Team:Newcastle/Labwork/31 July 2009
From 2009.igem.org
(→Results for BBa_C0056) |
|||
| (14 intermediate revisions not shown) | |||
| Line 2: | Line 2: | ||
{{:Team:Newcastle/Header}} | {{:Team:Newcastle/Header}} | ||
{{:Team:Newcastle/Left}} | {{:Team:Newcastle/Left}} | ||
| - | + | __NOTOC__ | |
| - | =Lab Session | + | [[Image:Team Newcastle 2009 iGEM ProbationaryP-Sign.PNG|50px|right]] |
| + | =Formal Lab Session - 31st July 2009= | ||
[[Image:Team Newcastle 2009 iGEM 31-07-09 IMG 0231.JPG|250px|center]] | [[Image:Team Newcastle 2009 iGEM 31-07-09 IMG 0231.JPG|250px|center]] | ||
| - | |||
| - | |||
| - | ==Today== | + | |
| - | ===Protocol=== | + | ==Introduction== |
| + | Over the past week we have attempted to transform five BioBricks from the Spring Distributions. Of these BioBricks, ''E. coli'' plus ''cinR'' ([http://partsregistry.org/Part:BBa_C0077 BBa_C0077]) and ''E. coli'' + ''cinI'' ([http://partsregistry.org/Part:BBa_C0076 BBa_C0076]) have not seemed to grow on LB + kan plates suggesting problems with transforming in ''JM109'' cells. With this scenario, we attempted one final transformation but this time using ''DH5-alpha'' ''E. coli'' cells. | ||
| + | |||
| + | However ''JM109'' ''E. coli'' transformations with the ''CinR'' sensitive promoter([http://partsregistry.org/Part:BBa_R0077 BBa_R0077]), ''cI'' coding sequence([http://partsregistry.org/Part:BBa_B1002 BBa_C0056]) and the double terminator([http://partsregistry.org/Part:BBa_B1002 BBa_B1002]) have all worked and grown on LB + amp plates. Three colonies from each set of BioBrick transformants were then used to inoculate 9 x 5ml LB tubes and allowed to grow overnight. | ||
| + | |||
| + | Today's lab session will see us make a decision on BioBricks ''BBa_C0077'' and ''BBa_C0076'' based on whether our final transformation attempt has worked. Today's lab session will also see the team carry out midi-preps from the inoculated LB tubes (one set of three tubes inoculated with ''BBa_C0056'', another set of three tubes inoculated with ''BBa_B1002'' and the final set of three tubes inoculated with ''BBa_R0077''). | ||
| + | |||
| + | ==Practical Outline== | ||
| + | [[Image:Team Newcastle 2009 iGEM 31-07-09 IMG 0227.JPG|200px|thumb|right]] | ||
| + | Here are the objectives for today: | ||
| + | <br> | ||
| + | # Carry out midi-preps on ''E. coli'' cultures containing ''BBa_C0056'', ''BBa_B1002'' and ''BBa_R0077'' | ||
| + | # Check LB and LB + kan plates for any ''DH5-alpha'' cells transformed with ''BBa_C0077'' and ''BBa_C0076'' | ||
| + | ## If unsuccessful, abandon, | ||
| + | ## If successful, prepare 5ml LB inoculations | ||
| + | <br> | ||
| + | |||
| + | ==Procedures== | ||
| + | ===1) Midi-preps of ''BBa_C0056'', ''BBa_B1002'', and ''BBa_R0077''=== | ||
| + | =====Protocol===== | ||
[[Image:Team Newcastle 2009 iGEM 31-07-09 IMG 0229.JPG|200px|thumb|Jess tipping off the supernatant after spinning the overnight cultures in the centrifuge]] | [[Image:Team Newcastle 2009 iGEM 31-07-09 IMG 0229.JPG|200px|thumb|Jess tipping off the supernatant after spinning the overnight cultures in the centrifuge]] | ||
We did a midi prep for the overnight cultures. We followed the protocol from GenElute ([http://www.sigmaaldrich.com/etc/medialib/docs/Sigma/Bulletin/na0200bul.Par.0001.File.tmp/na0200bul.pdf NA0200S_NA0200]). The list of plasmid kits can be accessed from [http://www.sigmaaldrich.com/life-science/molecular-biology/dna-and-rna-purification/hp-plasmid-kits.html Gen Elute's plasmid kits page]. | We did a midi prep for the overnight cultures. We followed the protocol from GenElute ([http://www.sigmaaldrich.com/etc/medialib/docs/Sigma/Bulletin/na0200bul.Par.0001.File.tmp/na0200bul.pdf NA0200S_NA0200]). The list of plasmid kits can be accessed from [http://www.sigmaaldrich.com/life-science/molecular-biology/dna-and-rna-purification/hp-plasmid-kits.html Gen Elute's plasmid kits page]. | ||
| - | ===Procedure=== | + | =====Procedure===== |
To transfer the binding column, we used two eppendorf tubes for each culture. The volume of the eppendorfs were 450ul and we used 45ul of sodium acetate buffer and 315ul of isopropanol for "DNA concentration" step. We centrifuged the tubes at 18000rpm for 30 minutes before adding ethanol to rinse the DNAs. We then centriguged the tubes again for 20 minutes at 13000rpm. If we had used the faster machine we would centrifuge the tubes at 18000rpm for 10 minutes. Since it was a slower machine, we used 20 minutes instead. | To transfer the binding column, we used two eppendorf tubes for each culture. The volume of the eppendorfs were 450ul and we used 45ul of sodium acetate buffer and 315ul of isopropanol for "DNA concentration" step. We centrifuged the tubes at 18000rpm for 30 minutes before adding ethanol to rinse the DNAs. We then centriguged the tubes again for 20 minutes at 13000rpm. If we had used the faster machine we would centrifuge the tubes at 18000rpm for 10 minutes. Since it was a slower machine, we used 20 minutes instead. | ||
At the end we air-dried the pellets until the ethanol evaporated. We then added 50ul of PCR water to each eppendorf tube and mixed the solution well. Two eppendorf tubes from the same colony were combined. | At the end we air-dried the pellets until the ethanol evaporated. We then added 50ul of PCR water to each eppendorf tube and mixed the solution well. Two eppendorf tubes from the same colony were combined. | ||
| - | ===Results=== | + | =====Results===== |
[[Image:Team Newcastle 2009 iGEM 31-07-09 IMG 0237.JPG|200px|thumb|Goksel adding one of the agents needed for the plasmid midiprep]] | [[Image:Team Newcastle 2009 iGEM 31-07-09 IMG 0237.JPG|200px|thumb|Goksel adding one of the agents needed for the plasmid midiprep]] | ||
Finally we measured the concentration of the DNA in the tubes using spectrometer. We used a sensitive pipet to wash the machine wth 2ul of water twice. We then loaded and measured the concentration for each tube. Between the steps we cleaned the machine with tissues. At the end we loaded with water again for a final cleaning procedure. | Finally we measured the concentration of the DNA in the tubes using spectrometer. We used a sensitive pipet to wash the machine wth 2ul of water twice. We then loaded and measured the concentration for each tube. Between the steps we cleaned the machine with tissues. At the end we loaded with water again for a final cleaning procedure. | ||
| Line 27: | Line 45: | ||
<br> | <br> | ||
| - | ====Results for BBa_C0056==== | + | =====Results for BBa_C0056===== |
We had high DNA concentration for the first tube. | We had high DNA concentration for the first tube. | ||
| Line 36: | Line 54: | ||
[[Image:Team_Newcastle_iGEM_2009_31-07-09_no_1.JPG|550px]] | [[Image:Team_Newcastle_iGEM_2009_31-07-09_no_1.JPG|550px]] | ||
| - | ====Results for BBa_B1002 ==== | + | =====Results for BBa_B1002 ===== |
We had low DNA concentration for the second tube. | We had low DNA concentration for the second tube. | ||
| Line 45: | Line 63: | ||
[[Image:Team_Newcastle_iGEM_2009_31-07-09_no_2.JPG|550px]] | [[Image:Team_Newcastle_iGEM_2009_31-07-09_no_2.JPG|550px]] | ||
| - | ====Results for BBa_R0077==== | + | =====Results for BBa_R0077===== |
The concentration of DNA: 53.8 ng/uL | The concentration of DNA: 53.8 ng/uL | ||
The ratio of DNA concentration to protein concentration(260/280): 1.75 | The ratio of DNA concentration to protein concentration(260/280): 1.75 | ||
| Line 51: | Line 69: | ||
[[Image:Team_Newcastle_iGEM_2009_31-07-09_no_3.JPG|550px]] | [[Image:Team_Newcastle_iGEM_2009_31-07-09_no_3.JPG|550px]] | ||
| + | <br> | ||
| + | ===2) ''BBa_C0077'' and ''BBa_C0076'' Transformants === | ||
| + | [[Image:Team Newcastle 2009 iGEM 31-07-09 IMG 0225.JPG|425px|center]] | ||
| + | <br> | ||
| + | At last, colonies were found on the plates which contained ''DH5-alpha'' transformed with ''BBa_C0077'' and ''BBa_c0076''! The 2 LB-alone plates for both ''BBa_C0077'' and ''BBa_C0076'' transformants was completely covered in a lawn of bacteria. The LB + kan plate with the ''DH5-alpha'' cells transformed with ''BBa_C0077'' yielded about 5 colonies whereas the LB + kan plate with the ''BBa_C0076'' transformants yielded 100+ colonies. | ||
| + | However the plates will be stored in the fridge, over the weekend, for LB inoculations and future mini-preps to take place later (hopefully in Monday's lab session). | ||
| + | {{:Team:Newcastle/Project/Labwork/CalTemplate}} | ||
{{:Team:Newcastle/Footer}} | {{:Team:Newcastle/Footer}} | ||
{{:Team:Newcastle/Right}} | {{:Team:Newcastle/Right}} | ||
Latest revision as of 17:26, 17 October 2009
Formal Lab Session - 31st July 2009
Introduction
Over the past week we have attempted to transform five BioBricks from the Spring Distributions. Of these BioBricks, E. coli plus cinR ([http://partsregistry.org/Part:BBa_C0077 BBa_C0077]) and E. coli + cinI ([http://partsregistry.org/Part:BBa_C0076 BBa_C0076]) have not seemed to grow on LB + kan plates suggesting problems with transforming in JM109 cells. With this scenario, we attempted one final transformation but this time using DH5-alpha E. coli cells.
However JM109 E. coli transformations with the CinR sensitive promoter([http://partsregistry.org/Part:BBa_R0077 BBa_R0077]), cI coding sequence([http://partsregistry.org/Part:BBa_B1002 BBa_C0056]) and the double terminator([http://partsregistry.org/Part:BBa_B1002 BBa_B1002]) have all worked and grown on LB + amp plates. Three colonies from each set of BioBrick transformants were then used to inoculate 9 x 5ml LB tubes and allowed to grow overnight.
Today's lab session will see us make a decision on BioBricks BBa_C0077 and BBa_C0076 based on whether our final transformation attempt has worked. Today's lab session will also see the team carry out midi-preps from the inoculated LB tubes (one set of three tubes inoculated with BBa_C0056, another set of three tubes inoculated with BBa_B1002 and the final set of three tubes inoculated with BBa_R0077).
Practical Outline
Here are the objectives for today:
- Carry out midi-preps on E. coli cultures containing BBa_C0056, BBa_B1002 and BBa_R0077
- Check LB and LB + kan plates for any DH5-alpha cells transformed with BBa_C0077 and BBa_C0076
- If unsuccessful, abandon,
- If successful, prepare 5ml LB inoculations
Procedures
1) Midi-preps of BBa_C0056, BBa_B1002, and BBa_R0077
Protocol
We did a midi prep for the overnight cultures. We followed the protocol from GenElute ([http://www.sigmaaldrich.com/etc/medialib/docs/Sigma/Bulletin/na0200bul.Par.0001.File.tmp/na0200bul.pdf NA0200S_NA0200]). The list of plasmid kits can be accessed from [http://www.sigmaaldrich.com/life-science/molecular-biology/dna-and-rna-purification/hp-plasmid-kits.html Gen Elute's plasmid kits page].
Procedure
To transfer the binding column, we used two eppendorf tubes for each culture. The volume of the eppendorfs were 450ul and we used 45ul of sodium acetate buffer and 315ul of isopropanol for "DNA concentration" step. We centrifuged the tubes at 18000rpm for 30 minutes before adding ethanol to rinse the DNAs. We then centriguged the tubes again for 20 minutes at 13000rpm. If we had used the faster machine we would centrifuge the tubes at 18000rpm for 10 minutes. Since it was a slower machine, we used 20 minutes instead.
At the end we air-dried the pellets until the ethanol evaporated. We then added 50ul of PCR water to each eppendorf tube and mixed the solution well. Two eppendorf tubes from the same colony were combined.
Results
Finally we measured the concentration of the DNA in the tubes using spectrometer. We used a sensitive pipet to wash the machine wth 2ul of water twice. We then loaded and measured the concentration for each tube. Between the steps we cleaned the machine with tissues. At the end we loaded with water again for a final cleaning procedure.
Tubes with plasmid DNAs were then placed to the -20C freezer.
Results for each tube are as below.
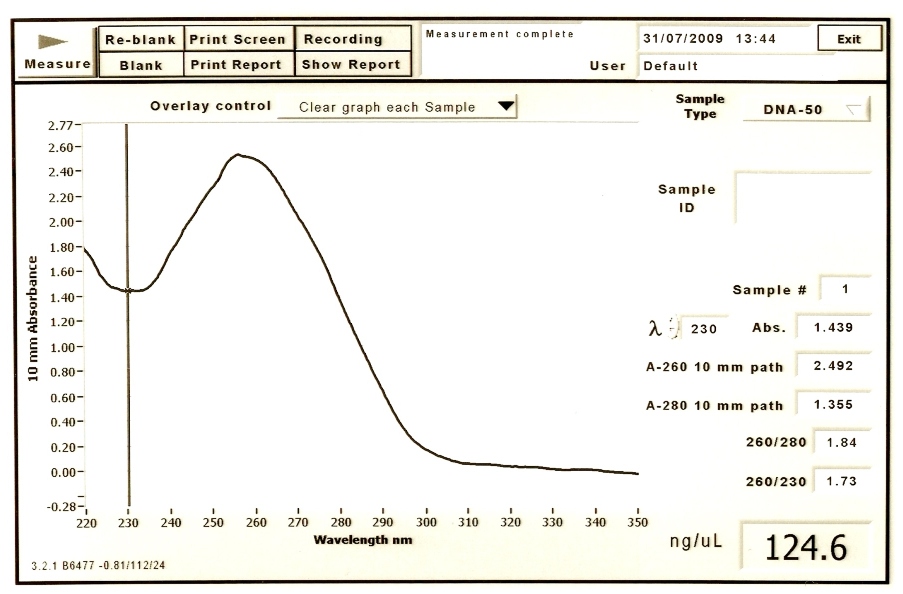
Results for BBa_C0056
We had high DNA concentration for the first tube.
The concentration of DNA: 124.6 ng/uL The ratio of DNA concentration to protein concentration(260/280): 1.84 The ratio of protein concentration to RNA concentration(260/230): 1.73
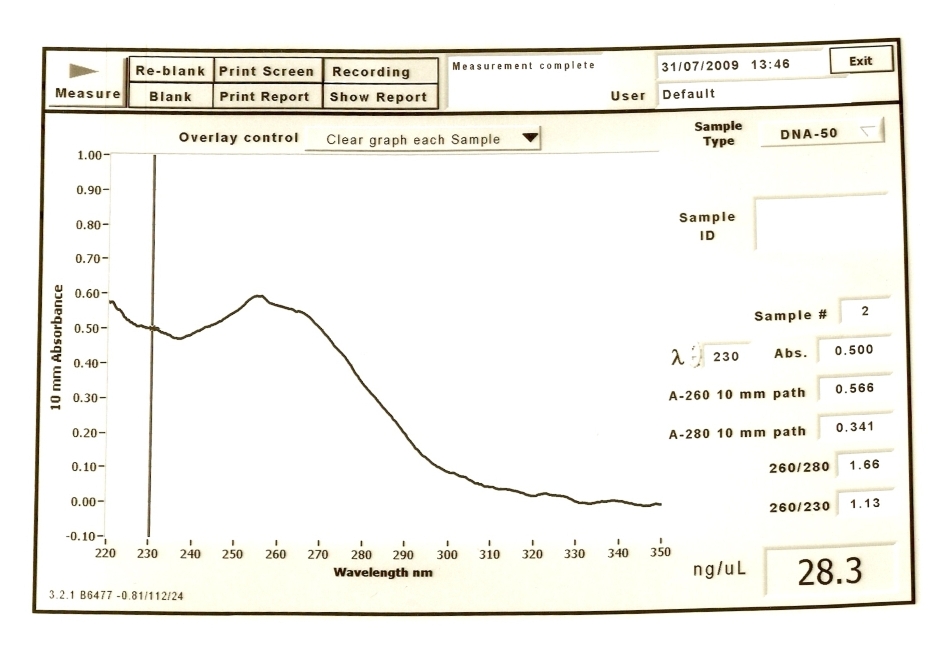
Results for BBa_B1002
We had low DNA concentration for the second tube.
The concentration of DNA: 28.3 ng/uL The ratio of DNA concentration to protein concentration(260/280): 1.66 The ratio of protein concentration to RNA concentration(260/230): 1.13
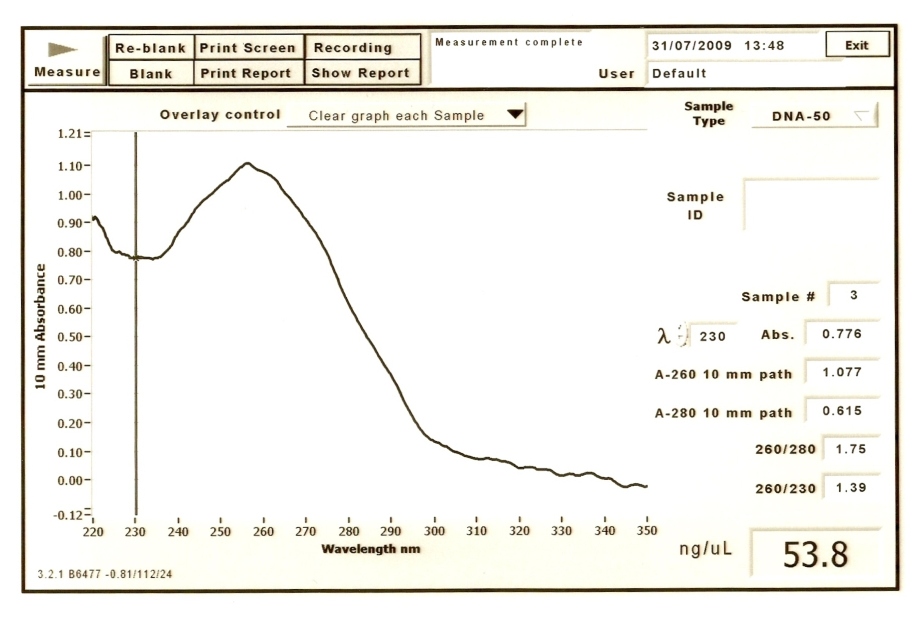
Results for BBa_R0077
The concentration of DNA: 53.8 ng/uL The ratio of DNA concentration to protein concentration(260/280): 1.75 The ratio of protein concentration to RNA concentration(260/230): 1.39
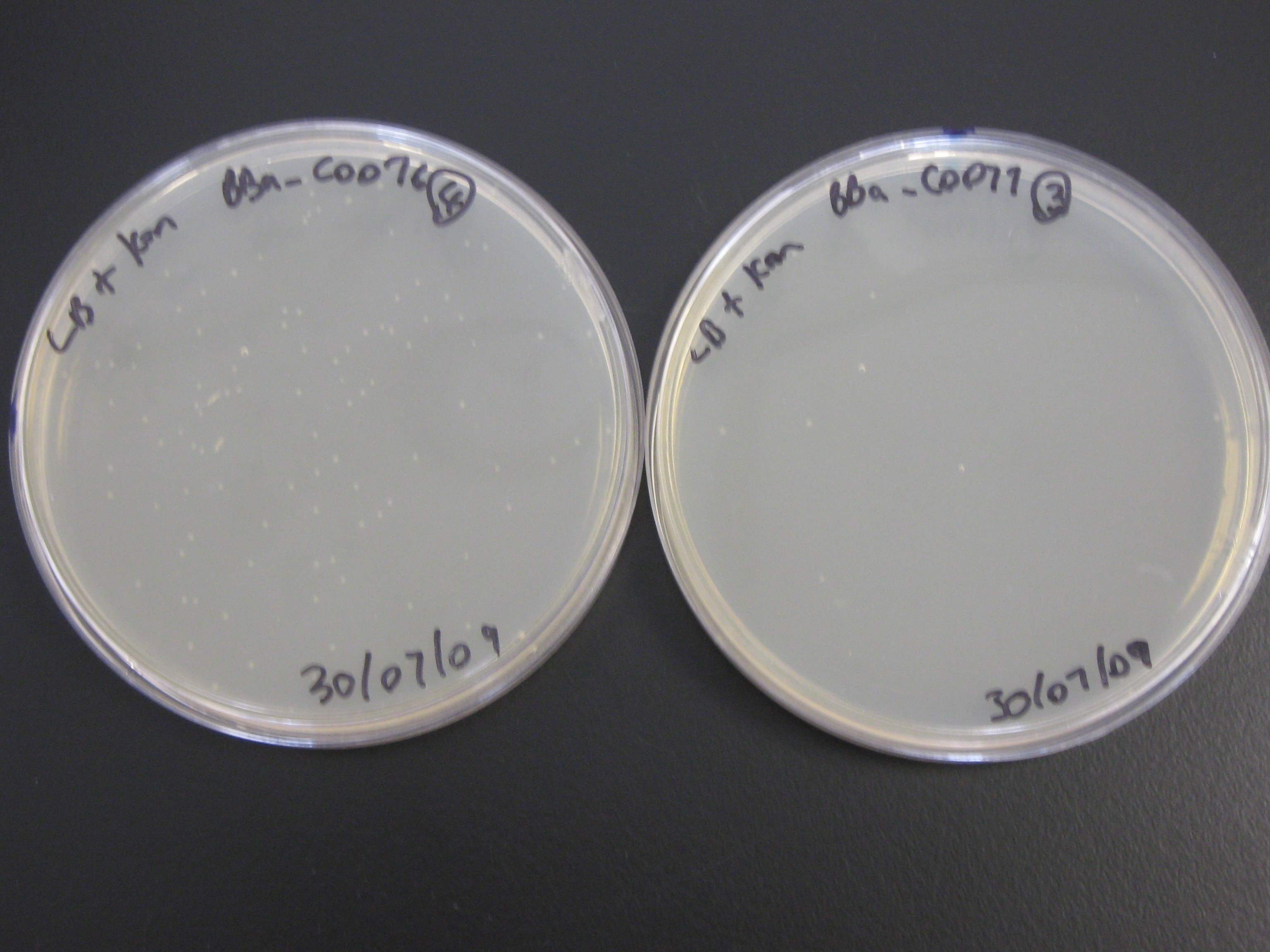
2) BBa_C0077 and BBa_C0076 Transformants
At last, colonies were found on the plates which contained DH5-alpha transformed with BBa_C0077 and BBa_c0076! The 2 LB-alone plates for both BBa_C0077 and BBa_C0076 transformants was completely covered in a lawn of bacteria. The LB + kan plate with the DH5-alpha cells transformed with BBa_C0077 yielded about 5 colonies whereas the LB + kan plate with the BBa_C0076 transformants yielded 100+ colonies.
However the plates will be stored in the fridge, over the weekend, for LB inoculations and future mini-preps to take place later (hopefully in Monday's lab session).
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
News
Events
- 20 – 21 June 2009 - Europe workshop (London)
- 23 – 24 June 2009 - UK iGEM meetup (Edinburgh)
- 23 October Practice Presentation (Newcastle)
- 23 October T-shirts are ready
- 27 October Practice Presentation (Sunderland)
- 27 October Poster is ready
- 30 October – 2 November 2009 - Jamboree (Boston)
Social Net
- Newcastle iGEM Twitter
- [http://www.facebook.com/home.php#/group.php?gid=131709337641 Newcastle on Facebook]
- [http://www.youtube.com/user/newcastle2009igem Newcastle Youtube Channel]
 "
"