Team:UNIPV-Pavia/Notebook/Week3Jul
From 2009.igem.org
(Difference between revisions)
(New page: {{UNIPV-Pavia/Month}} __NOTOC__ <div> = Week from July 6th, to July 12nd, 2009 = <html> <table width="100%"> <tr> <td align="left" width="50%"> <a href="https://2009.igem.org/Team:UNIPV-P...) |
(→Week from July 13rd, to July 19th, 2009) |
||
| (17 intermediate revisions not shown) | |||
| Line 3: | Line 3: | ||
<div> | <div> | ||
| - | = Week from July | + | <html><a name="week_start"></a></html> |
| + | = Week from July 13rd, to July 19th, 2009 = | ||
<html> | <html> | ||
<table width="100%"> | <table width="100%"> | ||
<tr> | <tr> | ||
<td align="left" width="50%"> | <td align="left" width="50%"> | ||
| - | <a href="https://2009.igem.org/Team:UNIPV-Pavia/Notebook/ | + | <a href="https://2009.igem.org/Team:UNIPV-Pavia/Notebook/Week2Jul#week_start"> |
<img src="https://2009.igem.org/Special:FilePath?file=Left_arrow.png"/> | <img src="https://2009.igem.org/Special:FilePath?file=Left_arrow.png"/> | ||
</a> | </a> | ||
| - | <a href="https://2009.igem.org/Team:UNIPV-Pavia/Notebook/ | + | <a href="https://2009.igem.org/Team:UNIPV-Pavia/Notebook/Week2Jul#week_start">Previous Week</a> |
</td> | </td> | ||
<td align="right" width="50%"> | <td align="right" width="50%"> | ||
| - | <a href="https://2009.igem.org/Team:UNIPV-Pavia/Notebook/ | + | <a href="https://2009.igem.org/Team:UNIPV-Pavia/Notebook/Week4Jul#week_start">Next Week</a> |
| - | <a href="https://2009.igem.org/Team:UNIPV-Pavia/Notebook/ | + | <a href="https://2009.igem.org/Team:UNIPV-Pavia/Notebook/Week4Jul#week_start"> |
<img src="https://2009.igem.org/Special:FilePath?file=Right_arrow.png"/> | <img src="https://2009.igem.org/Special:FilePath?file=Right_arrow.png"/> | ||
</a> | </a> | ||
| Line 23: | Line 24: | ||
</html> | </html> | ||
| - | == <html><font class="dayw_style">July, | + | == <html><font class="dayw_style">July, 13th</font></html> == |
| - | * | + | *This week we planned to assemble GFP protein generator under the control of Plac in our IPTG/lactose sensor (i.e. A11) in order to test it. We also planned to build up another aTc sensor in which tetR is expressed constitutively, but in smaller amount than in A9 BioBrick. Finally, we planned to put lacZ protein generator under the control of the lactose sensor in order to build up one of the final parts for our project: this part will express beta-galactosidase when lactose is present in the medium and this expression will be glucose-independent (see R0011 page in the Registry for more info). |
| - | *We | + | *We infected 5 ml of LB + Amp with 8 ul of the following glycerol stocks: |
{|cellpadding="20" | {|cellpadding="20" | ||
| - | | | + | |F2620 |
| - | | | + | |A11-1 (X2) |
| + | |A3-1 | ||
| + | |- | ||
| + | |A4 (X2) | ||
| + | |E0240 (X2) | ||
| + | |J23118 | ||
|} | |} | ||
| - | * | + | *We incubated the inocula at 37°C, 220 rpm overnight. |
| - | + | <div align="right"> | |
| + | [[#top|Top]] | ||
| + | </div> | ||
| - | + | == <html><font class="dayw_style">July, 14th</font></html> == | |
| - | * | + | *Miniprep for: |
| + | {|cellpadding="20" | ||
| + | |F2620 (for seq) | ||
| + | |A11-1 (X2) | ||
| + | |A3-1 | ||
| + | |- | ||
| + | |A4 (X2) | ||
| + | |E0240 (X2) | ||
| + | |J23118 | ||
| + | |} | ||
| + | *Digestion for: | ||
| + | {|cellpadding="20" | ||
| + | |A11-1(E-S) | ||
| + | |A11-1(S-P) | ||
| + | |A3-1(E-X) | ||
| + | |- | ||
| + | |A4(X-P)(X2) | ||
| + | |E0240(X-P)(X2) | ||
| + | |J23118(S-P) | ||
| + | |} | ||
| + | *Gel run/cut/purification: DNA yield was good for all the extracted parts, except A11-1(E-S), which was quantified as 3 ng/ul after Nanodrop measurement. Anyway, we decided to try to ligate it. | ||
| + | *Ligations: | ||
| + | **A12 = J23118(S-P) + A4(X-P) in ~pSB1A2 | ||
| + | **A13 = A11(E-S) + A3(E-X) in pSB1AK3 | ||
| + | **A14 = A11(S-P) + E0240(X-P) in pSB1A2 | ||
| - | + | *We incubated the ligations at 16°C overnight. | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | *We incubated the | + | |
<div align="right"> | <div align="right"> | ||
| Line 57: | Line 83: | ||
</div> | </div> | ||
| - | == <html><font class="dayw_style">July, | + | == <html><font class="dayw_style">July, 15th</font></html> == |
| - | * | + | *We received ''pdc'' and ''adhB'' genes from Mr Gene! we will call them MRGENE1 and MRGENE2. |
| + | **MRGENE1: 5 ug of liophylized DNA, Kan resistance plasmid; | ||
| + | **MRGENE2: 5 ug of liophylized DNA, Amp resistance plasmid. | ||
| - | |||
| - | * | + | *We transformed the overnight ligations in TOP10 and plated the transformed bacteria. We incubated the plates at 37°C overnight. |
| - | + | <div align="right"> | |
| + | [[#top|Top]] | ||
| + | </div> | ||
| - | + | == <html><font class="dayw_style">July, 16th</font></html> == | |
| - | * | + | *Plates results: |
| - | ** | + | **A12 showed colonies! and they were not red (as we expected, because RFP protein generator had been excided from BBa_J61002 plasmid); |
| - | ** | + | **A13 showed no colonies:( We will repeat this ligation one of the next days... |
| + | **A14 showed colonies! some of them seemed to be green under the sun light; this was probably caused to GFP accumulation due to the leaky activity of Plac promoter. | ||
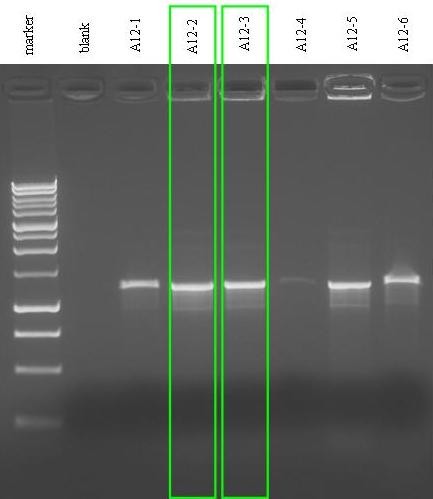
| + | *Colony PCR for A12. We didn't perform colony PCR screening for A14 because the insert was almost 1.5 Kb long and it contains two copies of B0015 BioBrick, which may corrupt PCR results. (Even A12 had B0015, but it was located at the end of the BioBrick!). During colony PCR, the colonies used for the reaction were inoculated in 1 ml of LB + Amp and incubated at 37°C, 220 rpm waiting for the end of the reaction. | ||
| - | '' | + | <table align="center"> |
| + | <tr> | ||
| + | <td> | ||
| + | <font class='didascalia'> | ||
| + | [[Image:pv_colonypcr_A12.jpg|thumb|500px|left|Colony PCR: all the screened colonies contained the correctly ligated plasmid.]] | ||
| + | </font> | ||
| + | </td> | ||
| + | </tr> | ||
| + | </table> | ||
| - | * | + | *Gel results: all the picked colonies had the insert with the expected length! we decided to keep and prepare a glycerol stock for: |
| + | **A12-2 (in case of failure) | ||
| + | **A12-3 (to use) | ||
| - | *We incubated the | + | *We re-filled (with 5 ml of LB + Amp) A12-2 and A12-3 cultures, using the remaining 250 ul. We incubated the cultures at 37°C, 220 rpm overnight. |
| + | *We picked 7 random colonies from A14 plate and infected 1 ml of LB + Amp. We incubated the inocula for 5 and 1/2 hours and then we prepared a glycerol stock for all of them. | ||
| - | + | *We re-filled the remaining 250 ul of bacterial culture with 5 ml of LB + Amp and incubated the new cultures at 37°C, 220 rpm overnight. Tomorrow they will be miniprepped and screened! | |
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| + | *We resuspended all the 5 ug of MRGENE1 and MRGENE2 with 20 ul of ddH2O. | ||
| - | + | *We transformed 1 ul of the resuspended DNA in TOP10. For each of the 2 plasmids, we plated 150 ul of transformed bacteria + SOC on an Amp plate and also on a Kan plate. In this way we could check if the plasmid had the expected antibiotic resistance. | |
| - | + | ||
| - | *We | + | |
| + | *We incubated the 4 plates at 37°C overnight. We stored the resuspended DNA at -20°C. | ||
<div align="right"> | <div align="right"> | ||
| Line 105: | Line 142: | ||
</div> | </div> | ||
| - | == <html><font class="dayw_style">July, | + | == <html><font class="dayw_style">July, 17th</font></html> == |
| - | * | + | *Plates results: |
| + | **MRGENE1 Amp plate - no colonies | ||
| + | **MRGENE1 Kan plate - bacterial carpet! | ||
| + | **MRGENE2 Amp plate - bacterial carpet! | ||
| + | **MRGENE2 Kan plate - no colonies | ||
| + | *The results were consistent with our expectations about the antibiotic resistances! | ||
| - | *We | + | *We picked a single colony from MRGENE1 and MRGENE2 plates and infected 3 ml of LB + suitable antibiotic. We incubated these inocula for about 6 hours. Then, we prepared three glycerol stocks for each of the two cultures in order to have good long term stocks. |
| - | * | + | *We aliquoted the remaining 750 ul (=3 ml-750x3) in three falcon tubes containing 250 ul of bacterial culture and then we re-filled the falcon tubes with 5 ml of LB + suitable antibiotic to grow 6 overnight cultures. |
| - | + | ||
| - | *We | + | *We also infected 5 ml of LB + Amp with 8 ul of B0030 glycerol stock to grow 3 identical 5 ml overnight cultures. |
| + | *All the 9 cultures were incubated at 37°C, 220 rpm overnight. | ||
| - | |||
| - | * | + | *Miniprep for the 7 cultures coming from the colonies of A14 plate. |
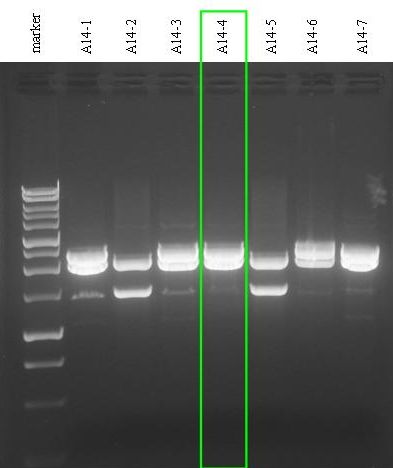
| + | *Digestion E-P of 1 ug of DNA for all the 7 miniprepped plasmids (20 ul reaction volume). | ||
| + | *Electrophoresis for the digestions. | ||
| + | <table align="center"> | ||
| + | <tr> | ||
| + | <td> | ||
| + | <font class='didascalia'> | ||
| + | [[Image:pv_digestion_A14.jpg|thumb|500px|left|Digestion screening: colonies 2 and 5 were negative, while the other colonies showed the correctly ligated plasmid, but with a smaller amount of unwanted plasmid.]] | ||
| + | </font> | ||
| + | </td> | ||
| + | </tr> | ||
| + | </table> | ||
| - | + | *Gel results: | |
| + | **A14-1 - positive insert(~2250 bp) + unwanted insert(~1400 bp) + unexpected band(~1100 bp) | ||
| + | **A14-2 - unwanted insert(~1400 bp) | ||
| + | **A14-3 - positive insert(~2250 bp) + unwanted insert(~1400 bp) + unexpected band(~1100 bp) | ||
| + | **A14-4 - positive insert(~2250 bp) + unwanted insert(~1400 bp) + unexpected band(~1100 bp) | ||
| + | **A14-5 - unwanted insert(~1400 bp) | ||
| + | **A14-6 - positive insert(~2250 bp) + unwanted insert(~1400 bp) | ||
| + | **A14-7 - positive insert(~2250 bp) + unwanted insert(~1400 bp) + unexpected band(~1100 bp) | ||
| - | * | + | *We decided to keep A14-4 glycerol stock in order to perform: i)inoculum, ii)miniprep, iii)dilution, iv) 5 pg transformation, v)plating, vi)digestion screening, as we performed for A8 and A9. |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | + | *Miniprep for A12-2 and A12-3: DNA yield was quite low...next week they will be inoculated and miniprepped again in order to prepare the samples for sequencing. | |
| - | + | ||
<div align="right"> | <div align="right"> | ||
| Line 153: | Line 198: | ||
</div> | </div> | ||
| - | == <html><font class="dayw_style">July, | + | == <html><font class="dayw_style">July, 18th</font></html> == |
| - | * | + | *Miniprep for: |
| - | + | {|cellpadding="20" | |
| - | + | |MRGENE1 | |
| - | + | |MRGENE1-2 | |
| - | + | |MRGENE1-3 | |
| - | + | |- | |
| - | + | |MRGENE2 | |
| - | {| | + | |MRGENE2-2 |
| - | | | + | |MRGENE2-3 |
| + | |- | ||
| + | |B0030(X3) | ||
|} | |} | ||
| - | + | *We stored the miniprepped DNA at -20°C. Next week we will be ready to assemble these bricks;) | |
| - | + | ||
| - | * | + | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
<div align="right"> | <div align="right"> | ||
[[#top|Top]] | [[#top|Top]] | ||
</div> | </div> | ||
| - | |||
| - | |||
| - | |||
<html> | <html> | ||
<hr/> | <hr/> | ||
| Line 187: | Line 222: | ||
<tr> | <tr> | ||
<td align="left" width="50%"> | <td align="left" width="50%"> | ||
| - | <a href="https://2009.igem.org/Team:UNIPV-Pavia/Notebook/Week2Jul"> | + | <a href="https://2009.igem.org/Team:UNIPV-Pavia/Notebook/Week2Jul#week_start"> |
<img src="https://2009.igem.org/Special:FilePath?file=Left_arrow.png"/> | <img src="https://2009.igem.org/Special:FilePath?file=Left_arrow.png"/> | ||
</a> | </a> | ||
| - | <a href="https://2009.igem.org/Team:UNIPV-Pavia/Notebook/Week2Jul">Previous Week</a> | + | <a href="https://2009.igem.org/Team:UNIPV-Pavia/Notebook/Week2Jul#week_start">Previous Week</a> |
</td> | </td> | ||
<td align="right" width="50%"> | <td align="right" width="50%"> | ||
| - | <a href="https://2009.igem.org/Team:UNIPV-Pavia/Notebook/Week4Jul">Next Week</a> | + | <a href="https://2009.igem.org/Team:UNIPV-Pavia/Notebook/Week4Jul#week_start">Next Week</a> |
| - | <a href="https://2009.igem.org/Team:UNIPV-Pavia/Notebook/Week4Jul"> | + | <a href="https://2009.igem.org/Team:UNIPV-Pavia/Notebook/Week4Jul#week_start"> |
<img src="https://2009.igem.org/Special:FilePath?file=Right_arrow.png"/> | <img src="https://2009.igem.org/Special:FilePath?file=Right_arrow.png"/> | ||
</a> | </a> | ||
Latest revision as of 09:05, 20 October 2009

|
|
|
|
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
Week from July 13rd, to July 19th, 2009
 Previous Week
Previous Week
|
Next Week

|
July, 13th
- This week we planned to assemble GFP protein generator under the control of Plac in our IPTG/lactose sensor (i.e. A11) in order to test it. We also planned to build up another aTc sensor in which tetR is expressed constitutively, but in smaller amount than in A9 BioBrick. Finally, we planned to put lacZ protein generator under the control of the lactose sensor in order to build up one of the final parts for our project: this part will express beta-galactosidase when lactose is present in the medium and this expression will be glucose-independent (see R0011 page in the Registry for more info).
- We infected 5 ml of LB + Amp with 8 ul of the following glycerol stocks:
| F2620 | A11-1 (X2) | A3-1 |
| A4 (X2) | E0240 (X2) | J23118 |
- We incubated the inocula at 37°C, 220 rpm overnight.
July, 14th
- Miniprep for:
| F2620 (for seq) | A11-1 (X2) | A3-1 |
| A4 (X2) | E0240 (X2) | J23118 |
- Digestion for:
| A11-1(E-S) | A11-1(S-P) | A3-1(E-X) |
| A4(X-P)(X2) | E0240(X-P)(X2) | J23118(S-P) |
- Gel run/cut/purification: DNA yield was good for all the extracted parts, except A11-1(E-S), which was quantified as 3 ng/ul after Nanodrop measurement. Anyway, we decided to try to ligate it.
- Ligations:
- A12 = J23118(S-P) + A4(X-P) in ~pSB1A2
- A13 = A11(E-S) + A3(E-X) in pSB1AK3
- A14 = A11(S-P) + E0240(X-P) in pSB1A2
- We incubated the ligations at 16°C overnight.
July, 15th
- We received pdc and adhB genes from Mr Gene! we will call them MRGENE1 and MRGENE2.
- MRGENE1: 5 ug of liophylized DNA, Kan resistance plasmid;
- MRGENE2: 5 ug of liophylized DNA, Amp resistance plasmid.
- We transformed the overnight ligations in TOP10 and plated the transformed bacteria. We incubated the plates at 37°C overnight.
July, 16th
- Plates results:
- A12 showed colonies! and they were not red (as we expected, because RFP protein generator had been excided from BBa_J61002 plasmid);
- A13 showed no colonies:( We will repeat this ligation one of the next days...
- A14 showed colonies! some of them seemed to be green under the sun light; this was probably caused to GFP accumulation due to the leaky activity of Plac promoter.
- Colony PCR for A12. We didn't perform colony PCR screening for A14 because the insert was almost 1.5 Kb long and it contains two copies of B0015 BioBrick, which may corrupt PCR results. (Even A12 had B0015, but it was located at the end of the BioBrick!). During colony PCR, the colonies used for the reaction were inoculated in 1 ml of LB + Amp and incubated at 37°C, 220 rpm waiting for the end of the reaction.
|
|
- Gel results: all the picked colonies had the insert with the expected length! we decided to keep and prepare a glycerol stock for:
- A12-2 (in case of failure)
- A12-3 (to use)
- We re-filled (with 5 ml of LB + Amp) A12-2 and A12-3 cultures, using the remaining 250 ul. We incubated the cultures at 37°C, 220 rpm overnight.
- We picked 7 random colonies from A14 plate and infected 1 ml of LB + Amp. We incubated the inocula for 5 and 1/2 hours and then we prepared a glycerol stock for all of them.
- We re-filled the remaining 250 ul of bacterial culture with 5 ml of LB + Amp and incubated the new cultures at 37°C, 220 rpm overnight. Tomorrow they will be miniprepped and screened!
- We resuspended all the 5 ug of MRGENE1 and MRGENE2 with 20 ul of ddH2O.
- We transformed 1 ul of the resuspended DNA in TOP10. For each of the 2 plasmids, we plated 150 ul of transformed bacteria + SOC on an Amp plate and also on a Kan plate. In this way we could check if the plasmid had the expected antibiotic resistance.
- We incubated the 4 plates at 37°C overnight. We stored the resuspended DNA at -20°C.
July, 17th
- Plates results:
- MRGENE1 Amp plate - no colonies
- MRGENE1 Kan plate - bacterial carpet!
- MRGENE2 Amp plate - bacterial carpet!
- MRGENE2 Kan plate - no colonies
- The results were consistent with our expectations about the antibiotic resistances!
- We picked a single colony from MRGENE1 and MRGENE2 plates and infected 3 ml of LB + suitable antibiotic. We incubated these inocula for about 6 hours. Then, we prepared three glycerol stocks for each of the two cultures in order to have good long term stocks.
- We aliquoted the remaining 750 ul (=3 ml-750x3) in three falcon tubes containing 250 ul of bacterial culture and then we re-filled the falcon tubes with 5 ml of LB + suitable antibiotic to grow 6 overnight cultures.
- We also infected 5 ml of LB + Amp with 8 ul of B0030 glycerol stock to grow 3 identical 5 ml overnight cultures.
- All the 9 cultures were incubated at 37°C, 220 rpm overnight.
- Miniprep for the 7 cultures coming from the colonies of A14 plate.
- Digestion E-P of 1 ug of DNA for all the 7 miniprepped plasmids (20 ul reaction volume).
- Electrophoresis for the digestions.
|
|
- Gel results:
- A14-1 - positive insert(~2250 bp) + unwanted insert(~1400 bp) + unexpected band(~1100 bp)
- A14-2 - unwanted insert(~1400 bp)
- A14-3 - positive insert(~2250 bp) + unwanted insert(~1400 bp) + unexpected band(~1100 bp)
- A14-4 - positive insert(~2250 bp) + unwanted insert(~1400 bp) + unexpected band(~1100 bp)
- A14-5 - unwanted insert(~1400 bp)
- A14-6 - positive insert(~2250 bp) + unwanted insert(~1400 bp)
- A14-7 - positive insert(~2250 bp) + unwanted insert(~1400 bp) + unexpected band(~1100 bp)
- We decided to keep A14-4 glycerol stock in order to perform: i)inoculum, ii)miniprep, iii)dilution, iv) 5 pg transformation, v)plating, vi)digestion screening, as we performed for A8 and A9.
- Miniprep for A12-2 and A12-3: DNA yield was quite low...next week they will be inoculated and miniprepped again in order to prepare the samples for sequencing.
July, 18th
- Miniprep for:
| MRGENE1 | MRGENE1-2 | MRGENE1-3 |
| MRGENE2 | MRGENE2-2 | MRGENE2-3 |
| B0030(X3) |
- We stored the miniprepped DNA at -20°C. Next week we will be ready to assemble these bricks;)
 Previous Week
Previous Week
|
Next Week

|
 "
"