Team:Groningen/Project/Transport
From 2009.igem.org
(→Heavy metal uptake coupled to citrate via efCitH bsCitM) |
(→Results) |
||
| (32 intermediate revisions not shown) | |||
| Line 1: | Line 1: | ||
{{Team:Groningen/Project/Header|}} | {{Team:Groningen/Project/Header|}} | ||
| + | <div style="float:left" >{{linkedImage|GroningenPrevious.png|Team:Groningen/Application}}</div> | ||
<div title="Arsie Says UP TO ACCUMULATION" style="float:right" >{{linkedImage|Next.JPG|Team:Groningen/Project/Accumulation}}</div> | <div title="Arsie Says UP TO ACCUMULATION" style="float:right" >{{linkedImage|Next.JPG|Team:Groningen/Project/Accumulation}}</div> | ||
| - | {| | + | {| style="clear:both" |
|<html><style type="text/css"> | |<html><style type="text/css"> | ||
.intro { margin-left:0px; margin-top:10px; padding:10px; border-left:solid 5px #FFF6D5; border-right:solid 5px #FFF6D5; text-align:justify;background:#FFFFE5; } | .intro { margin-left:0px; margin-top:10px; padding:10px; border-left:solid 5px #FFF6D5; border-right:solid 5px #FFF6D5; text-align:justify;background:#FFFFE5; } | ||
</style></html> | </style></html> | ||
<div class="intro"> | <div class="intro"> | ||
| - | < | + | <h1>Transport</h1> |
| - | + | <b>To isolate heavy metals from the environment we require uptake systems. Several different mechanisms to create such an import system are exist; metal transporters (coupled and uncoupled) and binding proteins in the periplasm. Import systems for several metals were found. We investigated HmtA for copper/zinc uptake. Cloning of the HmtA failed unfortunatly. Citrate coupled transporters, CitH and CitM were also considered as wel as the periplasmic accumulation operon Mer. | |
| - | + | Since we chose to focus on arsenic the final device was made with GlpF. GlpF a aquaglycerol porin was found to import not only glycerol but also arsenite and arsenate. This importer was cloned as a BioBrick part and transformed into ''E. coli''. An uptake assay was performed and a metal sensitivity assay, which showed functionality of the GlpF transporter.</b> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
<br><br><br> | <br><br><br> | ||
</div> | </div> | ||
| Line 29: | Line 18: | ||
==Arsenite uptake via GlpF== | ==Arsenite uptake via GlpF== | ||
| - | <!--[[Image:GlpF.jpeg|200px|thumb|right|73As(III) and 125Sb(III) uptake into cells of E. coli is facilitated by the aquaglyceroporin channel GlpF.]]--> | + | <!--[[Image:GlpF.jpeg|200px|thumb|right|73As(III) and 125Sb(III) uptake into cells of ''E. coli'' is facilitated by the aquaglyceroporin channel GlpF.]]--> |
===GlpF=== | ===GlpF=== | ||
| Line 37: | Line 26: | ||
The GlpF aquaglycerol porin is a membrane protein with a symmetric arrangement of four independent GlpF channels. One monomer of this tetramer GlpF porin consists of six transmembrane and two half membrane-spanning α-helices that form a right-handed helical bundle around the channel. The channel has a diameter of ~15Å at the periplasmid end, which constricts towards a diameter of ~3.8Å at the beginning of a 28 Å long selective channel that ends at the cytoplasmic end [[Team:Groningen/Literature#Fu, DX, et al.2000|(Fu, DX, et al.2000)]]. | The GlpF aquaglycerol porin is a membrane protein with a symmetric arrangement of four independent GlpF channels. One monomer of this tetramer GlpF porin consists of six transmembrane and two half membrane-spanning α-helices that form a right-handed helical bundle around the channel. The channel has a diameter of ~15Å at the periplasmid end, which constricts towards a diameter of ~3.8Å at the beginning of a 28 Å long selective channel that ends at the cytoplasmic end [[Team:Groningen/Literature#Fu, DX, et al.2000|(Fu, DX, et al.2000)]]. | ||
The GlpF is a stereospecific channel that is thought to be more selective on molecular size than on chemical structure [[Team:Groningen/Literature#Fu, DX, et al.2000|(Fu, DX, et al.2000]], [[Team:Groningen/Literature#Heller, KB, et al.1980|(Heller, KB, et al.1980)]] . It does allow transport of a variance of non-charged compounds ranging from polyhydric alcohols, glycerol being one of them, arsenic to antimone [[Team:Groningen/Literature#Fu, DX, et al.2000|(Fu, DX, et al.2000]]), [[Team:Groningen/Literature#Meng, YL, et al.2004|(Meng, YL, et al.2004]]), [[Team:Groningen/Literature#Porquet, A, et al.2007|(Porquet, A, et al.2007)]], [[Team:Groningen/Literature#Rosen, BR, et al.2009|(Rosen, BR, et al.2009]]), [[Team:Groningen/Literature#Heller, KB, et al.1980|(Heller, KB, et al.1980)]]. Carbon sugars and ions are shown to be unable to be transported by GlpF [[Team:Groningen/Literature#Heller, KB, et al.1980|(Heller, KB, et al.1980)]]. At physiological pH arsenic and antimone are not present in their ionic state but rather as As(OH)3 and Sb(OH)3 [[Team:Groningen/Literature#Rosen, BR, et al.2009|(Rosen, BR, et al.2009)]]. These elements show a charge distribution similar to glycerol and a smaller but comparable volume. The structural similarities are thought to be the reason for the possibility of these elements to enter the cell by GlpF [[Team:Groningen/Literature#Porquet, A, et al.2007|(Porquet, A, et al.2007)]], GlpF facilitates transport of these compounds down there gradient (inside or outside the cell). | The GlpF is a stereospecific channel that is thought to be more selective on molecular size than on chemical structure [[Team:Groningen/Literature#Fu, DX, et al.2000|(Fu, DX, et al.2000]], [[Team:Groningen/Literature#Heller, KB, et al.1980|(Heller, KB, et al.1980)]] . It does allow transport of a variance of non-charged compounds ranging from polyhydric alcohols, glycerol being one of them, arsenic to antimone [[Team:Groningen/Literature#Fu, DX, et al.2000|(Fu, DX, et al.2000]]), [[Team:Groningen/Literature#Meng, YL, et al.2004|(Meng, YL, et al.2004]]), [[Team:Groningen/Literature#Porquet, A, et al.2007|(Porquet, A, et al.2007)]], [[Team:Groningen/Literature#Rosen, BR, et al.2009|(Rosen, BR, et al.2009]]), [[Team:Groningen/Literature#Heller, KB, et al.1980|(Heller, KB, et al.1980)]]. Carbon sugars and ions are shown to be unable to be transported by GlpF [[Team:Groningen/Literature#Heller, KB, et al.1980|(Heller, KB, et al.1980)]]. At physiological pH arsenic and antimone are not present in their ionic state but rather as As(OH)3 and Sb(OH)3 [[Team:Groningen/Literature#Rosen, BR, et al.2009|(Rosen, BR, et al.2009)]]. These elements show a charge distribution similar to glycerol and a smaller but comparable volume. The structural similarities are thought to be the reason for the possibility of these elements to enter the cell by GlpF [[Team:Groningen/Literature#Porquet, A, et al.2007|(Porquet, A, et al.2007)]], GlpF facilitates transport of these compounds down there gradient (inside or outside the cell). | ||
| - | If GlpF behaves as a nonsaturable transporter, a transport rate of 1umol of glycerol is transported per minute per mgr of cell protein [[Team:Groningen/Literature#Heller, KB, et al.1980|(Heller, KB, et al.1980)]]. | + | If GlpF behaves as a nonsaturable transporter, a transport rate of 1umol of glycerol is transported per minute per mgr of cell protein [[Team:Groningen/Literature#Heller, KB, et al.1980|(Heller, KB, et al.1980)]]. |
====Cloning strategy==== | ====Cloning strategy==== | ||
| Line 45: | Line 34: | ||
====Results==== | ====Results==== | ||
| - | The ability of GlpF (overexpressed under | + | The ability of GlpF (overexpressed under IPTG induction) to transport As(III) was tested by an arsenite uptake [https://2009.igem.org/Team:Groningen/Protocols assay]. Also the full accumulation device (<partinfo>BBa_K190038</partinfo>) was tested using this assay. '''Data and analysis can be found [https://2009.igem.org/Team:Groningen/Project/Accumulation here]. |
''' | ''' | ||
| - | |||
| - | ===Modelling uptake GlpF=== | + | [[Image:DeathAssayWT.png|310px|left]] |
| + | [[Image:DeathAssayGlpF.png|310px|left]] | ||
| + | [[Image:DeathAssayLow.png|310px|left]] | ||
| + | |||
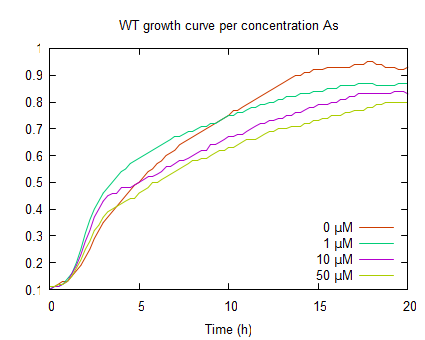
| + | The graphs above represent the result of the metal sensitivity [https://2009.igem.org/Team:Groningen/Protocols#Death_assay assay]. The lines in the graphs represent the average optical density (OD<sub>600</sub>) of a construct over time. The graph on the left shows that increased As(III) levels inhibits growth and as the As(III) concentration increases the plateau OD<sub>600</sub> level decreases. | ||
| + | |||
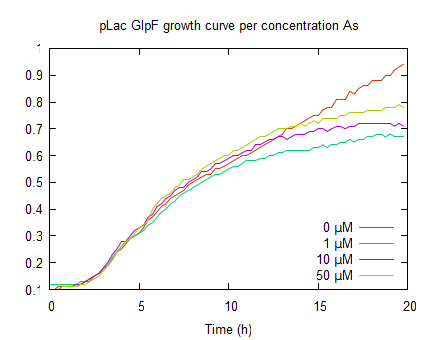
| + | The second graph represents the OD<sub>600</sub> values of a ''E. coli'' culture with pLac-GlpF. The growth curves are less steep in the log phase compared to WT, because of the protein expression by IPTG induction. In the absence of As(III) the plateau level equals the WT, but if arsenite is present the plateaus are lower (OD<sub>600</sub> <0.8) which implies functional expression of GlpF causing As(III) uptake to toxic levels. | ||
| + | |||
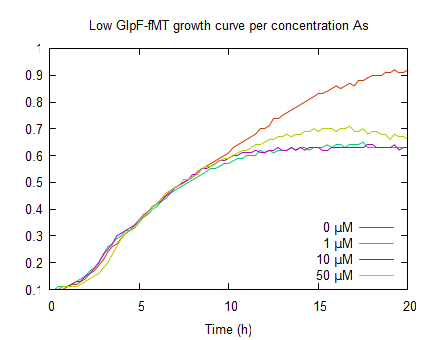
| + | In the graph on the right, shows growth curves of ''E. coli'' expressing GlpF and fMT under a low constitutive promoter. Also a similar growth in the log phase, compared to pLac GlpF, can be seen this is due to protein expression. Furthermore, like WT (0μM As(III)) it has its plateau above OD<sub>600</sub> 0.9. Upon incubation with various arsenite concentrations, the plateaus are lower (OD<sub>600</sub> <0.8) compared to WT. This is due to As(III) uptake by GlpF. The reduced growth is also an indicator for arsenite uptake. It is difficult to see if fMT has any effect because this assay can not show where the arsenite is and how fMT interferes with the cells' detoxification. | ||
| + | |||
| + | ==={{anchor|Modelling}}Modelling uptake GlpF=== | ||
<html> | <html> | ||
<script type="text/javascript" src="/Team:Groningen/Modelling/Model.js?action=raw"></script> | <script type="text/javascript" src="/Team:Groningen/Modelling/Model.js?action=raw"></script> | ||
| Line 114: | Line 112: | ||
<br> | <br> | ||
| - | === | + | ===Export of arsenicum via Ars operon=== |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
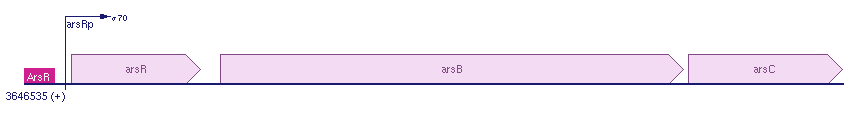
| - | + | GlpF is the importer of arsenicum. After arsenicum enters the cell, in response the Ars operon produces ArsR. At the same time, ArsB is also produced by Ars operon. This happens because the Ars operon contains three open reading frames: the first is ArsR, second ArsB and the last one is ArsC. ArsB is the exporter of arsenicum. The ars operon is located on the chromosomal DNA of E. coli. | |
| + | For more information see: [http://biocyc.org/ECOLI/NEW-IMAGE?type=GENE-IN-CHROM-BROWSER&object=EG12235 biocyc]. | ||
| + | |||
| + | [[Image:ArsRBC_operon.PNG|600px]] | ||
===Additional sources=== | ===Additional sources=== | ||
| Line 139: | Line 131: | ||
===HmtA=== | ===HmtA=== | ||
====Introduction==== | ====Introduction==== | ||
| - | HmtA(heavy metal transporter A) from <i>Pseudomonas aeruginosa</i> [http://www.ncbi.nlm.nih.gov/protein/81857196 Q9I147] is a P-type ATPase importer. This membrane protein mediates the uptake of copper (Cu) and zinc (Zn) and was functionally expressed in E.coli ([http://www.ncbi.nlm.nih.gov/pubmed/19264958 Lewinson 2009]). We want to use this membrane protein to accumulate copper and zinc into the cells. we believe this ATP-driven pump is capable of generating an elevated intracellular concentration of these compounds enabling the harvesting of copper and zinc from the medium. | + | HmtA(heavy metal transporter A) from <i>Pseudomonas aeruginosa</i> [http://www.ncbi.nlm.nih.gov/protein/81857196 Q9I147] is a P-type ATPase importer. This membrane protein mediates the uptake of copper (Cu) and zinc (Zn) and was functionally expressed in ''E. coli'' ([http://www.ncbi.nlm.nih.gov/pubmed/19264958 Lewinson 2009]). We want to use this membrane protein to accumulate copper and zinc into the cells. we believe this ATP-driven pump is capable of generating an elevated intracellular concentration of these compounds enabling the harvesting of copper and zinc from the medium. |
====Cloning strategy==== | ====Cloning strategy==== | ||
| - | |||
There are several restriction sites to be modified from [https://static.igem.org/mediawiki/2009/8/85/PBAD-HmtA-ClonemanagerFile.zip Lewinson's] pBAD construct. A vector with amp resistance with L-arabinose inducible HmtA-6HIS. The restriction sites have been silently mutated maintaining the amino acid sequence. | There are several restriction sites to be modified from [https://static.igem.org/mediawiki/2009/8/85/PBAD-HmtA-ClonemanagerFile.zip Lewinson's] pBAD construct. A vector with amp resistance with L-arabinose inducible HmtA-6HIS. The restriction sites have been silently mutated maintaining the amino acid sequence. | ||
We will create these mutations via PCR than digest the old methylated template and clone the product into competent cells. | We will create these mutations via PCR than digest the old methylated template and clone the product into competent cells. | ||
| Line 148: | Line 139: | ||
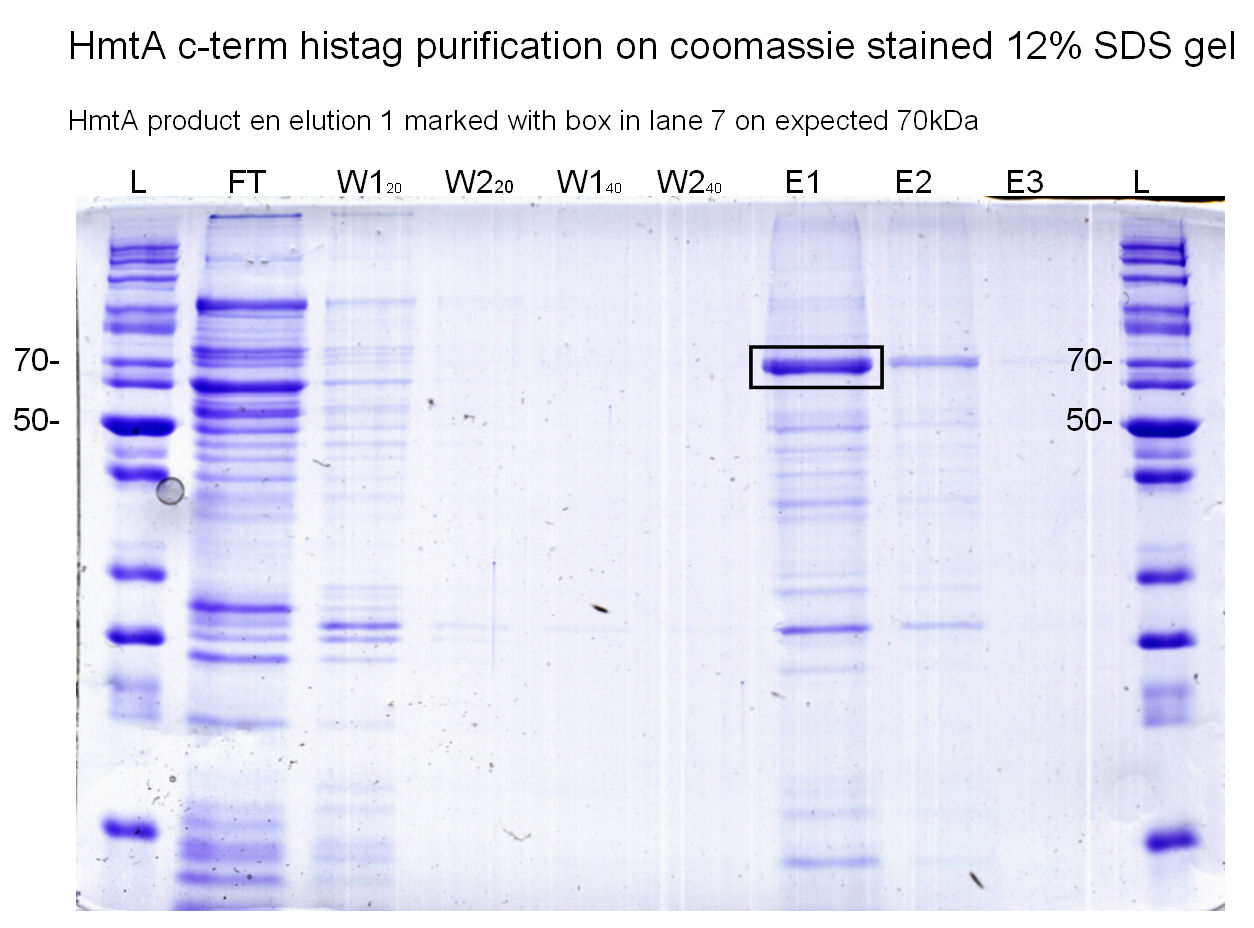
====Results==== | ====Results==== | ||
[[Image:HmtA_SDS_gel.jpg|200px|thumb|right|[Team:Groningen/Team|HmtA-6HIS on SDS-page]] | [[Image:HmtA_SDS_gel.jpg|200px|thumb|right|[Team:Groningen/Team|HmtA-6HIS on SDS-page]] | ||
| - | So far we have cloned HmtA as a biobrick without EcoRI site in the coding region into the iGEM vector. Unfortunately a mutation occurred at base 103 from the start of the orf. By a point mutation c to t in the first nucleotide of the codon changed arginine 35 to a Cysteine. We do not know the effects but we suspect it might have a great influence due to the very reactive side chain of Cysteine, eventhough it is not in the channel itself based on [http://www.cbs.dtu.dk/services/TMHMM/ TMHMM] predictions which indicate trans membrane helices of a protein. Further cloning is expected to be unsuccessful because the | + | So far we have cloned HmtA as a biobrick without EcoRI site in the coding region into the iGEM vector. Unfortunately a mutation occurred at base 103 from the start of the orf. By a point mutation c to t in the first nucleotide of the codon changed arginine 35 to a Cysteine. We do not know the effects but we suspect it might have a great influence due to the very reactive side chain of Cysteine, eventhough it is not in the channel itself based on [http://www.cbs.dtu.dk/services/TMHMM/ TMHMM] predictions which indicate trans membrane helices of a protein. Further cloning is expected to be unsuccessful because the IPTG induced clones grow even slightly better than the empty vector control. This is most likely cause by the missing pLAC-RBS in front of the gene. There was no positive controle with the L-arabinose inducable HmtA-6His in pBAD. We did do expression experiments with the pBAD construct to purified the membrane protein as quality controle. result shown in the figure on the right. |
==Heavy metal uptake coupled to citrate via ''ef''CitH ''bs''CitM== | ==Heavy metal uptake coupled to citrate via ''ef''CitH ''bs''CitM== | ||
| - | Force feeding of the heavy metals into the cell is possible when citrate is the only carbon source | + | Force feeding of the heavy metals into the cell is possible when citrate is the only available carbon source. Citrate in complex with heavy metals can be translocated over the membrane into the cell via citrate transporters. |
This can be a very efficient strategy to accumulate vast ammounts of heavy metals. | This can be a very efficient strategy to accumulate vast ammounts of heavy metals. | ||
| - | The | + | The two membrane proteins are CitM from ''Bacillus subtilis'' studied by [http://www.ncbi.nlm.nih.gov/pubmed/11053381 B.P Krom]. <i>Bs</i>CitM can transport citrate in complex with Mg<sup>2+</sup>, Ni<sup>2+</sup>, Mn<sup>2+</sup>, Co<sup>2+</sup>, and Zn<sup>2+</sup>. |
| - | The other | + | The other is CitH from ''Enterococcus faecalis'' described by [http://www.ncbi.nlm.nih.gov/pubmed/17042778 V.S Blancato]. <i>Ef</i>CitH catalyzes translocation of the citrate in complex with Ca<sup>2+</sup>, Sr<sup>2+</sup> Mn<sup>2+</sup> Mn<sup>2+</sup> Cd<sup>2+</sup> and Pb<sup>2+</sup>. |
| - | ===Additional sources=== | + | ====Additional sources==== |
More information on this topic can be found in: | More information on this topic can be found in: | ||
| Line 172: | Line 163: | ||
As the cells die after uptake of Mg (and induction of the Mer transporter), this system is not very well usable for our project. The dead cells will not produce the gas vesicles (it may be used however by having the gas vesicles consitutively expressed), thereby bouyancy may be a problem ([[Team:Groningen/Literature#Pennella2005|Pennella 2005]], [[Team:Groningen/Literature#Kao2008|Kao 2008]]). | As the cells die after uptake of Mg (and induction of the Mer transporter), this system is not very well usable for our project. The dead cells will not produce the gas vesicles (it may be used however by having the gas vesicles consitutively expressed), thereby bouyancy may be a problem ([[Team:Groningen/Literature#Pennella2005|Pennella 2005]], [[Team:Groningen/Literature#Kao2008|Kao 2008]]). | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
Latest revision as of 23:16, 21 October 2009
[http://2009.igem.org/Team:Groningen http://2009.igem.org/wiki/images/f/f1/Igemhomelogo.png]
|
|---|
TransportTo isolate heavy metals from the environment we require uptake systems. Several different mechanisms to create such an import system are exist; metal transporters (coupled and uncoupled) and binding proteins in the periplasm. Import systems for several metals were found. We investigated HmtA for copper/zinc uptake. Cloning of the HmtA failed unfortunatly. Citrate coupled transporters, CitH and CitM were also considered as wel as the periplasmic accumulation operon Mer.
Since we chose to focus on arsenic the final device was made with GlpF. GlpF a aquaglycerol porin was found to import not only glycerol but also arsenite and arsenate. This importer was cloned as a BioBrick part and transformed into E. coli. An uptake assay was performed and a metal sensitivity assay, which showed functionality of the GlpF transporter.
|
Arsenite uptake via GlpF
GlpF
Introduction
GlpF is an aquaglycerol porin of E.coli which facilitates not only glycerol import, but also arsenic (As) and antimone (Sb) import (Fu, DX, et al.2000), (Meng, YL, et al.2004), (Porquet, A, et al.2007), (Rosen, BR, et al.2009) . It has homologues in other organisms; Fps1p has shown to facilitate arsenic import in yeast and AQP9 is the mammalian homologue (Porquet, A, et al.2007), (Rosen, BR, et al.2009) . The GlpF aquaglycerol porin is a membrane protein with a symmetric arrangement of four independent GlpF channels. One monomer of this tetramer GlpF porin consists of six transmembrane and two half membrane-spanning α-helices that form a right-handed helical bundle around the channel. The channel has a diameter of ~15Å at the periplasmid end, which constricts towards a diameter of ~3.8Å at the beginning of a 28 Å long selective channel that ends at the cytoplasmic end (Fu, DX, et al.2000). The GlpF is a stereospecific channel that is thought to be more selective on molecular size than on chemical structure (Fu, DX, et al.2000, (Heller, KB, et al.1980) . It does allow transport of a variance of non-charged compounds ranging from polyhydric alcohols, glycerol being one of them, arsenic to antimone (Fu, DX, et al.2000), (Meng, YL, et al.2004), (Porquet, A, et al.2007), (Rosen, BR, et al.2009), (Heller, KB, et al.1980). Carbon sugars and ions are shown to be unable to be transported by GlpF (Heller, KB, et al.1980). At physiological pH arsenic and antimone are not present in their ionic state but rather as As(OH)3 and Sb(OH)3 (Rosen, BR, et al.2009). These elements show a charge distribution similar to glycerol and a smaller but comparable volume. The structural similarities are thought to be the reason for the possibility of these elements to enter the cell by GlpF (Porquet, A, et al.2007), GlpF facilitates transport of these compounds down there gradient (inside or outside the cell). If GlpF behaves as a nonsaturable transporter, a transport rate of 1umol of glycerol is transported per minute per mgr of cell protein (Heller, KB, et al.1980).
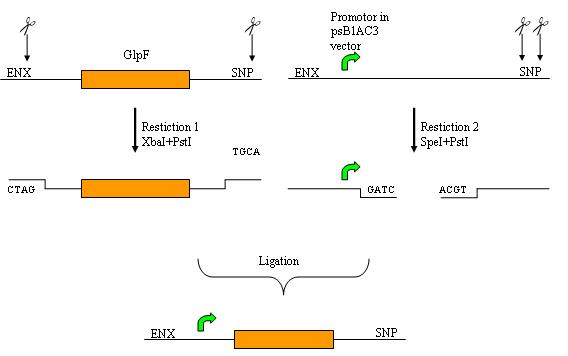
Cloning strategy
This part has been obtained from the genome of E.coli 356 in two steps with PCR. First the whole gene was obtained from the genome by using PCR and in the second step an EcoR1 restiction site was removed.
The GlpF PCR product was restricted with XbaI and PstI and a psB1AC3 vector with a pMed promotor was restricted with SpeI and PstI. The restriction products were ligated. This resulted in a psB1AC3 vector with a promotor and GlpF.
Results
The ability of GlpF (overexpressed under IPTG induction) to transport As(III) was tested by an arsenite uptake assay. Also the full accumulation device () was tested using this assay. Data and analysis can be found here.
The graphs above represent the result of the metal sensitivity assay. The lines in the graphs represent the average optical density (OD600) of a construct over time. The graph on the left shows that increased As(III) levels inhibits growth and as the As(III) concentration increases the plateau OD600 level decreases.
The second graph represents the OD600 values of a E. coli culture with pLac-GlpF. The growth curves are less steep in the log phase compared to WT, because of the protein expression by IPTG induction. In the absence of As(III) the plateau level equals the WT, but if arsenite is present the plateaus are lower (OD600 <0.8) which implies functional expression of GlpF causing As(III) uptake to toxic levels.
In the graph on the right, shows growth curves of E. coli expressing GlpF and fMT under a low constitutive promoter. Also a similar growth in the log phase, compared to pLac GlpF, can be seen this is due to protein expression. Furthermore, like WT (0μM As(III)) it has its plateau above OD600 0.9. Upon incubation with various arsenite concentrations, the plateaus are lower (OD600 <0.8) compared to WT. This is due to As(III) uptake by GlpF. The reduced growth is also an indicator for arsenite uptake. It is difficult to see if fMT has any effect because this assay can not show where the arsenite is and how fMT interferes with the cells' detoxification.
Modelling uptake GlpF
The import of As(III) via GlpF is modelled as a simple import reaction with Michaelis-Menten kinetics, in part because this makes it easy to specify, but also because we only have very high level data. The following allows a comparison with the data acquired from figure 1B from Meng 2004.
|
Loading graph...
|
To determine the constants v5 and K5 we performed the following steps:
- Read the wild-type line in figure 1B of Meng 2004 by pasting it in a drawing program and aligning/scaling the axes and then manually determining the coordinates of each data point.
- Converted to units of concentration using the data in Meng 2004 and [http://gchelpdesk.ualberta.ca/CCDB/cgi-bin/STAT_NEW.cgi the CCDB] (assuming that the cells are resting/non-growing), see our [http://spreadsheets.google.com/pub?key=t4gilzCbEaCFAvpEVWUE_zQ Google Docs spreadsheet]. Here we disregarded the fact that the measurements were made by taking out 0.1mL samples, as this does not change the concentrations. Specifically (note that uptake is in nmol/mg):
- uptaketotal (nmol) = uptake · 8mg · 0.3 The ratio between dry and wet weight is 0.3 (see the [http://gchelpdesk.ualberta.ca/CCDB/cgi-bin/STAT_NEW.cgi CCDB]).i
- As(III)ex (µM=nmol/mL) = (10nmol/mL · 1mL - uptaketotal) / (1.1-0.0073)mL The experiment started with 1mL of a 10µM=10nmol/mL solution of As(III). After adding the cells the total volume of the solution was 1.1mL, and 0.0073mL is an estimate of the total volume of cells in the solution, see below.i
- uptaketotal (nmol) = uptake · 8mg · 0.3
- Fit the Michaelis-Menten equation to find the constants v5 and K5 in Mathematica (see [http://igemgroningen.googlecode.com/svn/trunk/buoyant/Models/Meng2004%20Figure%201B.nb the Mathematica notebook in SVN]) using the method from Goudar 1999 (a least squares fit of a closed-form solution of the differential equation).
Export of arsenicum via Ars operon
GlpF is the importer of arsenicum. After arsenicum enters the cell, in response the Ars operon produces ArsR. At the same time, ArsB is also produced by Ars operon. This happens because the Ars operon contains three open reading frames: the first is ArsR, second ArsB and the last one is ArsC. ArsB is the exporter of arsenicum. The ars operon is located on the chromosomal DNA of E. coli. For more information see: [http://biocyc.org/ECOLI/NEW-IMAGE?type=GENE-IN-CHROM-BROWSER&object=EG12235 biocyc].
Additional sources
- Meng 2004 (As(III) and Sb(III) Uptake by GlpF and Efflux by ArsB in Escherichia coli)
- Rosen 2009 (Transport pathways for arsenic and selenium: A minireview)
- Porquet, A, et al.2007 (structural similarity between As(OH)3 and glycerol)
- Fu, DX, et al.2000 (Structure of the GlpF channel)
- Heller, KB, et al.1980 (Glycerol transport properties of GlpF)
Copper/zinc uptake via HmtA
HmtA
Introduction
HmtA(heavy metal transporter A) from Pseudomonas aeruginosa [http://www.ncbi.nlm.nih.gov/protein/81857196 Q9I147] is a P-type ATPase importer. This membrane protein mediates the uptake of copper (Cu) and zinc (Zn) and was functionally expressed in E. coli ([http://www.ncbi.nlm.nih.gov/pubmed/19264958 Lewinson 2009]). We want to use this membrane protein to accumulate copper and zinc into the cells. we believe this ATP-driven pump is capable of generating an elevated intracellular concentration of these compounds enabling the harvesting of copper and zinc from the medium.
Cloning strategy
There are several restriction sites to be modified from Lewinson's pBAD construct. A vector with amp resistance with L-arabinose inducible HmtA-6HIS. The restriction sites have been silently mutated maintaining the amino acid sequence. We will create these mutations via PCR than digest the old methylated template and clone the product into competent cells.
Results
So far we have cloned HmtA as a biobrick without EcoRI site in the coding region into the iGEM vector. Unfortunately a mutation occurred at base 103 from the start of the orf. By a point mutation c to t in the first nucleotide of the codon changed arginine 35 to a Cysteine. We do not know the effects but we suspect it might have a great influence due to the very reactive side chain of Cysteine, eventhough it is not in the channel itself based on [http://www.cbs.dtu.dk/services/TMHMM/ TMHMM] predictions which indicate trans membrane helices of a protein. Further cloning is expected to be unsuccessful because the IPTG induced clones grow even slightly better than the empty vector control. This is most likely cause by the missing pLAC-RBS in front of the gene. There was no positive controle with the L-arabinose inducable HmtA-6His in pBAD. We did do expression experiments with the pBAD construct to purified the membrane protein as quality controle. result shown in the figure on the right.
Heavy metal uptake coupled to citrate via efCitH bsCitM
Force feeding of the heavy metals into the cell is possible when citrate is the only available carbon source. Citrate in complex with heavy metals can be translocated over the membrane into the cell via citrate transporters. This can be a very efficient strategy to accumulate vast ammounts of heavy metals. The two membrane proteins are CitM from Bacillus subtilis studied by [http://www.ncbi.nlm.nih.gov/pubmed/11053381 B.P Krom]. BsCitM can transport citrate in complex with Mg2+, Ni2+, Mn2+, Co2+, and Zn2+. The other is CitH from Enterococcus faecalis described by [http://www.ncbi.nlm.nih.gov/pubmed/17042778 V.S Blancato]. EfCitH catalyzes translocation of the citrate in complex with Ca2+, Sr2+ Mn2+ Mn2+ Cd2+ and Pb2+.
Additional sources
More information on this topic can be found in:
Bastiaan Krom. Citrate transporters of Bacillus subtilis PhD thesis. http://dissertations.ub.rug.nl/faculties/science/2002/b.p.krom/ Dissertation Groningen
Jessica B. Warner. Regulation and expression of the metal citrate transporter CitM PhD thesis. http://dissertations.ub.rug.nl/faculties/science/2002/j.b.warner/ Dissertation Groningen
Periplasmic accumulation of heavy metals via Mer Operon
Periplasmic accumulation of heavy metals via Mer proteins enables the harvesting of heavy metals from the medium by binding the cytosolic and periplasmic metals to metallothionein and transporting the metal-protein complex into the periplasm. The MerR family consists of different proteins for one specific metal (i.e. PbrR (lead), CueR (copper), ZntR (zinc), MerR (mercury), ArsR (arsenic), CadR (cadmium)).
As the cells die after uptake of Mg (and induction of the Mer transporter), this system is not very well usable for our project. The dead cells will not produce the gas vesicles (it may be used however by having the gas vesicles consitutively expressed), thereby bouyancy may be a problem (Pennella 2005, Kao 2008).
 "
"