stabilized by h-bonds to the beta sheet. These bonds are supposed to be disrupted by the conformational change in the dark state. The residues 513 seems to be involved in stabilisation of FMN through hydrogen bonds. We hope it is linked to beta sheet, more precisely the residue 414. There is a picture of the situation, residue ASN 414 is on the left (beta sheet), GLN513 in the middle and the FMN is in red. All the hydrogen bonds we investigated over the simulation are pictured.
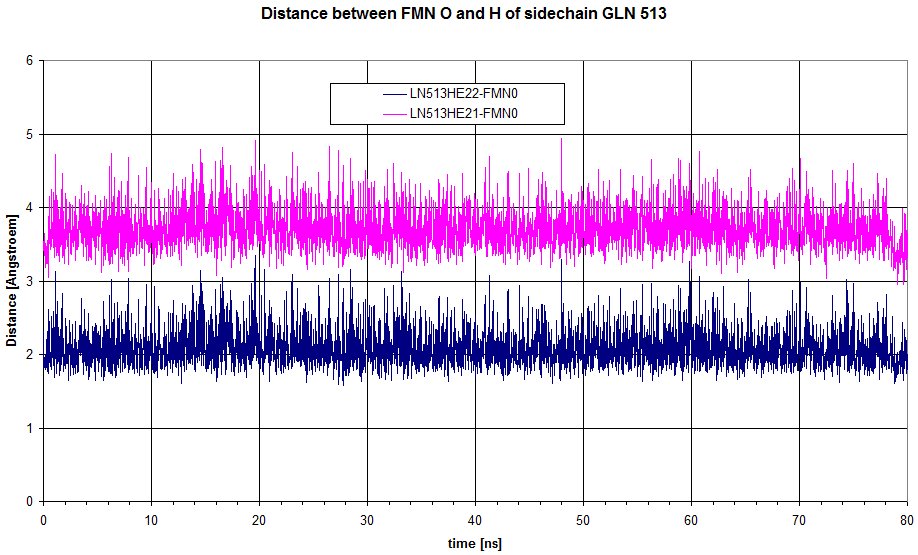
Here is a plot of the distance between the 2 hydrogens from sidechain of GLN513 to the oxygen of FMN. HE22 is definitely involved in an hydrogen bond, but doesn't move enough to loose the interaction.
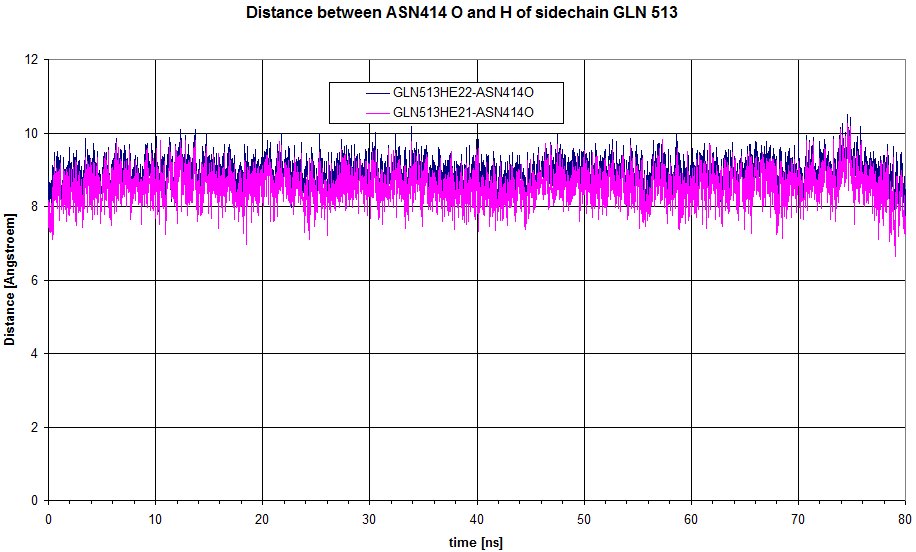
We can have a look at the distance between O of sidechain of ASN 414 to Hs of GLN513.
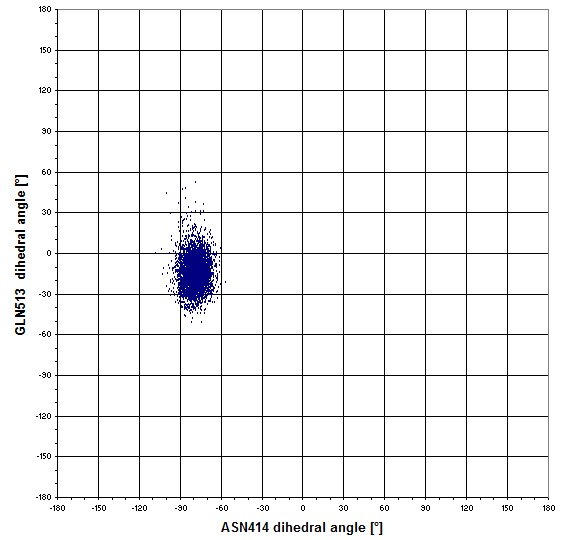
Maybe the sidechains of GLN513 moves in regard the the position of ASN414? -> no, there is a single combination on the next graph.
II. Equilibration of light state
After having modified some parameters in the parameter files, here is the movie concerning the light state of the protein with the FMN: Light State
Light state
Analysis
1. Validation of our run
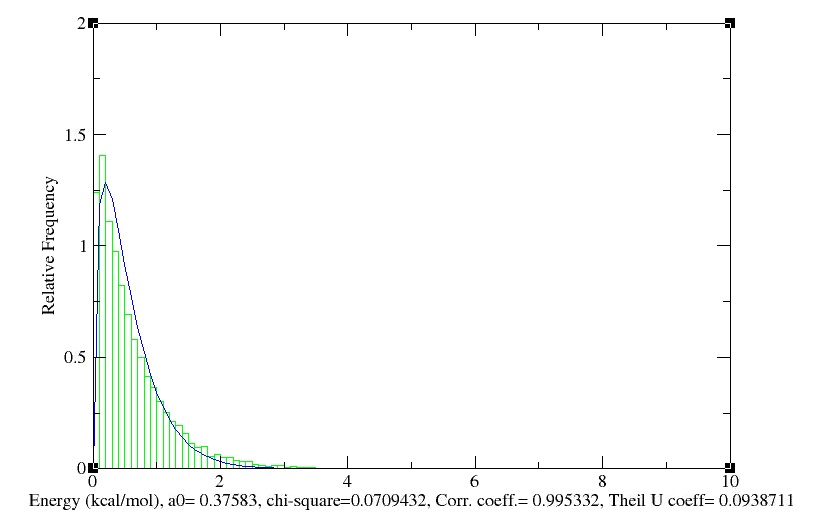
Maxwell-Boltzmann Energy Distribution
We obtain the following histogramm!
Temperature
Using EXCEL, we obtain the following graph, which represents the evolution of the temperature in function of time:
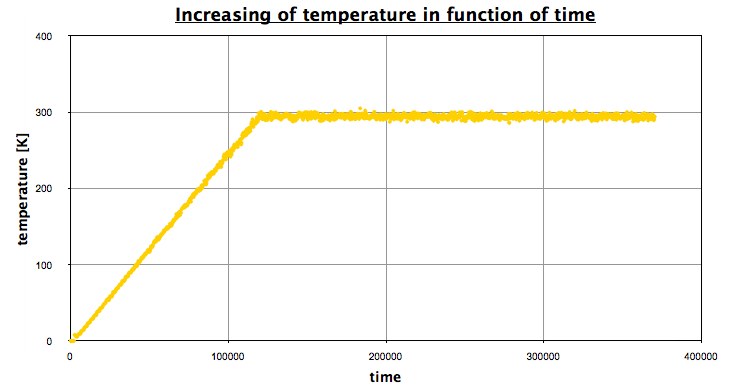
The first part corresponds the the heating, then we let the system reach an equilibrium (NPT state), a NVT portion, and finally a NPT portion again. This is exactly what we wanted, and this prove we achieved to find a minimum in energy on the plateau.
Density
Using EXCEL, we obtain the following graph, which represents the evolution of the density in function of time:
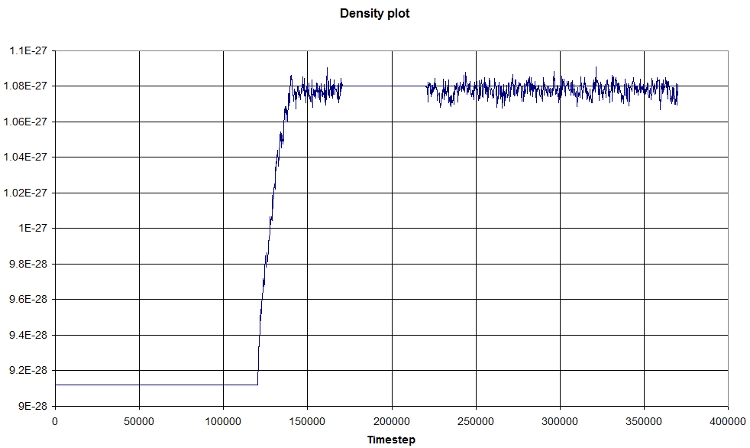
The first part corresponds the the heating, then we let the system reach an equilibrium (NPT state), a NVT portion, and finally a NPT portion again. The density plot follow the general scheme described above, and corroborate our findings.
Pressure
Here is a small plot of pressure and temperature in function of time
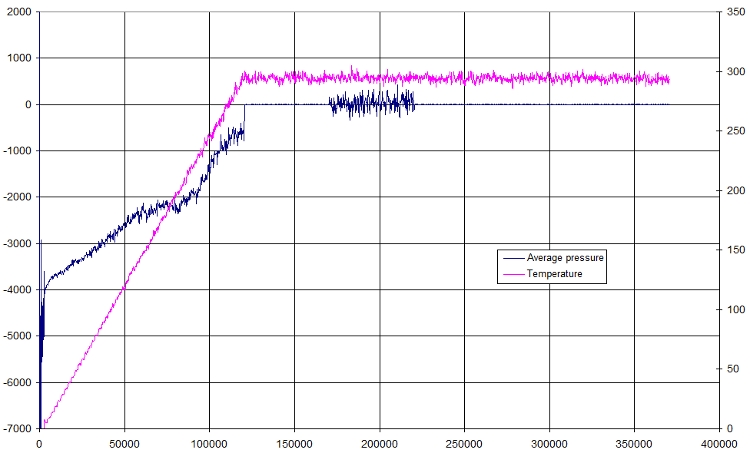
Again, it prove that we achieved our minimization, because the pressure following the increase in temperature, then during the NPT, the pressure remains constant (as stated!). The following portion is a NVT portion: this time, pressure is allowed to fluctuate to compensate other boundaries of volume and temperature. Finally, the last part is again a NPT part, and pressure is also constant.
RMSD
The root mean square deviation is a measure of the differences between predicted values and the values actually observed.
We obtain the following picture:
| RMSD of residue within 3 angström | RMSD of residue within 6 angström |
|---|---|
| We can see that the residues that move the most are the residue number: 425, 451, 453 | |
| We can see that the residues that move the most are the residue number: 424, 425, 464, 468 |
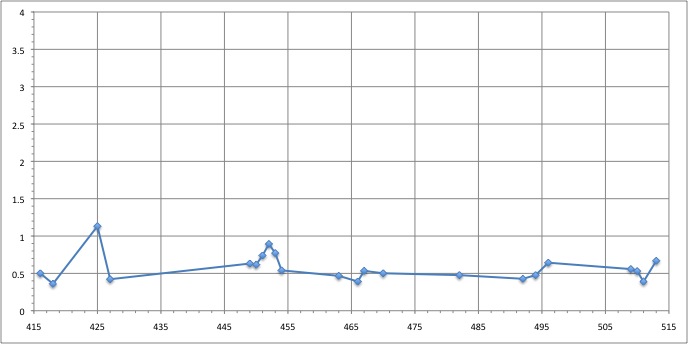
RMSD of residue within 3 angström of the FMN
We can see that the residues that move the most are the residue number: 425, 451, 453
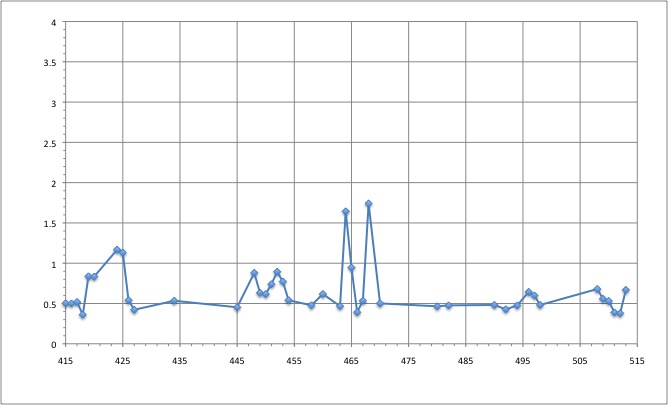
RMSD of residue within 6 angström of the FMN
We can see that the residues that move the most are the residue number: 424, 425, 464, 468
RMSD of selected atoms compared to initial position along time
Here is a fast graph of the output of the average RMSD of the atoms in function of time. It seems normal.
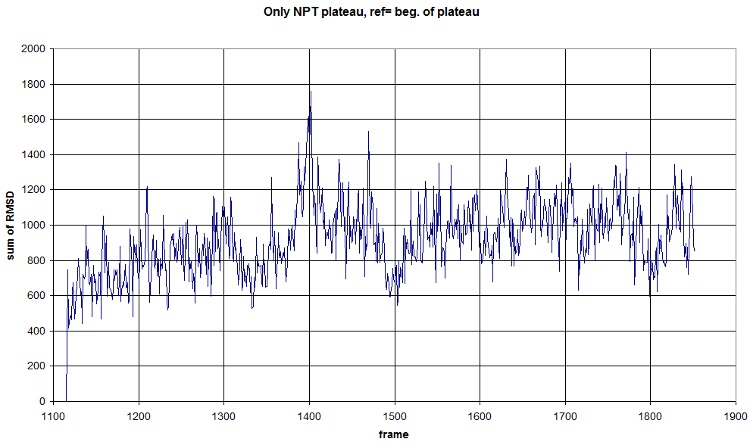
Here is what we got with FIRST_FRAME=1115 REFERENCE_FRAME=1115. Average=921.477, standard deviation=202.1708
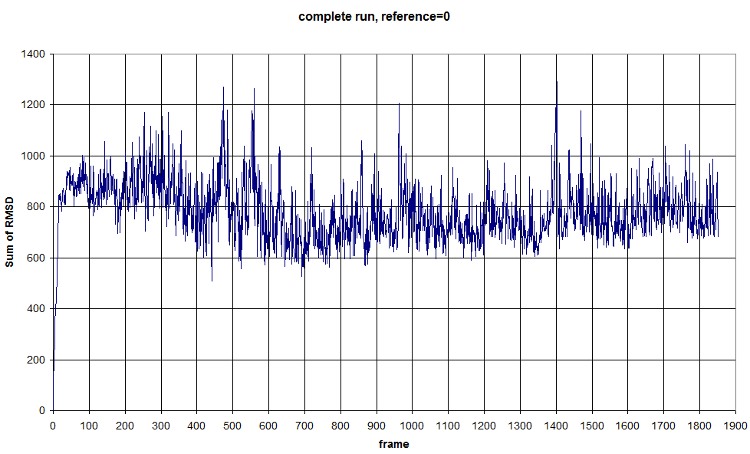
FIRST_FRAME=0 REFERENCE_FRAME=0. The difference of the sum probably comes from the new selection of atoms from the backbone. We should compute an average value to normalize amplitude. (fluctuation is conserved, anyway) Average=781.3913, standard deviation=118.1393
Salt bridges
Here is a plot for one of the bridges. We have to look for the max distance for a salt bridge.
RMSF
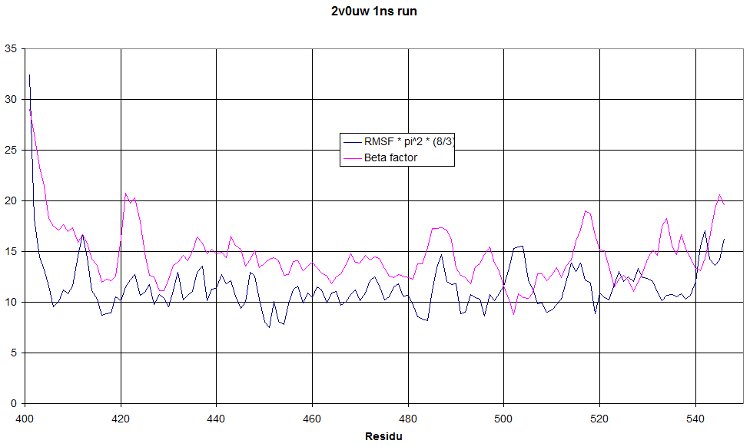
This is a 1 nanosecond NPT run at 300°K. We hope to see a RMSF curve identical to the beta factor. It should only be shifted higher because of the increased temperature. But having a similar tendency would mean our simulation show oscillations similar to what was observed during crystallography. This is really a quite nice validation of our run!
2. Differential analysis
Some useful distances
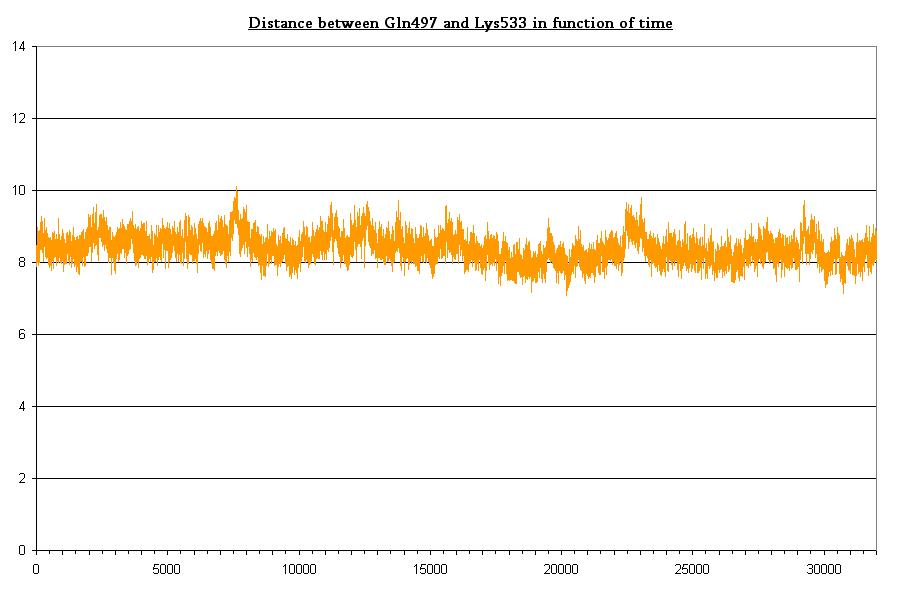
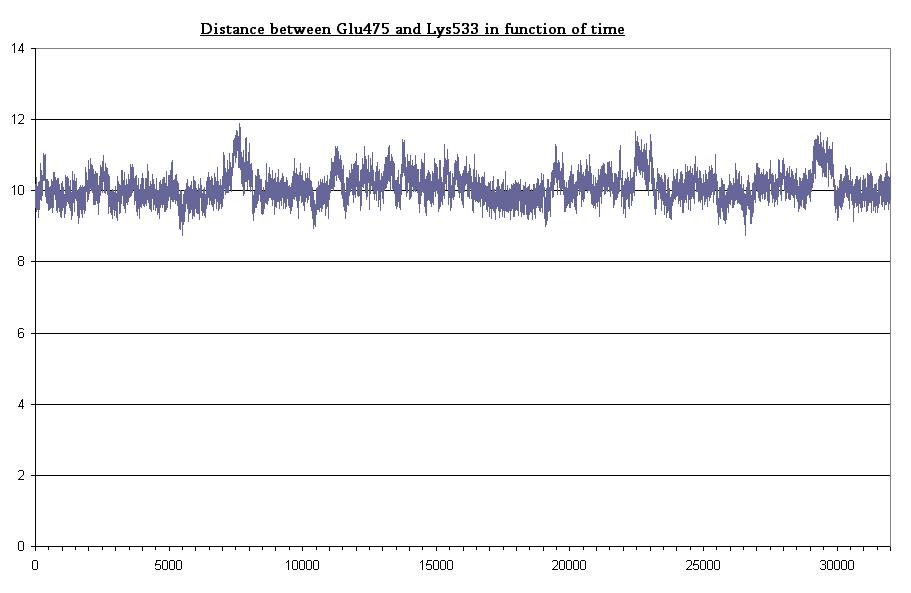
The Jα helix is anchored to the β structure by two H-bond networks the first involving Lys533 (Jα) and the couple Glu475, Gln497 (β structure), the second involving Lys413 (Jα) and Thr535 (β structure). Here we plot the H-bond distances (two in the former and one in the latter).
- Bond between Gln497 and Lys533 in dark state
- Bond between Gln475 and Lys533 in dark state
 "
"