Team:Warsaw/Project/invasion
From 2009.igem.org
Contents |
Entrance of bacteria into eukaryotic cells
The main aim of our project is to make, using synthetic biology methods, an Escherichia coli capable of entering mammalian cells. We've decided to use proteins that are already used in the same manner by enteropathogenic species like Listeria sp. or Yersinia sp. (see safety page for details about safety issues)
Theoretical basis
Many bacterial species are capable of entering mammalian cells. One of the crucial proteins for this process is invasin. It is capable of selective interaction with integrins, which are present on external eukaryotic membrane. This triggers signalling cascade, indispensable to start endocytosis [9]. This interaction occurs via the invasin N-terminal domain, which is capable of promoting effective endocytosis of bacteria that synthesize invasin by cells normally unable to undertake phagocytosis [3]. The other protein which is involved in process of entering mammalian cells is internalin, which can be found on the surface of Listeria monocytogenes. Internalin which selecively interacts with cadherins found on the external eukaryotic membranes.
Details
Invasion process
Invasion process has to be driven by 2 proteins: invasin from Yersinia sp. or Internalin A from Listeria monocytogenes. In the presence of listeriolysin from Listeria monocytogenes both proteins should effectively bind to the surface of mammalian cells and trigger internalisation of the whole bacteria. We would like to test both invasin and Internalin A and define which one is most effective.
Listeriolysin will be fused with secretion system because, in the opposite to invasin and internalin A, it doesn't contain its own secretion signal. Listeriolysin is also important for the next step of invasion - escape from the endosome.
Regulation
Our invasion module is controlled by a bistable switch, composed by 2 promoters and 2 repressors. Invasion proteins are under the control of the bacteriophage promoter (pR) which is repressed by the presence of the cI protein. The second part of the operon is the cIts gene that encodes a thermosensitive version of the cI repressor protein from the λ phage. It is under the control of the lactose promoter (Plac). This λ system of promoters and regulatory genes creates two mutually exclusive negative feedback loops and is dubbed the bistable switch [19]. It has two states depending on the conditions within the cell. cI protein binds to the pR promoter inhibiting its activity and expression of the invasion operon genes. cIts protein is inactivated by high temperature 42°C which allows for the expression of the invasion operon that codes for lacI. The lacI stops the gene expression from the Plac promoter ( the expression of cI protein). IPTG that binds and inactivates lacI can be used to switch on the expression from the Plac promoter. Another way to control the operon is in trans expression of cI or lacI genes. Activated invasion operon causes internalisation of bacteria by mammalian cells. Expression of the green fluorescence protein allows for the observation of the process using fluorescence microscopy.
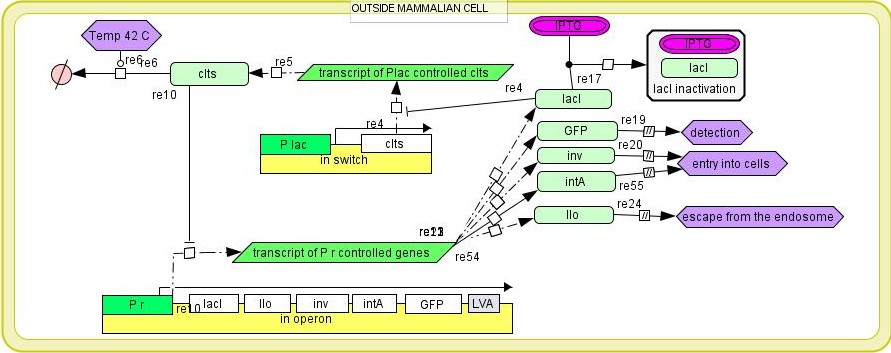
Fig 2. The overview of the invasion operon. It is based on a lacI/cI bistable switch. After thermal activation (42°C) the expression of the genes that enable the invasion of mammalian cells is activated. The invasion operon is composed of lacI – lactose operon repressor, llo – hemolysin from Listeria monocytogenes, inv – invasin from Yersinia sp., GFP – Green Fluorescent Protein from Aqueora victoria.
 "
"