Team:Newcastle/Labwork/19 August 2009
From 2009.igem.org
TubularWorld (Talk | contribs) (New page: {{:Team:Newcastle/CSS}} {{:Team:Newcastle/Header}} {{:Team:Newcastle/Left}} =Lab 19/08/09= ==<u>Sporulation Tuning/ Chassis Team</u>== ===Summary=== [[Image:Newcastle 19 August Jane inspe...) |
(→Lab 19/08/09) |
||
| Line 14: | Line 14: | ||
[[Image:Newcastle 19 August sleB count 102.jpg|thumb|counting colonies on the sleB/cwlJ plate 10<sup>-2</sup>.]] | [[Image:Newcastle 19 August sleB count 102.jpg|thumb|counting colonies on the sleB/cwlJ plate 10<sup>-2</sup>.]] | ||
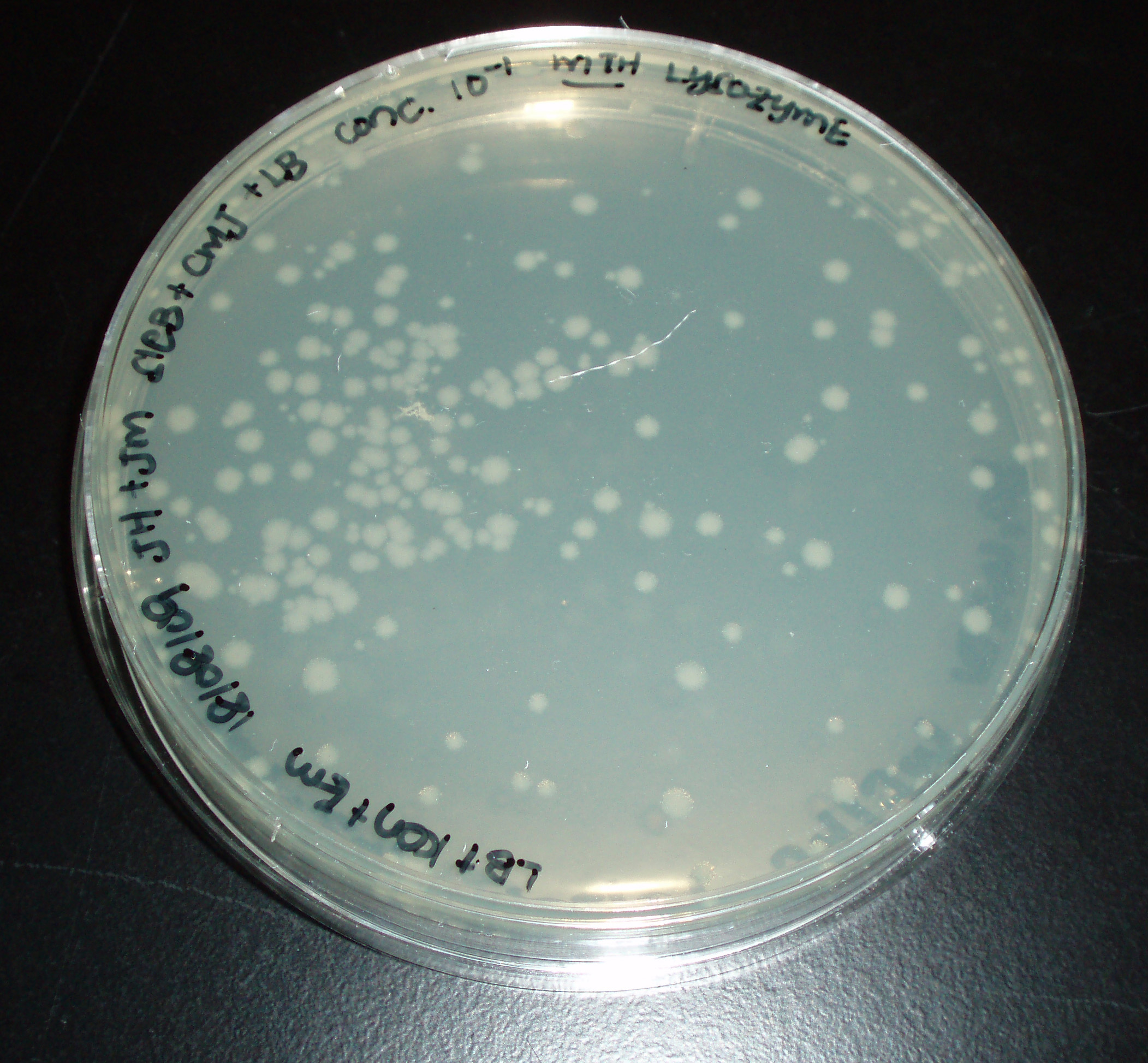
[[Image:Newcastle 19 August sleB noly.jpg|thumb|sleB/cwlJ no lysozyme plate.]] | [[Image:Newcastle 19 August sleB noly.jpg|thumb|sleB/cwlJ no lysozyme plate.]] | ||
| + | <br> | ||
| + | ==<u>Metal Sensing Team</u>== | ||
| + | ===Introduction=== | ||
| + | So far in the lab, we have prepared the BioBrick ''BBa_J33206'' (''arsR'' promoter + coding sequence) from the repository and attempted transformation of this DNA in ''E.coli''. Once this procedure had been completed, the Metal Sensing team then streaked the bacteria on an LB + ampicillin plate and left it on 37°C incubation overnight. | ||
| + | |||
| + | '''Today’s procedure involves searching the plate for any colonies and if any colonies are present, picking three of them and inoculating 3ml LB with them'''. These grown up cultures will then be used for a mini-prep of the ''BBa_J33206'' BioBrick. If no colonies are present, we must attempt transformation again. Because the plasmid involved in this BioBrick (''pSB1A2'') is a high copy plasmid, we can inoculate 3ml LB + ampicillin in the morning; by the afternoon the ''E.coli'' should have grown in the LB solution sufficiently to further inoculate 50ml LB solution in the afternoon (for midi-preps). | ||
| + | <br> | ||
| + | ===Practical Outline=== | ||
| + | These are the steps that the Metal Sensing Team want to have completed by the end of the day: | ||
| + | <br> | ||
| + | # Innoculate three tubes (each with 3ml of LB) with three of the ''E.coli'' cultures containing ''BBa_J33206'' - morning | ||
| + | # Start preparing for a test PCR for the ''pGFP-rrnb'' plasmid in ''Bacillus subtilis''. | ||
| + | # Should the ''E.coli'' cultures (containing ''BBa_J33206'') have grown sufficiently in the LB tubes, further inoculate 3 flasks (containing 50ml of LB) with these bacteria – afternoon. | ||
| + | <br> | ||
| + | ===Observations and Procedure=== | ||
| + | When looking at the plate following an overnight incubation it could be seen that the transformation rate for the ''DH5α E.coli'' containing ''BBa_J33206'' DNA was very low – only five colonies were spotted on the plate. | ||
| + | Nevertheless three colonies were removed from the plate and, under aseptic conditions, used to inoculate 3 tubes containing 3ml LB each. These tubes were then placed in the shaking incubator at 37°C for the rest of the day. This step was done first thing in the morning. The bacteria which grow in these LB tubes will be used for a mini-prep of the ''BBa_J33206'' BioBrick tomorrow | ||
| + | <br> | ||
| + | In the afternoon, the team returned to the lab to observe the progress of the ''E.coli'' bacterial growth in the three LB tubes. The appearance of the three tubes was noted as cloudy which indicated the presence of sufficient numbers of ''E.coli'' bacteria. To save time in the labs, it was decided to inoculate 3 flasks (each containing 50ml LB) with these three samples – these 50ml flasks will then be used for midi-preps tomorrow should the mini-preps indicate that the E.coli have been successfully transformed. | ||
| + | <br> | ||
| + | To inoculate the 3 flasks containing 50ml LB each, the team took 0.5ml from each of the 3ml of cultures and pipette them into the separate flasks. These flasks were then placed into the orbital shaking incubator at 37°C overnight. | ||
{{:Team:Newcastle/Footer}} | {{:Team:Newcastle/Footer}} | ||
{{:Team:Newcastle/Right}} | {{:Team:Newcastle/Right}} | ||
Revision as of 20:35, 23 August 2009
Contents |
Lab 19/08/09
Sporulation Tuning/ Chassis Team
Summary
Metal Sensing Team
Introduction
So far in the lab, we have prepared the BioBrick BBa_J33206 (arsR promoter + coding sequence) from the repository and attempted transformation of this DNA in E.coli. Once this procedure had been completed, the Metal Sensing team then streaked the bacteria on an LB + ampicillin plate and left it on 37°C incubation overnight.
Today’s procedure involves searching the plate for any colonies and if any colonies are present, picking three of them and inoculating 3ml LB with them. These grown up cultures will then be used for a mini-prep of the BBa_J33206 BioBrick. If no colonies are present, we must attempt transformation again. Because the plasmid involved in this BioBrick (pSB1A2) is a high copy plasmid, we can inoculate 3ml LB + ampicillin in the morning; by the afternoon the E.coli should have grown in the LB solution sufficiently to further inoculate 50ml LB solution in the afternoon (for midi-preps).
Practical Outline
These are the steps that the Metal Sensing Team want to have completed by the end of the day:
- Innoculate three tubes (each with 3ml of LB) with three of the E.coli cultures containing BBa_J33206 - morning
- Start preparing for a test PCR for the pGFP-rrnb plasmid in Bacillus subtilis.
- Should the E.coli cultures (containing BBa_J33206) have grown sufficiently in the LB tubes, further inoculate 3 flasks (containing 50ml of LB) with these bacteria – afternoon.
Observations and Procedure
When looking at the plate following an overnight incubation it could be seen that the transformation rate for the DH5α E.coli containing BBa_J33206 DNA was very low – only five colonies were spotted on the plate.
Nevertheless three colonies were removed from the plate and, under aseptic conditions, used to inoculate 3 tubes containing 3ml LB each. These tubes were then placed in the shaking incubator at 37°C for the rest of the day. This step was done first thing in the morning. The bacteria which grow in these LB tubes will be used for a mini-prep of the BBa_J33206 BioBrick tomorrow
In the afternoon, the team returned to the lab to observe the progress of the E.coli bacterial growth in the three LB tubes. The appearance of the three tubes was noted as cloudy which indicated the presence of sufficient numbers of E.coli bacteria. To save time in the labs, it was decided to inoculate 3 flasks (each containing 50ml LB) with these three samples – these 50ml flasks will then be used for midi-preps tomorrow should the mini-preps indicate that the E.coli have been successfully transformed.
To inoculate the 3 flasks containing 50ml LB each, the team took 0.5ml from each of the 3ml of cultures and pipette them into the separate flasks. These flasks were then placed into the orbital shaking incubator at 37°C overnight.
News
Events
- 20 – 21 June 2009 - Europe workshop (London)
- 23 – 24 June 2009 - UK iGEM meetup (Edinburgh)
- 23 October Practice Presentation (Newcastle)
- 23 October T-shirts are ready
- 27 October Practice Presentation (Sunderland)
- 27 October Poster is ready
- 30 October – 2 November 2009 - Jamboree (Boston)
Social Net
- Newcastle iGEM Twitter
- [http://www.facebook.com/home.php#/group.php?gid=131709337641 Newcastle on Facebook]
- [http://www.youtube.com/user/newcastle2009igem Newcastle Youtube Channel]
 "
"