Team:PKU Beijing/Parts/Parts improvement
From 2009.igem.org
| (17 intermediate revisions not shown) | |||
| Line 2: | Line 2: | ||
{{PKU_Beijing/Sidebar_Parts}} | {{PKU_Beijing/Sidebar_Parts}} | ||
{{PKU_Beijing/Header2}} | {{PKU_Beijing/Header2}} | ||
| + | [[Team:PKU_Beijing/Parts|Parts]] > [[Team:PKU_Beijing/Parts/Parts_improvement|Parts improvement]] | ||
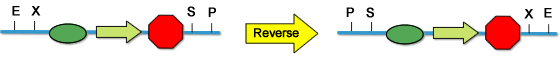
==='''The Reversed Standard'''=== | ==='''The Reversed Standard'''=== | ||
| - | ===''' | + | Standard assembly employs four reistriction sites, EcoRI, XbaI, SpeI and PstI. EcoRI and XbaI is used as the prefix, and SpeI PstI the suffix. |
| + | |||
| + | What if the prefix and suffix are reversed? At the first sight, it may seem stupid to do this. | ||
| + | However, it is a quite useful strategy to avoid interference in your synthetic system. We have, in our project, used this method to construct the sensor part. The activator (repressor) expression set is in an opposite direction of the inducible promoter. This avoids the constitutive expressing promoter (repressor) leaking down. | ||
| + | |||
| + | [[Image:PKU_Reverse.png|500px|center]] | ||
| + | |||
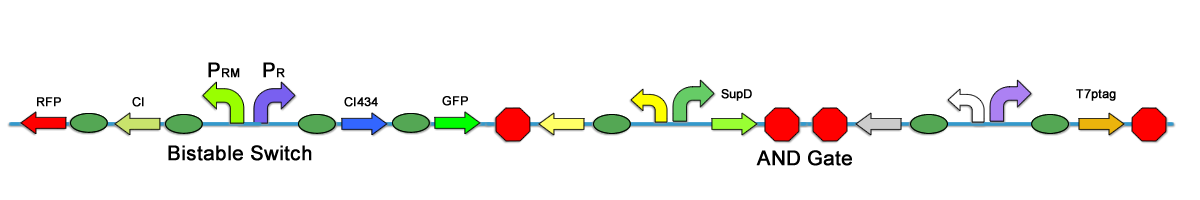
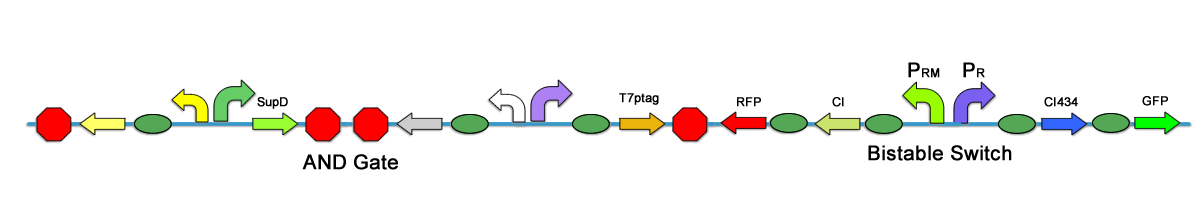
| + | One may introduce transcription terminator to avoid interference between modules, however, in our experiment, we found that terminator cannot completely shut down the leakage. There are still significant interference between modules. In our first stage assembly work, one of the strategies is to place the bistable switch on the same plasmid of the AND Gate, there are two submethods, one is to put the bistable upstream of the AND Gate, and the other is to place it downstream. The 2 methods leads to different results. | ||
| + | |||
| + | [[Image:PKU_Qianjie.png|600px|center|thumb|Fig1. Bistable switch upstream of the AND Gate]] | ||
| + | [[Image:PKU_assemble_SS.png|600px|center|thumb|Fig2. Result of Bistable switch upstream of the AND Gate, left to right is the not induced group and induced group]] | ||
| + | |||
| + | [[Image:PKU_Houjie.png|600px|center|thumb|Fig3. Bistable switch downstream of the AND Gate]] | ||
| + | [[Image:PKU_XP1_No_Yes.jpg|600px|center|thumb|Fig4. Result of Bistable switch downstream of the AND Gate, left to right is the not induced group and induced group]] | ||
| + | |||
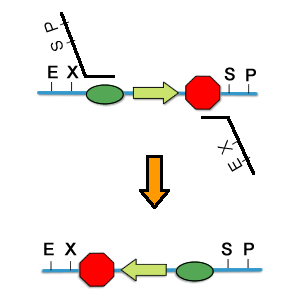
| + | [[Image:PKU_PCR_Reverse.png|400px|left| thumb|fig5. PCR to reverse a part]] | ||
| + | |||
| + | We explain the phenomenon as below: | ||
| + | |||
| + | The terminator is to some extent leaky, so when bistable switch is upstream, it can increase the expression level of both T7ptag and SupD, so that the AND Gate outputs more to trigger the bistable switch. Thus the colonies turn read when induced. In addition, the constitutive promoter that expresses the activator can also increase the CI level. However, when the bistable switch is downstream, it is the promoters in the AND Gate can leak downstream and enhance the CI434 state, while the bistable has little effect on the AND Gate. | ||
| + | |||
| + | With the reversed standard, we can arrange the transcription direction of each part, which, in simple systems, can totally avoid interference. For complex systems like ours, it can also help out.For complex systems like ours, it can also help out by reducing the interferences that we should consider. | ||
| + | |||
| + | Parts can be reversed by PCR. As shown in Fig5. Actually, for basic parts, we can assembly them in the normal way, and it is after the completion of one expression set that we consider to reverse the whole costruct. | ||
| + | |||
| + | ==='''The Blue White Screening Plasmid'''=== | ||
| + | |||
| + | ====Introduction==== | ||
| + | |||
| + | The blue-white screen is a molecular technique that allows for the detection of successful ligations in vector-based gene cloning in which DNA fragment of interest is ligated into a vector. The vector has a short portion of the β-galactosidase gene (the alpha fragment) whose synthesis can be induced by IPTG. This alpha fragment is capable of intra-allelic complementation with a defective form of β-galactosidase called omega fragment encoded by host chromosome. In the presence of IPTG in growth medium, bacteria synthesize both fragments of the enzyme. Both the fragments can assemble to form a functional protein, which degrades X-gal and turns the bacterial colony blue on media with X-gal. | ||
| + | |||
| + | Insertion of foreign DNA into the lac Z alpha fragment gene on the vector causes insertional inactivation of this gene abolishing intra-allelic complementation. Thus bacteria carrying recombinant plasmids can’t hydrolyse X-gal, giving rise to white colonies, which can be distinguished on culture media from non-recombinant cells, which are blue. | ||
| + | |||
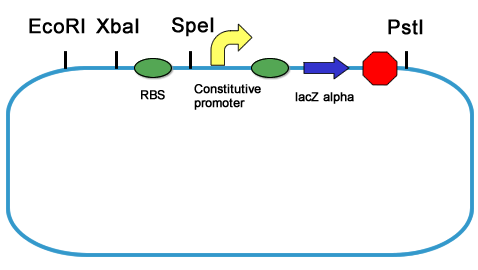
| + | Each biobrick is commonly flanked on the upstream end by EcoRI and XbaI restriction sites and on the downstream end by SpeI and PstI restriction sites. In normal cases, gaps between EcoRI and XbaI, or SpeI and PstI restriction sites just contain NotI restriction site. Some biobrick parts, such as promoter parts, rbs parts and terminator parts are too small to be the insertion fragment in standard assembly because of the Gel purification failure. To assemble these parts, we usually cut them with EcoRI XbaI or SpeI PstI to make vectors, and cut large fragments as inserts. Assessment is needed because the incomplete digestion of the vector creates false positive. In order to save the assessment time, we designed the following strategy to introduce blue white selection to standard assembly. | ||
| + | |||
| + | ====Design==== | ||
| + | |||
| + | The Design is simply shown in fig1. | ||
| + | |||
| + | [[Image: PKU_Blue_White.png]] | ||
| + | |||
| + | The lacZ alpha fragment, which is from standard part BBa_I732006, performs intra-allelic complementation with the omega fragment encoded by host chromosome to restore β-galactosidase activity. | ||
| + | |||
| + | In standard assembly, we cut the plasmid with an RBS part with SpeI and PstI, so that the lacZ constitutive generator is removed. And the coding sequence to be placed downstream is inserted to between the SpeI and PstI site. Although there will be religation of the lacZ constitutive generator fragment, the false positive colonies can be distinguished by its color when plated on an X-gal plate. We can accelerate the cloning process by saving the assessment time. | ||
| + | |||
| + | An similar construct is the constitutive promoter family by Berkley in 2006. They have an RBS-RFP part placed in between the SpeI and PstI site, so that the constitutive promoter can drive the expression of the RFP. J61002. They explained the function of this plasmid as 1. Insertion of a promoter element between XbaI and SpeI sites results in a RFP reporter while retaining the ability to do biobrick assembly. 2. Insertion of a protein generating device or RNA gene (cutting the part with XbaI/PstI, inserting into SpeI/PstI of J61002) results in a standard pSB1A2 plasmid containing the constitutive promoter and its downstream parts. In point 2, the correct colonies and the false positive can be differentiated by its color, red colonies are false positive ones. | ||
| + | |||
| + | However, there are limits: 1. The rbs-RFP part is so large that gel purification should be carried out to get the vector. 2. The rbs – RFP can express only when placed downstream of a constitutive promoter. In our design, however, lacZ is a small fragment with a length of only 200bp, we can purify the vector simply by purification without gel electrophoresis (200bp fragment gets lower purification ratio). Also a constitutive promoter is packed with the lacZ-alpha fragment, ensuring the expression of the lacZ-alpha gene no matter where it is placed. Two types of basic parts can be moved to this improved vector, the promoter part, the rbs part, which is too small to be an insert. To the terminator part, the constitutive generator of lacZ should be placed in between EcoRI and XbaI site. | ||
| - | |||
{{PKU_Beijing/Foot}} | {{PKU_Beijing/Foot}} | ||
__NOTOC__ | __NOTOC__ | ||
Latest revision as of 13:50, 21 October 2009
|
||||||||||||||
|
||||||||||||||
 "
"