Team:UNIPV-Pavia/Notebook/Week4Jul
From 2009.igem.org
(Difference between revisions)
(→July, 23rd) |
(→July, 24th) |
||
| Line 215: | Line 215: | ||
<font class='didascalia'> | <font class='didascalia'> | ||
{|align="center" | {|align="center" | ||
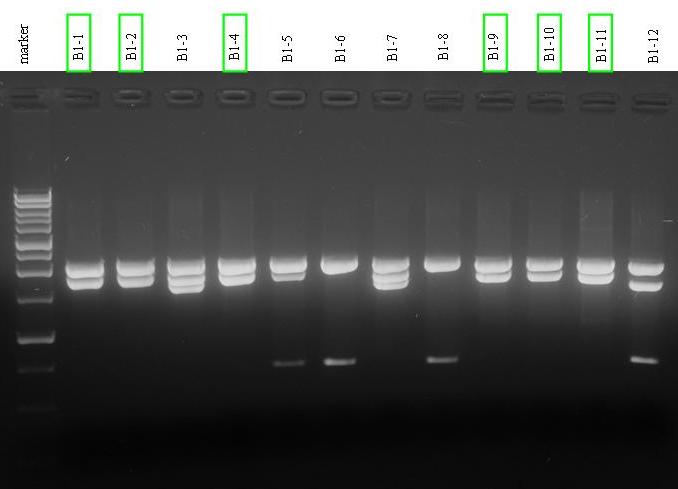
| - | |[[Image:pv_digestion_B1.jpg|thumb|500px|left|]] | + | |[[Image:pv_digestion_B1.jpg|thumb|500px|left|B1 digestion E-P (samples 1 to 12)]] |
|} | |} | ||
</font> | </font> | ||
<font class='didascalia'> | <font class='didascalia'> | ||
{|align="center" | {|align="center" | ||
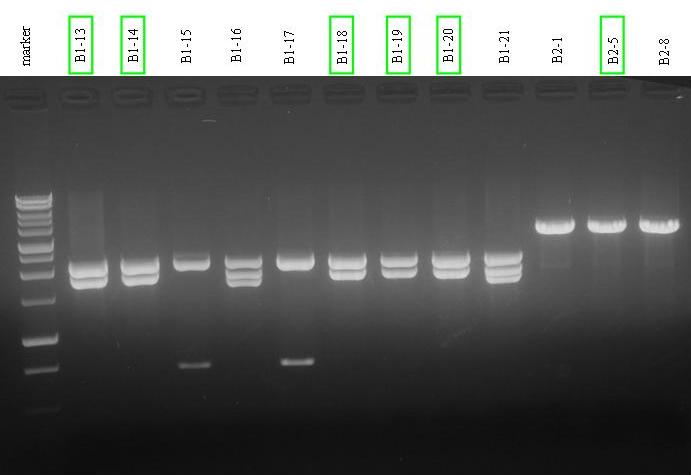
| - | |[[Image:pv_digestion_B1_B2.jpg|thumb|500px|left|]] | + | |[[Image:pv_digestion_B1_B2.jpg|thumb|500px|left|lanes 2 to 10: B1 digestion E-P (samples 13 to 21), lanes 11 to 13: B2 digestion S-P]] |
|} | |} | ||
</font> | </font> | ||
Revision as of 12:01, 30 August 2009

|
|
|
|
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
Week from July 20th, to July 26th, 2009
 Previous Week
Previous Week
|
Next Week

|
July, 20th
- Overnight digestion (20 ul reaction volume) for:
| MRGENE1(X-P) | MRGENE1-2(X-P) | MRGENE1-3(X-P) |
| MRGENE2(X-P)(X2) | MRGENE2-2(X-P) | MRGENE2-3(X-P) |
| B0030(S-P)(X3) |
- NOTE: reactions which involve excision(X-P) were performed on 3 ug of DNA, while S-P digestions were performed with 1 ug of DNA. We decided to have 3 replicates to be sure to have an acceptable yield.
- We decided to perform gel run/cut/purification for MRGENE2 and for B0030, while we decided to perform DNA precipitation with sodium acetate for MRGENE1.
- We transformed about 2.5 pg of A14-4 in TOP10 in order to filter the wanted plasmids. We plated the transformed bacteria and incubated the plates at 37°C overnight.
- We infected 5 ml of LB + Amp with 10 ul of A12-2 and A12-3 glycerol stocks. Tomorrow they will be miniprepped and sent to sequence!
Preparation of experiment with Tecan F200
- We infected 5 ml of LB + Kan with a single colony taken from the native plate of B0015.
- We incubated the inoculum for 6 hours (37°C, 220 rpm).
Experiment with Tecan F200
- Description
- Purpose:
- Materials & Methods
- Protocol
- Results
July, 21st
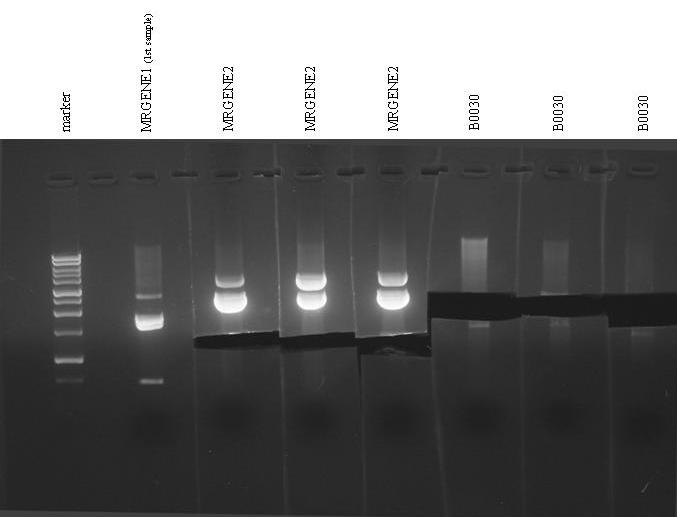
- Gel run for:
- MRGENE1(for check)
- MRGENE2
- MRGENE2-2
- MRGENE2-3
- B0030(X3)
- We noticed unwanted bands in MRGENE1 run...So, we performed an analysis on Mr Gene plasmids and we found an unwanted PstI site in pMK-RQ that gives the noticed bands...
- We decided to proceed to ligation and to perform a massive screening of the ligated inserts in the next days.
- Gel purification for:
- MRGENE2
- MRGENE2-2
- MRGENE2-3
- B0030(X3)
- DNA precipitation with sodium acetate for MRGENE1, MRGENE1-2, MRGENE1-3.
- Results: after quantifications at Nanodrop, all the purified DNA samples had a good yield! let's proceed to ligation!
- Ligation:
- B1 = B0030(S-P) + MRGENE1(X-P)
- B2 = B0030(S-P) + MRGENE2(X-P)
- We incubated the ligations at 16°C overnight.
- Miniprep for A12-2 and A12-3. We sent purified DNA to BMR Genomics for sequencing.
- A14pg plate showed colonies! we picked 7 colonies and infected 5 ml of LB + Amp. We incubated the 7 inocula at 37°C, 220 rpm overnight.
- We received sequencing results for F2620: sequence was not consistent, which confirmed the results of iGEM 2009 QC.
July, 22nd
- We received the following stabs from iGEM HQ:
| K116001 | K116002 | K112405 |
| P0412 | I746902 | I746903 |
| K101017 |
- while F2620 will be shipped soon.
- We transformed B1 and B2 overnight ligations in TOP10. Plates were incubated at 37°C overnight.
- Glycerol stocks and miniprep for the 7 overnight cultures of A14pg.
- Digestion E-P for the miniprepped DNA.
- Gel run for these 7 samples.
- Gel results: there was the expected ligated insert band in all the colonies, but some of them also have the non ligated insert (very small amount) or the same unexpected band (~1100 bp). None of these colonies was pure...We will discuss this result in the next days. If someone have suggestions about this problem, please e-mail lorenzo.pasotti@unipv.it . Thank you!
July, 23rd
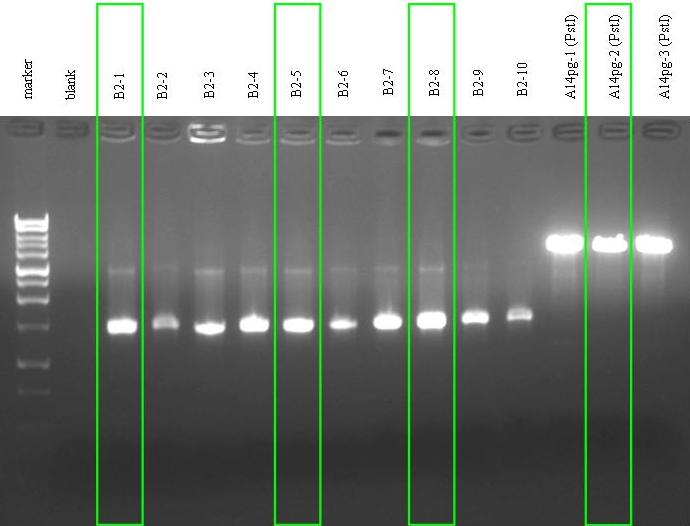
- Colony PCR for B2. The screened colonies were inoculated in 1 ml of LB + Amp and incubated at 37°C, 220 rpm waiting for the end of the PCR.
- We also linearized three samples from A14pg previously extracted DNA, cutting in PstI. We chose to perform this test to check if the unexpected extra band was due to double or single digestion.
- Electrophoresis for B2 PCR results and for the linearized samples of A14pg.
- Gel results:
- All the screened colonies from B2 had the insert with expected length (~1450 bp). Anyway, in this gel the 250 pb band in the ladder was lost and so we do not know if the ten colonies were pure or not. We decided to keep colonies 1, 2, 3, 5, 7 and 8 for further screening. We prepared a glycerol stock for them.
- The first and the third A14pg DNA sample had the unexpected extra band (~1100 bp), while the second seemed not to have it...we still don't know what it is. Moreover, all the three samples had the unwanted band (difficult to notice, because it was near the brightest one). We decided to keep the second DNA sample and to test TOP10 bearing this plasmid.
- We re-filled the remaining 250 ul of B2-1, B2-5 and B2-8 bacterial culture with 5 ml of LB + Amp to grow an overnight culture.
- We also inoculated 21 colonies from B1 plate in 5 ml of LB + Amp. Tomorrow they will be miniprepped and screened! we chose such a high number of colonies because the ligation of B1 had the simultaneous presence of our wanted insert and other digestion products due to an extra PstI site in the vector provided by Mr Gene.
- We incubated the 24 cultures at 37°C, 220 rpm overnight.
- We sent A11-1 purified DNA (stored at -20°C) to BMR Genomics for sequencing.
July, 24th
- Miniprep for the 24 overnight cultures.
- Digestion E-P for all the 21 B1 samples.
- Digestion S-P for B2-1, B2-5, B2-8 samples.
- Electrophoresis for digestion products (two medium-size 1% gels).
- Gel results:
- B1 - positive colonies containing pdc gene: 1, 2, 4, 9, 10, 11, 13, 14, 18, 19, 20. They will be screened to check for non ligated plasmid presence.
- B2 - all the three samples showed the expected length for plasmid+insert, but also the expected length for non ligated plasmid...
- We received TWO stabs for F2620 from iGEM HQ.
 Previous Week
Previous Week
|
Next Week

|
 "
"