Team:Cambridge/Project/Violacein
From 2009.igem.org
(→Violacein Pigment) |
(→Violacein Pigment) |
||
| Line 6: | Line 6: | ||
{{Template:Cambridgetemplatetop}} | {{Template:Cambridgetemplatetop}} | ||
[[#Background | Background]] | [[#Background | Background]] | ||
| - | [[# | + | [[#Design | Design]] |
| - | [[# | + | [[#Characterisation | Characterisation ]] |
| - | + | ||
[[# | ]] | [[# | ]] | ||
{{Template:Cambridgetemplatebottom}} | {{Template:Cambridgetemplatebottom}} | ||
| Line 32: | Line 31: | ||
*vio gene cluster complete cds AB032799 and AF172851. | *vio gene cluster complete cds AB032799 and AF172851. | ||
| - | == Action plan of our team == | + | === Action plan of our team === |
Our action plan is as follows: | Our action plan is as follows: | ||
| Line 42: | Line 41: | ||
3. Attach to the promoters of the processing system | 3. Attach to the promoters of the processing system | ||
| - | == | + | == Design == |
| + | |||
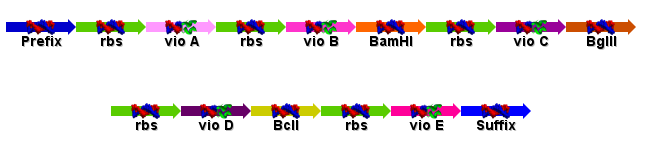
| + | The Vio operon had numerous forbidden restriction sites. We thus had to synthesize it, removing these restriction sites and optimizing codon usage for E. coli, to create the following biobrick: | ||
| + | |||
| + | [[Image:violaceinoperon.jpg]] | ||
| + | |||
| + | As DNA2.0 very generously agreed to synthesise the entire operon for us, we designed it to include all the five genes, each preceded by a ribosome binding site, and flanked by the prefix and suffix. All forbidden restriction sites were removed from the operon. The final plan for the inserted operon is shown below: | ||
| + | |||
| + | [[Image:Design sent to DNA 2.0.PNG]] | ||
| + | |||
| + | This will be held under a repressible promoter on the pJexpress cloning cassette from DNA2.0. The restriction sites will allow us to easily remove the vioC and vioD which are the genes responsible for chaging pigment colour. | ||
| + | |||
| + | |||
| + | == Characterisation == | ||
=== Proof of pigment production=== | === Proof of pigment production=== | ||
| Line 59: | Line 71: | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
== Characterisation of Biobricks and colour output == | == Characterisation of Biobricks and colour output == | ||
Revision as of 14:01, 19 October 2009
Categories :
Project :
-
Overview
Sensitivity Tuner
--- Characterisation
--- Modelling
Colour Generators
--- Carotenoids (Orange/Red)
--- Melanin (Brown)
--- Violacein (Purple/Green)
The Future
Safety
Notebook :
Team Logistics :
Violacein Pigment
Background
Violacein Biosynthesis
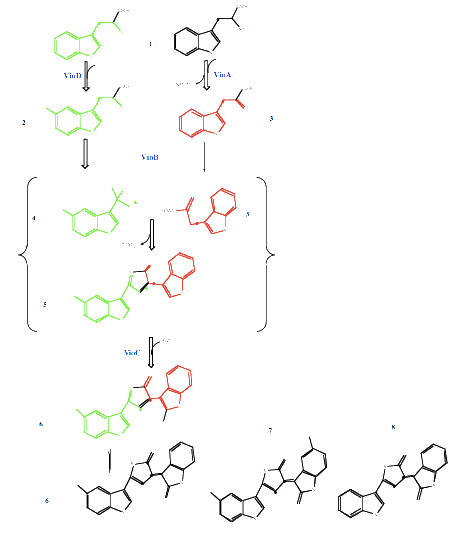
The Violacein pigment is produced from L-tryptophan via a pathway involving five enzymes, VioA-E. This forms a purple colour which remains within the individual cell colonies. This synthesis pathway is shown below:
From P.R. August, T.H. Grossman, C. Minor, M.P. Draper, I.A. MacNeil, J.M. Pemberton, K.M. Call, d. Holt, and M. S. Osbourne, Sequence Analysis and Functional Characterization of the Violacein Biosynthetic Pathway from Chromobacterium violaceum, J. Mol. Microbiol. Biotechnol. (2000) 2(4): 513-519. [http://www.horizonpress.com/jmmb/v2/v2n4/26.pdf]
The vioE is used in the step just after the vioB for the 1-2 shift of the indole ring. César Sánchez, Dr., Alfredo F. Braña, Prof. Dr., Carmen Méndez, Prof. Dr., José A. Salas, Prof. Dr. Reevaluation of the Violacein Biosynthetic Pathway and its Relationship to Indolocarbazole Biosynthesis http://www3.interscience.wiley.com/cgi-bin/fulltext/112732008/HTMLSTART
Further, as module 5 is Aqua, expressing the genes under different promoters will allow us to produce at least two different colours. The Sanchez paper suggests that removing vioD can produce a dark blue, while removing vio C produces a dark green.
Vio Operon
Our VioA-E genes are from Chromobacterium voilaceum ATCC 12472 in the pPSX vio+ plasmid. This was kindly provided by John Pemberton; Department of Microbiology and parasitology, University of Queensland, Brisbane, Australia. (Sarovich & Pemberton (2007) Plasmid 57:306-313)
- pPSX sequence ID FJ422118
- vio gene cluster complete cds AB032799 and AF172851.
Action plan of our team
Our action plan is as follows:
1. Test for violacein pigment production
2. Synthesize the violacein operon without any forbidden restriction sites. The finished gene will be BioBrick compatible and will have restriction sites around the vioC and vioD to remove these genes and form new colours.
3. Attach to the promoters of the processing system
Design
The Vio operon had numerous forbidden restriction sites. We thus had to synthesize it, removing these restriction sites and optimizing codon usage for E. coli, to create the following biobrick:
As DNA2.0 very generously agreed to synthesise the entire operon for us, we designed it to include all the five genes, each preceded by a ribosome binding site, and flanked by the prefix and suffix. All forbidden restriction sites were removed from the operon. The final plan for the inserted operon is shown below:
This will be held under a repressible promoter on the pJexpress cloning cassette from DNA2.0. The restriction sites will allow us to easily remove the vioC and vioD which are the genes responsible for chaging pigment colour.
Characterisation
Proof of pigment production
Successful Pigment Production
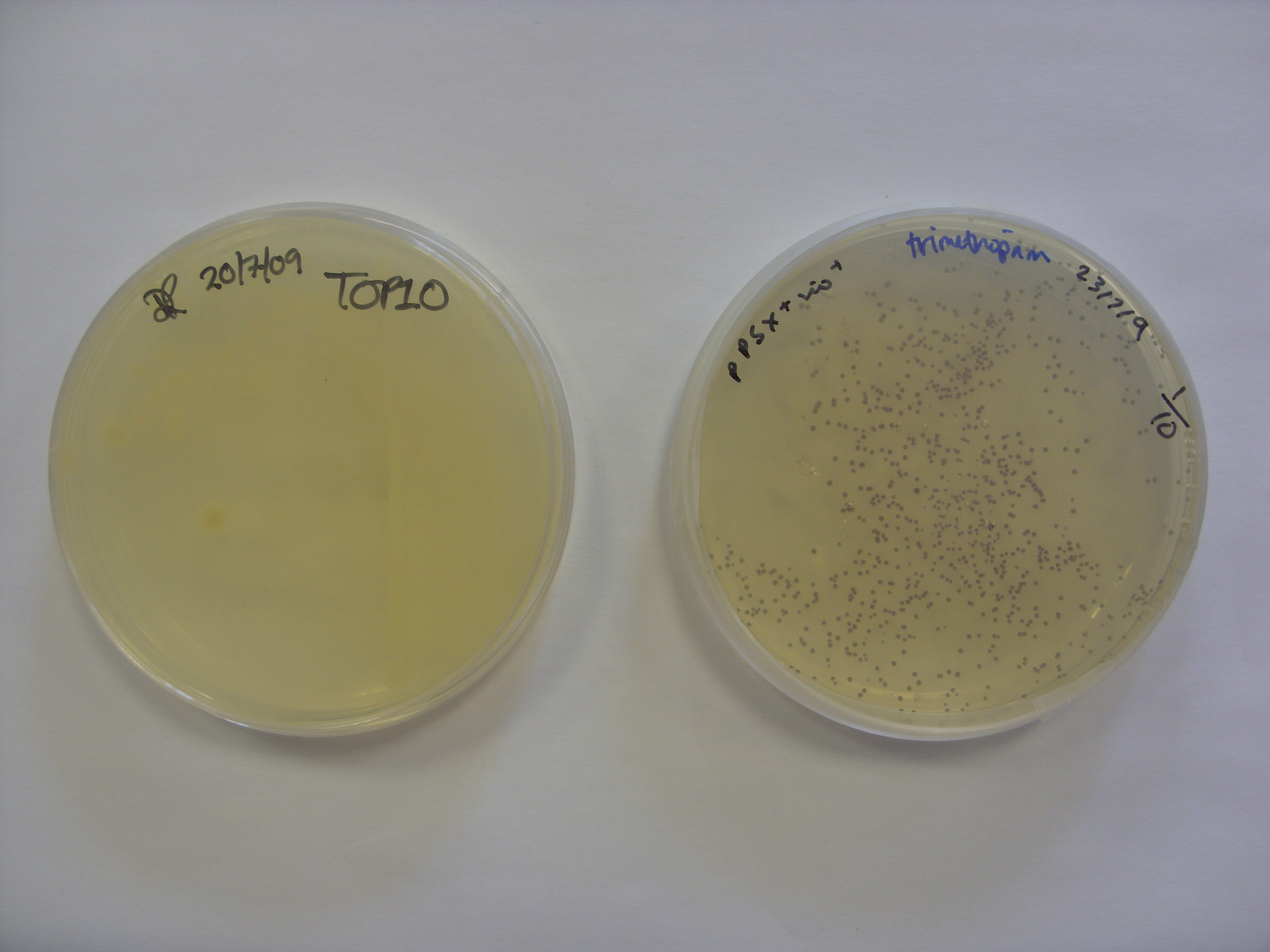
We transformed Top10 with pPSX-Vio+. After three colour eventually appeared, as shown below. Interestingly, the pigment appears to remain within the bacteria, with little or no bleeding into the media. We took the violacein pigment bacteria (right plate in photo) out of the fridge to find that the purple colour had started to develop. They were therefore left at room temperature overnight. The colour appears to be within the bacteria, with little or no bleeding into the media. The control plate (left plate) is the untransformed TOP10 E. coli.
Left: control plate - untransformed TOP10 E. coli, Right: Top10 transformed with pPSX-Vio+.
Pigment production efficienty
The Vio operon is currently on a very low copy number plasmid; moving it onto a higher copy number plasmid may accelerate pigment production.
Characterisation of Biobricks and colour output
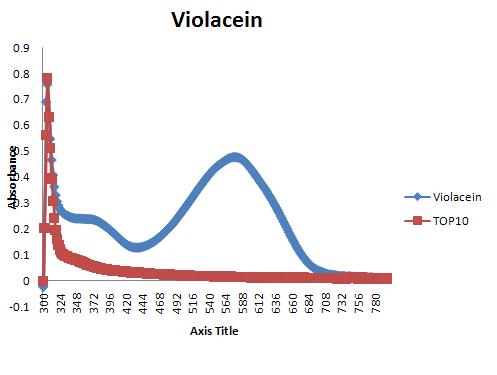
We characterised the violacein pigment by carrying out the acetone extraction protocol used for carotene. The results were normalised for OD and then plotted as a graph of absorption units against wavelength:
 "
"