Team:Groningen/Notebook/12 August 2009
From 2009.igem.org
m (→GVP Cluster) |
m (→GVP Cluster) |
||
| (10 intermediate revisions not shown) | |||
| Line 7: | Line 7: | ||
:→ {{done}} isolate plasmids from o.n. cultures | :→ {{done}} isolate plasmids from o.n. cultures | ||
:→ {{done}} run gel to check plasmid size (should be ~9000bp) | :→ {{done}} run gel to check plasmid size (should be ~9000bp) | ||
| - | :→ {{ | + | :→ {{done}} analyse results of gel |
| + | :→ {{done}} restriction analysis of plasmids with EcoRI/PstI | ||
| + | :→ {{done}} plate remaining o.n. culture on plates for glycerol stocks | ||
| + | :→ {{done}} restriction of GVP biobrick plasmid, pSB1AC3-H, pSB3K3-L/M/H for assembly | ||
| + | :→ {{done}} gel purification of wanted fragments | ||
| + | |||
| + | '''Plates''' | ||
| + | |||
| + | Showed single colony growth and were stored in the fridge for future preculture growth. | ||
'''Over Night Cultures''' | '''Over Night Cultures''' | ||
| Line 27: | Line 35: | ||
From each cup with isolated plasmid 5μL was mixed with 1μL 6x loading buffer and incubated for 10 minutes. Together with 1kB ladder all samples were loaded on a 1% agarose gel with 4μL EtBr. | From each cup with isolated plasmid 5μL was mixed with 1μL 6x loading buffer and incubated for 10 minutes. Together with 1kB ladder all samples were loaded on a 1% agarose gel with 4μL EtBr. | ||
| + | [[Image:Gel Mic no1.jpg|450px]] [[Image:Generulers_1kb_marker_Fermentas.jpg]] | ||
| - | :→ Gel shows bands at ~ | + | :→ From left to right:1kB ladder, 4x pSB1AC3-med.promoter-GVP cluster, 4x pSB1AC3-low.promoter-GVP |
| + | |||
| + | :→ Gel shows bands at ~9000bp and might indicate correct ligation of GVP cluster into pSB1AC3 vector with medium and low promoters. The absence of clear lines at ~10000bp is due to the uncut round shape of the plasmids. | ||
:→ The concentration of plasmids is higher for the low constitutive promoter, which might indicate more difficulty for E.coli to grow at higher GVP expression (to much energy needed). | :→ The concentration of plasmids is higher for the low constitutive promoter, which might indicate more difficulty for E.coli to grow at higher GVP expression (to much energy needed). | ||
:→ Next will be restriction analysis of the plasmids!! | :→ Next will be restriction analysis of the plasmids!! | ||
| Line 72: | Line 83: | ||
'''Restriction Analysis''' | '''Restriction Analysis''' | ||
| + | |||
| + | The vector [http://partsregistry.org/wiki/index.php?title=Part:pSB1AC3 pSB1AC3] containing the [http://partsregistry.org/wiki/index.php?title=Part:BBa_J23106 medium] and [http://partsregistry.org/wiki/index.php?title=Part:BBa_J23109 low] constitutive promoters with [http://partsregistry.org/wiki/index.php?title=Part:BBa_I750016 GVP] were cut with EcoRI/PstI for control of insert. | ||
| + | |||
| + | * 9 μL MQ | ||
| + | * 7μL plasmid (med. promoter) in MQ (0.8-1.0μg) (2x) | ||
| + | * 2μL Fast digest buffer | ||
| + | * 1μL PstI fast digest enzyme | ||
| + | * 1μL EcoRI fast digest enzyme | ||
| + | |||
| + | |||
| + | * 14 μL MQ | ||
| + | * 2μL plasmid (low promoter) in MQ (0.8-1.0μg) (2x) | ||
| + | * 2μL Fast digest buffer | ||
| + | * 1μL PstI fast digest enzyme | ||
| + | * 1μL EcoRI fast digest enzyme | ||
| + | |||
| + | and put at 37°C for 30 minutes, followed by addition of 4μL 6x loading buffer. | ||
| + | |||
| + | '''Gel''' | ||
| + | |||
| + | [[Image:Gel Mic no.2.jpg|250px]] [[Image:Generulers_1kb_marker_Fermentas.jpg]] | ||
| + | |||
| + | :→ From left to right:1kB ladder, 2x pSB1AC3-med.promoter-GVP cluster EcoRI/PstI, 2x pSB1AC3-low.promoter-GVP EcoRI/PstI. | ||
| + | |||
| + | :→ Gel shows bands at ~6200bp and ~3000bp, which would indicate GVP+promoter and vector pSB1AC3. If the GVP cluster would still be in its original vector fragments of ~7000bp and ~2000bp would be expected. | ||
| + | |||
| + | '''Plates''' | ||
| + | |||
| + | LB-agar plates with 50 μg/mL ampicillin (which were freshly prepared yesterday) and plates with 25 μg/mL ampicillin/50 μg/mL chloram. were used, and one drop of each o.n. preculture checked with restriction was spotted and subsequently swiped three times with sterile needle. The plates were stored at 37°C overnight. | ||
| + | |||
| + | '''Restriction for Assembly''' | ||
| + | |||
| + | The vector [http://partsregistry.org/wiki/index.php?title=Part:pSB3K3 pSB3K3] containing the [http://partsregistry.org/wiki/index.php?title=Part:BBa_J23100 high], [http://partsregistry.org/wiki/index.php?title=Part:BBa_J23106 medium] and [http://partsregistry.org/wiki/index.php?title=Part:BBa_J23109 low] constitutive promoters, the [http://partsregistry.org/wiki/index.php?title=Part:BBa_J GVP] biobrick plasmid, and the vector [http://partsregistry.org/wiki/index.php?title=Part:pSB1AC3 pSB1AC3] containing the [http://partsregistry.org/wiki/index.php?title=Part:BBa_J23100 high] constitutive promoter. | ||
| + | |||
| + | '''Purification''' | ||
| + | |||
| + | [[Image:Gel Mic no.3.jpg|450px]] [[Image:Generulers_1kb_marker_Fermentas.jpg]] | ||
| + | |||
| + | :→ From left to right:1kB ladder, Empty Slot, 2x GVP, Empty Slot, pSB1AC3-high.promoter, pSB3K3-H/M/L | ||
| + | |||
| + | '''Concentration''' | ||
| + | |||
| + | {|cellpadding="2" cellspacing="1" border="4" | ||
| + | |'''Plasmid''' | ||
| + | |'''Conc. ng/μL''' | ||
| + | |'''260/280 | ||
| + | |'''260/230 ''' | ||
| + | |'''-20 box (michael | ||
| + | |'''Restriction Control''' | ||
| + | |- | ||
| + | |GVP-cluster (XbaI/PstI) | ||
| + | |10.4 | ||
| + | |1.43 | ||
| + | |0.84 | ||
| + | |Not Stored | ||
| + | |No | ||
| + | |- | ||
| + | |pSB1AC3-BBa_J23100 (SpeI/PstI) | ||
| + | |16.1 | ||
| + | |1.63 | ||
| + | |1.44 | ||
| + | |Not Stored | ||
| + | |No | ||
| + | |- | ||
| + | |pSB3K3-BBa_J23100(H) (SpeI/PstI) | ||
| + | |5.0 | ||
| + | |1.65 | ||
| + | |0.84 | ||
| + | |Not Stored | ||
| + | |No | ||
| + | |- | ||
| + | |pSB3K3-BBa_J23106(M) (SpeI/PstI) | ||
| + | |5.4 | ||
| + | |1.25 | ||
| + | |0.88 | ||
| + | |Not Stored | ||
| + | |No | ||
| + | |- | ||
| + | |pSB3K3-BBa_J23109(L) (SpeI/PstI) | ||
| + | |6.5 | ||
| + | |1.52 | ||
| + | |0.88 | ||
| + | |Not Stored | ||
| + | |No | ||
| + | |} | ||
===Transporters=== | ===Transporters=== | ||
| Line 85: | Line 181: | ||
==Dry== | ==Dry== | ||
| - | + | We updated the literature list of all the projects. | |
{{Team:Groningen/Notebook/Day/Footer}} | {{Team:Groningen/Notebook/Day/Footer}} | ||
Latest revision as of 11:28, 17 August 2009
[http://2009.igem.org/Team:Groningen http://2009.igem.org/wiki/images/f/f1/Igemhomelogo.png]
|
|---|
Wet
GVP Cluster
- → DONE isolate plasmids from o.n. cultures
- → DONE run gel to check plasmid size (should be ~9000bp)
- → DONE analyse results of gel
- → DONE restriction analysis of plasmids with EcoRI/PstI
- → DONE plate remaining o.n. culture on plates for glycerol stocks
- → DONE restriction of GVP biobrick plasmid, pSB1AC3-H, pSB3K3-L/M/H for assembly
- → DONE gel purification of wanted fragments
Plates
Showed single colony growth and were stored in the fridge for future preculture growth.
Over Night Cultures
- → All cultures showed growth, but the low constitutive promoter containing cells showed higher growth compared to the medium promoter.
- → Also the E.coli cells with medium promoter were more concentrated on the bottem of the tubes, as compared to the low promoter, which showed a more homogenus solution.
Plasmid Purification
Plasmid isolation was performed on the cultures of E.coli TOP10 containing plasmids [http://partsregistry.org/wiki/index.php?title=Part:pSB1AC3 pSB1AC3] with [http://partsregistry.org/wiki/index.php?title=Part:BBa_J23100 medium] and [http://partsregistry.org/wiki/index.php?title=Part:BBa_J23100 low] constitutive promoters and GVP with the "Sygma-Aldrich™ [http://www.sigmaaldrich.com/life-science/molecular-biology/dna-and-rna-purification/plasmid-miniprep-kit.html GenElute™] Plasmid Miniprep Kit".
- From each tube 4mL of culture was collected in a 2.0mL cup, and the cells were pelleted by centrifugation for 1 min. at max. speed and the supernatant discarded.
- Plasmids were eluted with 20μL MQ and stored in the fridge
Gel
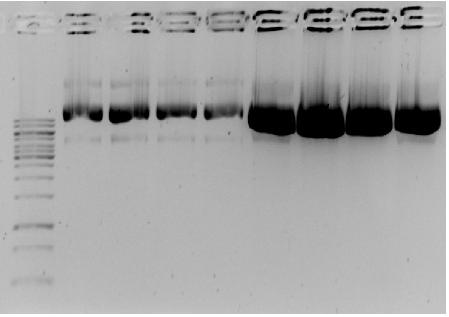
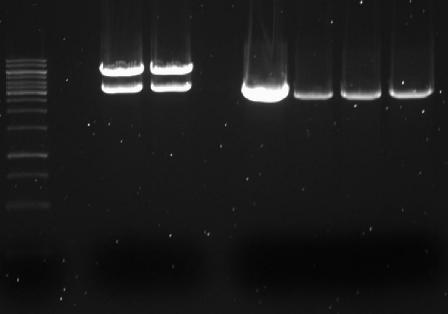
From each cup with isolated plasmid 5μL was mixed with 1μL 6x loading buffer and incubated for 10 minutes. Together with 1kB ladder all samples were loaded on a 1% agarose gel with 4μL EtBr.
- → From left to right:1kB ladder, 4x pSB1AC3-med.promoter-GVP cluster, 4x pSB1AC3-low.promoter-GVP
- → Gel shows bands at ~9000bp and might indicate correct ligation of GVP cluster into pSB1AC3 vector with medium and low promoters. The absence of clear lines at ~10000bp is due to the uncut round shape of the plasmids.
- → The concentration of plasmids is higher for the low constitutive promoter, which might indicate more difficulty for E.coli to grow at higher GVP expression (to much energy needed).
- → Next will be restriction analysis of the plasmids!!
Concentration
| Plasmid | Conc. ng/μL | 260/280 | 260/230 | -20 box (michael | Restriction Control |
| pSB1AC3-BBa_J23106-GVP (med. no.1) | 125.0 | 1.79 | 2.31 | ? | No |
| pSB1AC3-BBa_J23106-GVP (med. no.2) | 134.4 | 1.83 | 2.35 | ? | No |
| pSB1AC3-BBa_J23109-GVP (low no.5) | 528.9 | 1.90 | 2.34 | ? | No |
| pSB1AC3-BBa_J23109-GVP (low no.5) | 708.4 | 1.90 | 2.17 | ? | No |
Restriction Analysis
The vector [http://partsregistry.org/wiki/index.php?title=Part:pSB1AC3 pSB1AC3] containing the [http://partsregistry.org/wiki/index.php?title=Part:BBa_J23106 medium] and [http://partsregistry.org/wiki/index.php?title=Part:BBa_J23109 low] constitutive promoters with [http://partsregistry.org/wiki/index.php?title=Part:BBa_I750016 GVP] were cut with EcoRI/PstI for control of insert.
- 9 μL MQ
- 7μL plasmid (med. promoter) in MQ (0.8-1.0μg) (2x)
- 2μL Fast digest buffer
- 1μL PstI fast digest enzyme
- 1μL EcoRI fast digest enzyme
- 14 μL MQ
- 2μL plasmid (low promoter) in MQ (0.8-1.0μg) (2x)
- 2μL Fast digest buffer
- 1μL PstI fast digest enzyme
- 1μL EcoRI fast digest enzyme
and put at 37°C for 30 minutes, followed by addition of 4μL 6x loading buffer.
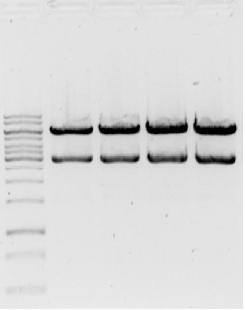
Gel
- → From left to right:1kB ladder, 2x pSB1AC3-med.promoter-GVP cluster EcoRI/PstI, 2x pSB1AC3-low.promoter-GVP EcoRI/PstI.
- → Gel shows bands at ~6200bp and ~3000bp, which would indicate GVP+promoter and vector pSB1AC3. If the GVP cluster would still be in its original vector fragments of ~7000bp and ~2000bp would be expected.
Plates
LB-agar plates with 50 μg/mL ampicillin (which were freshly prepared yesterday) and plates with 25 μg/mL ampicillin/50 μg/mL chloram. were used, and one drop of each o.n. preculture checked with restriction was spotted and subsequently swiped three times with sterile needle. The plates were stored at 37°C overnight.
Restriction for Assembly
The vector [http://partsregistry.org/wiki/index.php?title=Part:pSB3K3 pSB3K3] containing the [http://partsregistry.org/wiki/index.php?title=Part:BBa_J23100 high], [http://partsregistry.org/wiki/index.php?title=Part:BBa_J23106 medium] and [http://partsregistry.org/wiki/index.php?title=Part:BBa_J23109 low] constitutive promoters, the [http://partsregistry.org/wiki/index.php?title=Part:BBa_J GVP] biobrick plasmid, and the vector [http://partsregistry.org/wiki/index.php?title=Part:pSB1AC3 pSB1AC3] containing the [http://partsregistry.org/wiki/index.php?title=Part:BBa_J23100 high] constitutive promoter.
Purification
- → From left to right:1kB ladder, Empty Slot, 2x GVP, Empty Slot, pSB1AC3-high.promoter, pSB3K3-H/M/L
Concentration
| Plasmid | Conc. ng/μL | 260/280 | 260/230 | -20 box (michael | Restriction Control |
| GVP-cluster (XbaI/PstI) | 10.4 | 1.43 | 0.84 | Not Stored | No |
| pSB1AC3-BBa_J23100 (SpeI/PstI) | 16.1 | 1.63 | 1.44 | Not Stored | No |
| pSB3K3-BBa_J23100(H) (SpeI/PstI) | 5.0 | 1.65 | 0.84 | Not Stored | No |
| pSB3K3-BBa_J23106(M) (SpeI/PstI) | 5.4 | 1.25 | 0.88 | Not Stored | No |
| pSB3K3-BBa_J23109(L) (SpeI/PstI) | 6.5 | 1.52 | 0.88 | Not Stored | No |
Transporters
Metal Accumulation
MBP-ArsR fusion protein ligation was transformed for a second try, plated out on LB-amp100 (50 μL and concentrated after centrifugation) Positive control (pSB1AC3-high constitutive promotor) ∞ Negative control (pSB1AC3-ccdb)1 colony on the concentrated plate Ligations had no colonies.
Vectors
Dry
We updated the literature list of all the projects.
|
|
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 "
"