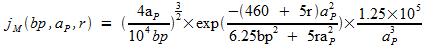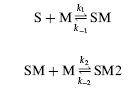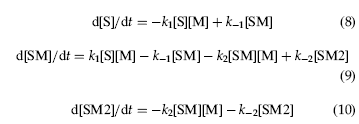Team:BIOTEC Dresden/Modeling v2
From 2009.igem.org
Looping probability simulation
Interpolation formula [Stewart et. al. 1999] allows to calculate the probability of two cites of DNA to meet by looping.
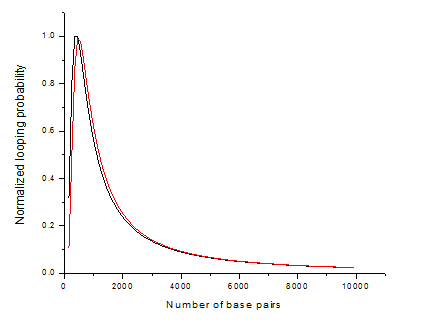
The key parameters of the model are the distance between cites (in number of base pairs), persistence length of DNA (in nm) and the proximity of two DNA ends necessary for looping to occur (nm). Probability is expressed as the local molar concentration of one site with respect to the other. On the Fig. 1 results of a simulation according to this formula are shown. Persistence length is considered to be 50 nm [Hagerman, 1988], proximity of two cites is 10 nm (black line), compared to 0 nm (red line). Probability is normalized with respect to the maximum value (0.1194 10^-6 M) reached at ~400 bp. Both lines are consistent with experiment and show no looping below the persistence length (~150 bp). Our simulation (performed with MATLAB) proves that the optimal distance for looping to occur is ~400 bp and the probability of two cites to meet decreases with growing distance between them. As can be seen from the picture, accounting for non-zero distance between to cites to form a loop, gives higher probability of looping to happen.
Theory behind FLP-FRT recombination
In genetics, FLP-FRT recombination is a site-directed recombination technology used to manipulate an organism's DNA under controlled conditions in vivo. It is analogous to Cre-Lox recombination. It involves the recombination of sequences between short Flippase Recognition Target (FRT) sites by the Flippase recombination enzyme (FLP or Flp) derived from the 2µ plasmid of the baker's yeast Saccharomyces cerevisiae.
The 34bp long FRT site sequence is : 5'-GAAGTTCCTATTCtctagaaaGTATAGGAACTTC-3'. Flippase (flp) binds to the 13-bp 5'-GAAGTTCCTATTC-3' and to the reverse complement of 5'-GTATAGGAACTTC-3' (5'-GAAGTTCCTATAC-3'). The FRT site is cleaved just before 5'-tctagaaa-3', the 8bp asymmetric core region, on the top strand and behind this sequence on the bottom strand.[1]
Several variant FRT sites exist. Recombination can occur between two identical FRT sites but generally not between non-identical FRT sites.
Many available constructs include the sequence 5'-GAAGTTCCTATTCC-3' immediately upstream the FRT site (resulting in 5'-GAAGTTCCTATTCCGAAGTTCCTATTCtctagaaaGTATAGGAACTTC-3') but this sequence is dispensable for recombination.
Because the recombination activity can be targeted to only one target organ, or a low level of recombination activity can be used to consistently alter the DNA of only a subset of cells, FLP-FRT can be used to construct genetic mosaics in multicellular organisms. Using this technology, the loss or alteration of a gene can be studied in one target organ of interest, even if experimental animals could not survive the loss of the gene in other organs.
The effect of altering a gene can also be studied over time, by using an inducible promoter to trigger the recombination activity late in development - this prevents the alteration from affecting overall development of an organ, and allows single cells lacking the gene to be compared to normal neighboring cells in the same environment.
http://www.recombineering.net/img/A202_01.gif
pCAGGS-FLPe-IRESpuro expression vector.
Unlike transcriptional regulation, this method gives true all-or-none induction due to covalent modification of DNA by Flp recombinase. As the wild-type enzyme is very sensitive to temperature, we are using the F70L variant because its degradation is sufficiently slow to give a time course. Determining the transfer curve of inter-FRT site distance versus average recombination time allows the onset of gene expression to be predicted. We then apply this [http://partsregistry.org/cgi/partsdb/pgroup.cgi?pgroup=iGEM2009&group=BIOTEC_Dresden Flp reporter system] as a powerful PoPS measurement device.
Recombination pathway of FLP: design of mathematical model
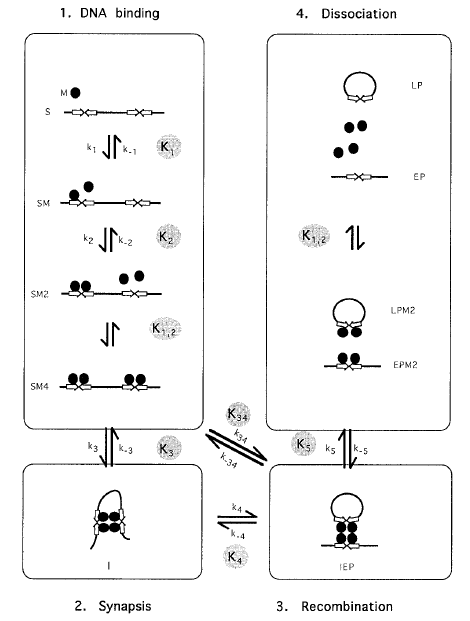
The recombination pathway upon which the mathematical model for recombination was based is shown in Figure 1. The model describes an excision reaction on a linear DNA substrate. The steps of DNA binding are well characterised for FLP. The enzyme binds the inverted repeat target site first as a monomer, which is then joined by a second monomer to form a dimer Andrews et al., 1987; Hoess et al., 1984; Mack et al., 1992). In the model, we have assumed that the protein is monomeric in solution, based on the behaviour of FLP and Cre in sucrose density gradients under similar buffer conditions to those used in this study (Abremski & Hoess, 1984; Qian et al., 1990).
Figure 1 Steps in FLP and Cre
excision recombination reaction.
Inverted repeat target sites are
shown as open arrows. Recombinase
monomers are shown as filled
circles. Each step in the reaction is
reversible and has a forward and a
backward rate, indicated by small
arrows. The forward and backward
rate constants for each step are
indicated on the Figure beside the
arrows (small kn or k-n). The equilibrium
constant for the conversion
of one complex to another is given
by the quotient of the backward
and forward rate constants. Equilibrium
constants are indicated (Kn).
Names of species used for mathematical
modelling are shown
beside each complex (Ringrose et al., 1998, reproduced with permission).
In our scheme, the second-order association rate
constants for the binding of the first and second
monomers are named k1 and k2, respectively. This
process must occur twice to occupy both target
sites of the excision substrate. In the model, all
DNA binding and dissociation steps are described
by k1 and k2, and their corresponding first-order
dissociation rate constants, k-1 and k-2 (Figure 1).
A reduced model, describing the sequential binding
of recombinase monomers to a single full site
target comprising two half sites, in terms of the
four parameters k1, k2, k-1 and k-2 is described by
equations (7) to (10) (see below).
After DNA binding, the next step in the pathway
is synapsis. FLP synapsis occurs by random collision
(Beatty et al., 1986). In Figure 1, synapsis of the fully occupied
excision substrate is described by a single first-order
rate constant, k3. The model only allows for
intramolecular synapsis, although in reality intermolecular
recombination can also occur. However,
for recombination assays, standard experimental
conditions have been used, under which the frequency of
intermolecular recombination is negligible. The
multiple steps of catalysis are well characterised
for FLP (for reviews, see Stark et al., 1992;
Jayaram, 1994; Sadowski, 1995), and are described
in Figure 1 by a single pair of rate constants, k4
and k-4, the forward and back rates of catalysis,
respectively.
At present, we do not have a means of accurately
measuring the formation of the synaptic
complex. For this reason, we have further simplified the model by combining the constants k3 and
k4 to give an apparent constant, k34,
describing the rate of conversion of the fully
bound substrate (SM4) into the excised synaptic
complex (IEP) (Figure 1). The back rate, k-34,
describes the reverse process. The relationship
between k34, k-34 and their components k3, k-3, k4
and k-4 is given by:
In this scheme, the dissociation of the synapse is
represented as a reversal of its assembly: dissociation
gives rise first to two products, each of
which is bound by a recombinase dimer (Figure 1).
This process is described by the first-order rate
constant, k5. The dissociation of recombinase from
DNA is assumed to occur in a stepwise manner,
and is described in the model by the rate constants
k-1 and k-2 (Figure 1). This assumption is based on
the most simple and logical pathway. The dissociation
mechanism of FLP has not been
studied extensively, and there is disagreement in
the literature regarding the steps involved. In experiments with synthetic Holliday
junctions, it has been reported that the resolution
of such structures requires two (Dixon &
Sadowski, 1993), three (Qian & Cox, 1995) or four
molecules of FLP (Lee et al., 1996). Based on experiments
using full recombination substrates, Waite
& Cox (1995) proposed a mechanism for FLP dissociation
in which one or more monomers leave
the synapse after recombination whilst others
remain bound for longer. In the absence of a consensus
on the mechanism of dissociation, Ringrose et al. (1998) proposed
the mechanism shown in Figure 1, and point
out that other mechanisms could easily be incorporated
into the model by modification of
equations (11) to (24) (see below).
The DNA binding rate constants k1, k-1, k2, k-2, and their corresponding equilibrium constants K1 and K2, have been directly measured using the gel mobility shift assay. If the DNA binding constants are known, this leaves two pairs of unknown rate constants: k34 and k-34, and k5 and k-5 (Figure 1). The mathematical model describes a general excision recombination reaction in which four protein monomers are required to reversibly recombine a single substrate, giving two products. The values of all rate constants, and of protein and substrate input can be varied to represent specific cases.
Simulation of FLP recombination
Figure 8. Simulation of FLP recombination. a) Substrate
titration (see below). The
input values for protein, substrate, k1, k-1, k2 and k-2 are
taken from Ringrose et al., (1998). Simulated
curves for total nM excision product
at three minutes and 60 minutes
were fitted to the experimental
data for FLP by simultaneous optimisation
of the rate constants k34, k-34,
k5 and k-5. Continuous lines, simulated
recombination at three and
60 minutes; 25.6 nM FLP, 0.05 to
10 nM substrate. Open circles,
recombination observed at three
minutes. Filled circles,
recombination observed at 60 minutes. b)
Time course. The optimised parameters in Ringrose et al.,(1998) were used to
simulate time course curves for
FLP. Continuous
line, simulated recombination time
course at 25.6 nM FLP, 0.4 nM substrate;
filled circles, data from time
course experiment at 25.6 nM FLP, 0.4 nM substrate. c) Protein titration. Recombination time course curves were simulated for FLP at various protein inputs, with 0.4 nM substrate input. The simulated recombination after 60 minutes is plotted against protein concentration. Continuous line, simulated recombination parameters as in Ringrose et al.,(1998). Open circles, data points from protein titration experiment.
Mathematical modelling I: DNA binding
Using the nomenclature of Figure 1, the binding of recombinase monomers (M) to a full site substrate (S) is described by the kinetic equations:
k1 and k-1 are the association and dissociation rate constants, respectively, for the species SM. k2 and k-2 are the association and dissociation rate constants for the species SM2. For the DNA binding model the assumption is made that the on and off rates for each half site, a and b, are identical. The evolution of species M, S, SM and SM2 over time is described by the differential equations:
dt is the time interval in seconds. The terms in square brackets refer to molar concentrations at time, t. Equations (7) to (10) were implemented in R code, and solved by finite difference numerical integration. The rate constants k1, k-1, k2 and k-2 were determined by fitting the computed time courses of the SM and SM2 concentrations to the experimentally observed values simultaneously. Starting values of k1, k-1, k2 and k-2 were determined by choosing values which gave an approximate agreement with the experimental data. Using these starting values, the model of equations (7) to (10) was integrated in time using a semi-implicit Euler scheme (Press et al., 1992). The time courses were fitted to the experimental data by minimising an error function using the method of Powell (1965), a derivative-free version of the conjugate gradient method. The error function was constructed by summing the squared deviations of the experimental and computed SM and SM2 concentrations. As each set of rate constants corresponds to a unique solution of equations (7) to (10), and hence a given value of the error function, optimal values of the rate constants correspond to optimal agreement between experimental and modelled time courses.
Mathematical modelling II: the recombination reaction
The proposed pathway of recombination is shown in Figure 1. The names of most species and rate constants used in the model are shown in the Figure. Additional species described by the model and not shown in Figure 1 are as follows. The distinction is made in the model between SM2A (equation (14)), which has a single monomer bound at each target site, and SM2B (equation (15)), which has two monomers bound at one site, leaving the other site unoccupied. Only SM2B is indicated in Figure 1. Either SM2A or SM2B can give rise to SM3 (equation (16)), in which one site is bound by two monomers while the other is bound by only one monomer. Later species which are also not shown in Figure 1 describe the stepwise dissociation of monomers from DNA. EPM2 and LPM2 (Figure 1), both with two monomers bound, can each release a single monomer to give rise to EPM and LPM respectively (equations (21) and (22)). The release of the last bound monomer from EPM and LPM gives rise to EP and LP (equations (23) and (24); Figure 1). Based on classical kinetic equations, the differential equations describing the rate of change of each species over time (in seconds) are as follows:
Equations (11) to (24) were implemented and solved
as described above for the DNA binding model. To find
values for the unknown rate constants k34, k-34, k5 and
k-5, computed time courses were fitted to experimental
data by varying the input values of unknown rate constants.
The quantity taken to represent the total excision
product, EPtot, is given by IEP + EPM2 + EPM + EP.
This quantity is considered to describe the experimentally
observed excision product because only the linear
excision product, and not the circle, is labelled and quanti
fied. In addition, because recombination reactions are
terminated with proteinase K, the DNA seen as excision
product includes the DNA within the excised synaptic
complex (IEP in the model) and all subsequent bound
 "
"