Formal Lab Session - 12th August 2009

The Stochastic Switch team and the Chassis Team discuss ideas with Neil
Metal Sensing Team
Recap
In the last session the Metal Sensing team attempted to transform B. subtilis with pGFP-rrnB plasmid (an integration vector). We carried out the procedure and plated the 'transformants' onto 4 plates:
- LB alone - (MM competence medium MINUS B. subtilis)
- LB + Chloramphenicol - (Bacillus subtilis + water (NO DNA))
- LB + Chloramphenicol - 50ul of B. subtilis + pGFP-rrnB
- LB + Chloramphenicol - 200ul of B. subtilis + pGFP-rrnB
Today, the Metal Sensing team will assess the plates and judge whether the transformations have worked well.
Observations
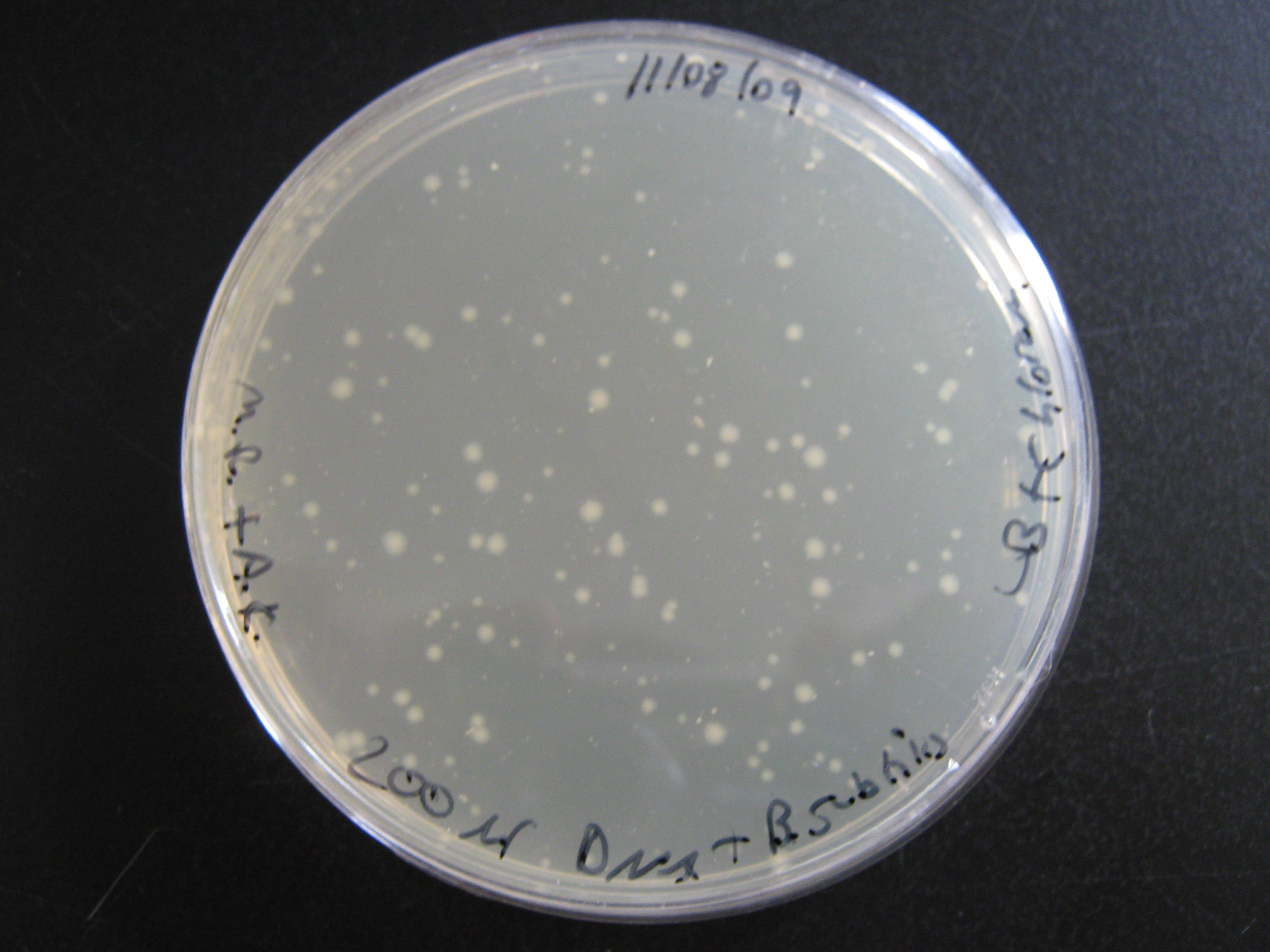
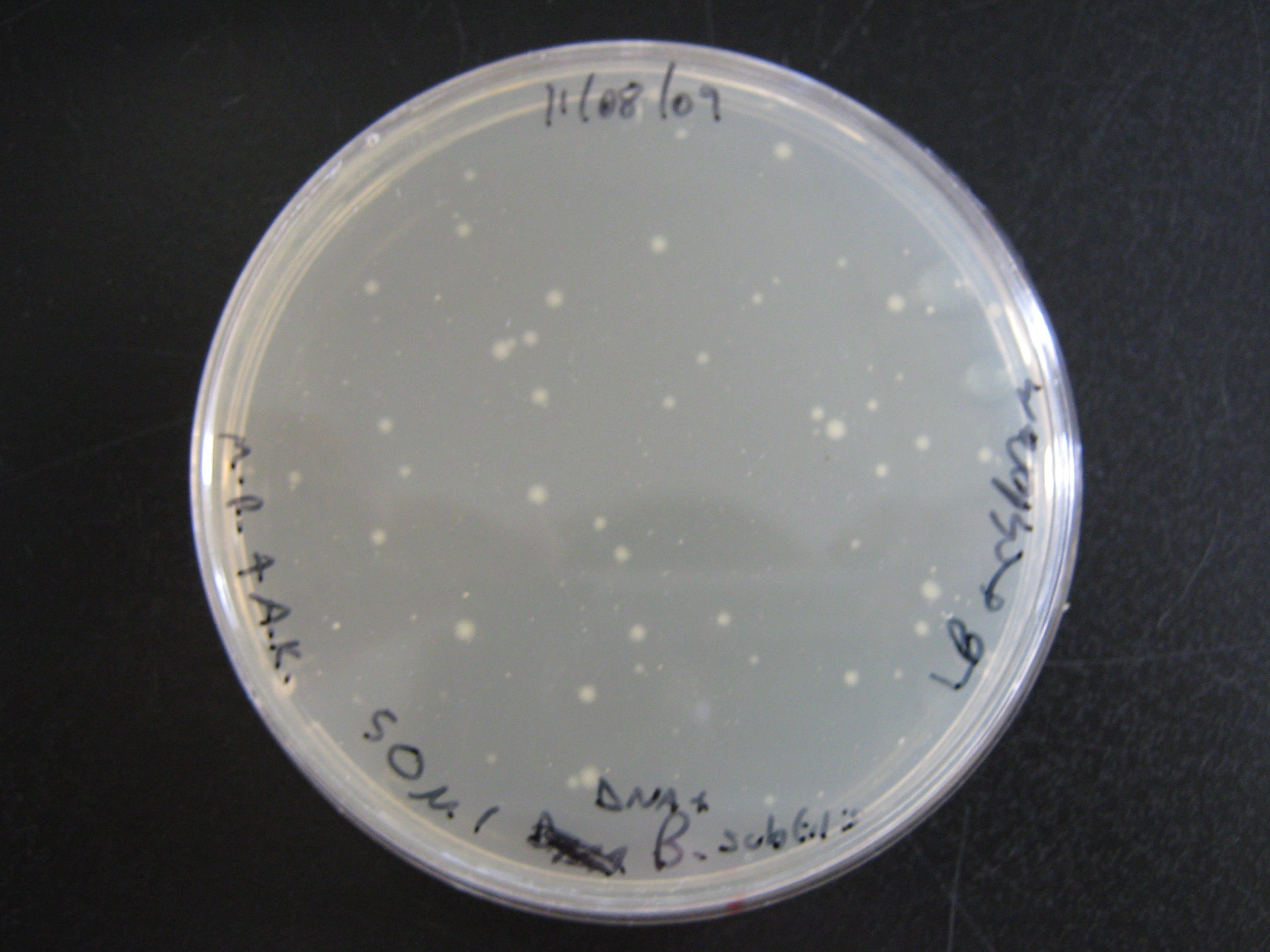
No colonies grew on the LB alone plate or the LB Chloramphenicol plate with B. subtilis lacking pGFP-rrnB. However there was considerable growth on both of the LB + Chloramphenicol plates which contained pGFP-rrnB E. coli transformants - more colonies could be seen in the 200ul portion than on the 50ul portion but that is simply down to concentration of cells. Both transformations were a success, as can be seen in the above photograph.
Stochastic Switch Team

Goksel and Jess are all prepared for the
Bacillus subtilis transformation procedure with these solutions made up
Today we are trying to transform Bacillus subtilis with gfp-rrnb integration vector. Metal sensor team kindly inoculated B. subtilis cells into flask tubes and placed them into the shaking incubator. We labelled them as 1,2,3 and control.
We prepared four plates
- LB + Bsubtilis (+ Control, cells should grow on this plate)
- LB + CHL + B. subtilis (- control, cells will not grow on this plate)
- LB + CHL + B. subtilis + plasmid DNA(Cells should transform with the DNA)
- LB + CHL + B. subtilis + diluted plasmid DNA (Cells should transform with the DNA)
- 2xcompetence medium and 2xstravation medium were prepared.
- We used the 2nd and the 3rd tubes for the tests
- Incubated the samplesfor three hours at 37C
- Added the starvation medium and incubated for another 2 hours
- For each of the two cultures, added 0.4ml of the final solution from the culture and 10ul of DNA into an eppendorf tube.
- Placed the tubes in the shaking incubator for an hour. To do this we taped the tubes to the base of the incubator. N.B- Chris in the lab swapped the temperatures of the incubators, so we used the one that is opposite the -80 freezer. The starter overnight cultures are also in this incubator -these are labelled 1,2,3 control (we used 2 and 3).
- The cultures were plated out using glass beads and put in the 37 incubator.
Sporulation Tuning/Chassis Team
Today, we made our first attempt to recover the cwlD spores which were kindly sent to us by Anne Moir from Sheffield University as mentioned on the 4th of August.

Jane pipetting the cwlD spores out of the eppendorf tubes
We used the protocol for Method A as mentioned.
Preparation
The Lysozyme Stock Solution was made on the 7th of August.
L-alanine
We need:
10mM L-alanine
0.01M L-alanine
We made 0.03L (30ml) of 0.05M L-alanine which we filter sterilised.



The amount of powdered L-alanine needed to make 30ml of 0.05M L-alanine solution is:
MW * Desired Volume (L) * Desired Molarity (M)
89.09* 0.03L * 0.05M = 0.134g
Our final solution volume inclusive of L-alanine is desired to be 1ml.
Therefore, the amount of L-alanine stock solution to add, such that the final concentration of L-alanine is 0.01M is:
0.05M (Stock solution) / 0.01M (Desired Molarity) = 5
In order to obtain a solution with a final concentration of 0.01M,
the stock solution needs to be diluted 5 times.
Desired volume = 1ml
1000ul / 5 = 200ul
200ul of L-alanine stock solution should be added to the buffer and lysozyme solution to make up approximately 1ml.
Final Volume = 1ml
Volume of buffer solution = 1000ul - 200ul = 800ul
Since 1ml of buffer solution requires 40ul of lysozyme stock solution,
800ul / 1000ul = 0.8
0.8 * 40ul = 32ul
Therefore, 800ul of buffer solution requires 32ul of lysozyme stock solution.
- Note
- Due to previous calculation errors, only 4ul of stock lysozyme was added to a ml of buffer solution. Instead, 40ml of stock lysozyme should have been added. As lysozyme is added to disrupt the bacterial cell wall, 4ul may not have been sufficient.
The experiment was performed again on Monday, 17th August. See Monday, 17th August for more details.
Protocol
In addition to the protocol for Method A, it is important to take note of the following points:
- 10ul of cwlD spores were added into the lysozyme and buffer solution.
- After the addition of L-alanine to the solution, which would supposedly initiate germination, the solution was left in the incubator for 10 minutes
- After 10 minutes, the eppendorf tube containing the solution was spinned down for approximately 1 minute.
- Note: Fill another eppendorf tube with water and put it on the opposite site to balance out the weight.
- The supernatant was removed from the eppendorf tube and the spores were resuspended in 1000ul of LB solution.
- Note
- The team faced difficulties in resuspending the spores.
- A serial dilution was performed as illustrated below, and 50ul of each solution was plated out using glass beads on LB + Cm plates.
- In addition, 5ul of the cwlD spores were plated out using glass beads on LB + Cm plates. No colonies should grow on this plate as the spores were not treated.
Results
| July
|
| M | T | W | T | F | S | S
|
|
|
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/1_July_2009&action=edit 1]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/2_July_2009&action=edit 2]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/3_July_2009&action=edit 3]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/4_July_2009&action=edit 4]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/5_July_2009&action=edit 5]
|
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/6_July_2009&action=edit 6]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/7_July_2009&action=edit 7]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/8_July_2009&action=edit 8]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/9_July_2009&action=edit 9]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/10_July_2009&action=edit 10]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/11_July_2009&action=edit 11]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/12_July_2009&action=edit 12]
|
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/13_July_2009&action=edit 13]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/14_July_2009&action=edit 14]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/15_July_2009&action=edit 15]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/16_July_2009&action=edit 16]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/17_July_2009&action=edit 17]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/18_July_2009&action=edit 18]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/19_July_2009&action=edit 19]
|
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/20_July_2009&action=edit 20]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/21_July_2009&action=edit 21]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/22_July_2009&action=edit 22]
| [http://2009.igem.org/Team:Newcastle/Labwork/23_July_2009 23]
| [http://2009.igem.org/Team:Newcastle/Labwork/24_July_2009 24]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/25_July_2009&action=edit 25]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/26_July_2009&action=edit 26]
|
| [http://2009.igem.org/Team:Newcastle/Labwork/27_July_2009 27]
| [http://2009.igem.org/Team:Newcastle/Labwork/28_July_2009 28]
| [http://2009.igem.org/Team:Newcastle/Labwork/29_July_2009 29]
| [http://2009.igem.org/Team:Newcastle/Labwork/30_July_2009 30]
| [http://2009.igem.org/Team:Newcastle/Labwork/31_July_2009 31]
|
|
| August
|
| M | T | W | T | F | S | S
|
|
|
|
|
|
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/1_August_2009&action=edit 1]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/2_August_2009&action=edit 2]
|
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/3_August_2009&action=edit 3]
| [http://2009.igem.org/Team:Newcastle/Labwork/4_August_2009 4]
| [http://2009.igem.org/Team:Newcastle/Labwork/5_August_2009 5]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/6_August_2009&action=edit 6]
| [http://2009.igem.org/Team:Newcastle/Labwork/7_August_2009 7]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/8_August_2009&action=edit 8]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/9_August_2009&action=edit 9]
|
| [http://2009.igem.org/Team:Newcastle/Labwork/10_August_2009 10]
| [http://2009.igem.org/Team:Newcastle/Labwork/11_August_2009 11]
| [http://2009.igem.org/Team:Newcastle/Labwork/12_August_2009 12]
| [http://2009.igem.org/Team:Newcastle/Labwork/13_August_2009 13]
| [http://2009.igem.org/Team:Newcastle/Labwork/14_August_2009 14]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/15_August_2009&action=edit 15]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/16_August_2009&action=edit 16]
|
| [http://2009.igem.org/Team:Newcastle/Labwork/17_August_2009 17]
| [http://2009.igem.org/Team:Newcastle/Labwork/18_August_2009 18]
| [http://2009.igem.org/Team:Newcastle/Labwork/19_August_2009 19]
| [http://2009.igem.org/Team:Newcastle/Labwork/20_August_2009 20]
| [http://2009.igem.org/Team:Newcastle/Labwork/21_August_2009 21]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/22_August_2009&action=edit 22]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/23_August_2009&action=edit 23]
|
| [http://2009.igem.org/Team:Newcastle/Labwork/24_August_2009 24]
| [http://2009.igem.org/Team:Newcastle/Labwork/25_August_2009 25]
| [http://2009.igem.org/Team:Newcastle/Labwork/26_August_2009 26]
| [http://2009.igem.org/Team:Newcastle/Labwork/27_August_2009 27]
| [http://2009.igem.org/Team:Newcastle/Labwork/28_August_2009 28]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/29_August_2009&action=edit 29]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/30_August_2009&action=edit 30]
|
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/31_August_2009&action=edit 31]
|
|
| September
|
| M | T | W | T | F | S | S
|
|
| [http://2009.igem.org/Team:Newcastle/Labwork/1_September_2009 1]
| [http://2009.igem.org/Team:Newcastle/Labwork/2_September_2009 2]
| [http://2009.igem.org/Team:Newcastle/Labwork/3_September_2009 3]
| [http://2009.igem.org/Team:Newcastle/Labwork/4_September_2009 4]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/5_September_2009&action=edit 5]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/6_September_2009&action=edit 6]
|
| [http://2009.igem.org/Team:Newcastle/Labwork/7_September_2009 7]
| [http://2009.igem.org/Team:Newcastle/Labwork/8_September_2009 8]
| [http://2009.igem.org/Team:Newcastle/Labwork/9_September_2009 9]
| [http://2009.igem.org/Team:Newcastle/Labwork/10_September_2009 10]
| [http://2009.igem.org/Team:Newcastle/Labwork/11_September_2009 11]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/12_September_2009&action=edit 12]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/13_September_2009&action=edit 13]
|
| [http://2009.igem.org/Team:Newcastle/Labwork/14_September_2009 14]
| [http://2009.igem.org/Team:Newcastle/Labwork/15_September_2009 15]
| [http://2009.igem.org/Team:Newcastle/Labwork/16_September_2009 16]
| [http://2009.igem.org/Team:Newcastle/Labwork/17_September_2009 17]
| [http://2009.igem.org/Team:Newcastle/Labwork/18_September_2009 18]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/19_September_2009&action=edit 19]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/20_September_2009&action=edit 20]
|
| [http://2009.igem.org/Team:Newcastle/Labwork/21_September_2009 21]
| [http://2009.igem.org/Team:Newcastle/Labwork/22_September_2009 22]
| [http://2009.igem.org/Team:Newcastle/Labwork/23_September_2009 23]
| [http://2009.igem.org/Team:Newcastle/Labwork/24_September_2009 24]
| [http://2009.igem.org/Team:Newcastle/Labwork/25_September_2009 25]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/26_September_2009&action=edit 26]
| [http://2009.igem.org/wiki/index.php?title=Team:Newcastle/Labwork/27_September_2009&action=edit 27]
|
| [http://2009.igem.org/Team:Newcastle/Labwork/28_September_2009 28]
| [http://2009.igem.org/Team:Newcastle/Labwork/29_September_2009 29]
| [http://2009.igem.org/Team:Newcastle/Labwork/30_September_2009 30]
|
|


 "
"






