Team:UNIPV-Pavia/Notebook/Week1Jun
From 2009.igem.org

|
|
|
|
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
Week from June 1st, to June 7th, 2009
 Previous Week
Previous Week
|
Next Week

|
June, 3rd
- We received lactose monohydrate, M9 minimal salts and Thiamine Hydrochloride from Sigma.
- DNA resuspension from iGEM 2009 plates. We resuspended the two parts we needed to re-built the inconsistent BioBrick Q04400, the five commonly used RBSs and a 3OC6-HSL inducible measurement system for GFP test at Tecan F200:
|
|
- Transformation of resuspended DNA (2 ul) in TOP10 E. coli, plated transformed bacteria and incubated the plates overnight at 37°C.
- We streaked a plate with a 2008 glycerol stock containing E0240 under the control of a constitutive promoter (we call this construct "01") in order to use it in the fluorescence test at Tecan F200 on June 5th.
June, 4th
- All the overnight plates showed colonies. 01 plate showed green fluorescence under UV rays, as we expected because GFP was expressed constitutively!
Unfortunately, B0034 and B0032 plates also showed a very small amount of unexpected red colonies...(@_@!?) These two parts had been resuspended from iGEM 2009 plate 1, just like Berkeley constitutive promoters, but it is not clear how they could be contaminated...
- Anyway, we decided to continue our work and we picked one colony from the following plates:
|
and infected 1 ml of LB + Amp. We incubated the inocula at 37°C, 220 rpm for 5 and 1/2 hours. Of course, we carefully avoided to pick red colonies from B0034 and B0032 plates:) Sequencing checks will tell us if our parts are correct.
- Glycerol stocks for all the grown cultures (except for 01, because we already had it).
- NOTE: we re-picked colonies from J23100, J23101 and J23118 plates because data analysis of experiment 1 at Tecan F200 showed that J23101 promoter seemed stronger than J23100. This is not in accordance to the ranking of the promoters (http://partsregistry.org/Promoters/Catalog/Anderson), so we decided to repeat the test at Tecan F200 with brand new colonies and to store these new glycerol stocks at -80°C.
|
|
|
|
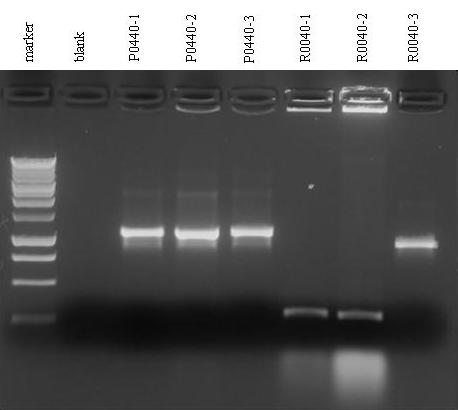
- Gel results: the 3 colonies of P0440 all have the plasmid with the correct length of the insert (1078 bp), while there have been problems with R0040 colonies. R0040-1 and R0040-2 show the correct length of the insert (292 bp) with a high weight contaminant and R0040-3 show an unexpected wrong length. Next week we are going to digest P0440 and R0040 to perform their assembly, so we will check the actual plasmid and insert length again.
June, 5th
Preparation of the second experiment with Tecan F200
|
|
|
Experiment with Tecan F200
June, 6th
Experiment with Tecan F200
- Results check
 Previous Week
Previous Week
|
Next Week

|
 "
"