Team:Cambridge/Project/CA03
From 2009.igem.org
(→Quantitative measurement of pigment production) |
(→In vivo expression of our devices in E.coli MG1655) |
||
| (9 intermediate revisions not shown) | |||
| Line 5: | Line 5: | ||
<!-- This is for the top grey / blue links bar !--> | <!-- This is for the top grey / blue links bar !--> | ||
{{Template:Cambridgetemplatetop}} | {{Template:Cambridgetemplatetop}} | ||
| - | [https://2009.igem.org/Team:Cambridge/Project/ | + | [https://2009.igem.org/Team:Cambridge/Project/CA01 Background] |
| - | [https://2009.igem.org/Team:Cambridge/Project/ | + | [https://2009.igem.org/Team:Cambridge/Project/CA02 Design] |
| - | [https://2009.igem.org/Team:Cambridge/Project/ | + | [https://2009.igem.org/Team:Cambridge/Project/CA03 Characterisation] |
| - | [https://2009.igem.org/Team:Cambridge/Project/ | + | [https://2009.igem.org/Team:Cambridge/Project/CA05 Datasheet] |
| - | [https://2009.igem.org/Team:Cambridge/Project/ | + | [https://2009.igem.org/Team:Cambridge/Project/CA04 Reference] |
{{Template:Cambridgetemplatebottom}} | {{Template:Cambridgetemplatebottom}} | ||
| - | == | + | == Testing and Characterising == |
=== In vivo expression of our devices in ''E.coli'' MG1655 === | === In vivo expression of our devices in ''E.coli'' MG1655 === | ||
Our preliminary research indicated that carotenoids production in ''E.coli'' TOP10 was very weak. We decided to use another strain, ''E.coli'' MG1655, for in vivo expression of our devices. The amount of carotenoids produced became visibly higher. Generally, the colour became visible after incubation for about 12 hours. | Our preliminary research indicated that carotenoids production in ''E.coli'' TOP10 was very weak. We decided to use another strain, ''E.coli'' MG1655, for in vivo expression of our devices. The amount of carotenoids produced became visibly higher. Generally, the colour became visible after incubation for about 12 hours. | ||
| Line 19: | Line 19: | ||
'''''Lycopene-producing device (constitutive)''''' | '''''Lycopene-producing device (constitutive)''''' | ||
{| style="color:#333; background-color:#FFFFFF;" cellpadding="6" cellspacing="0" border="1" | {| style="color:#333; background-color:#FFFFFF;" cellpadding="6" cellspacing="0" border="1" | ||
| - | |[[Image:Cam09_lyco_1.jpg]] | + | |[[Image:Cam09_lyco_1.jpg|400px]] |
| - | |[[Image:Cam09_lyco_2.jpg]] | + | |[[Image:Cam09_lyco_0.jpg]] |
| + | |||
| + | [[Image:Cam09_lyco_2.jpg]] | ||
|- style="color:#333; background-color:#FFFFFF;" cellpadding="6" cellspacing="0" border="1" | |- style="color:#333; background-color:#FFFFFF;" cellpadding="6" cellspacing="0" border="1" | ||
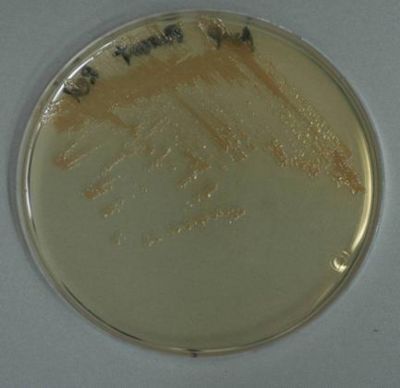
|''E.coli'' MG1655 transformed with K274110 and grown on agar plate overnight. | |''E.coli'' MG1655 transformed with K274110 and grown on agar plate overnight. | ||
| - | |Cell pellet of ''E.coli'' MG1655 transformed with K274210 (from | + | |Top: single colonies (enlarged). Bottom: Cell pellet of ''E.coli'' MG1655 transformed with K274210 (from 200mL LB culture at 37°C for 24 hours). |
|- style="color:#333; background-color:#FFFFFF;" cellpadding="6" cellspacing="0" border="1" | |- style="color:#333; background-color:#FFFFFF;" cellpadding="6" cellspacing="0" border="1" | ||
|} | |} | ||
| Line 31: | Line 33: | ||
{| style="color:#333; background-color:#FFFFFF;" cellpadding="6" cellspacing="0" border="1" | {| style="color:#333; background-color:#FFFFFF;" cellpadding="6" cellspacing="0" border="1" | ||
|[[Image:Cam09_caro_1.jpg]] | |[[Image:Cam09_caro_1.jpg]] | ||
| - | |[[Image:Cam09_caro_2.jpg]] | + | |[[Image:Cam09_caro_0.jpg]] |
| + | |||
| + | [[Image:Cam09_caro_2.jpg]] | ||
|- style="color:#333; background-color:#FFFFFF;" cellpadding="6" cellspacing="0" border="1" | |- style="color:#333; background-color:#FFFFFF;" cellpadding="6" cellspacing="0" border="1" | ||
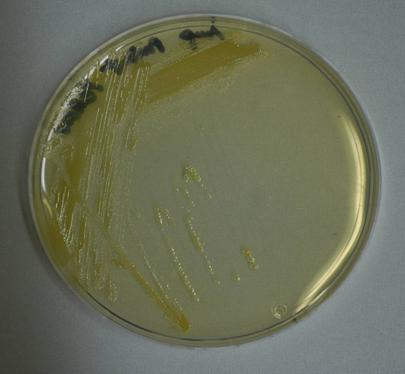
|''E.coli'' MG1655 transformed with K274210 and grown on agar plate overnight. | |''E.coli'' MG1655 transformed with K274210 and grown on agar plate overnight. | ||
| - | |Cell pellet of ''E.coli'' MG1655 transformed with K274220 (from | + | |Top: single colonies (enlarged). Bottom: Cell pellet of ''E.coli'' MG1655 transformed with K274220 (from 200mL LB culture at 37°C for 24 hours). |
|- style="color:#333; background-color:#FFFFFF;" cellpadding="6" cellspacing="0" border="1" | |- style="color:#333; background-color:#FFFFFF;" cellpadding="6" cellspacing="0" border="1" | ||
|} | |} | ||
| - | |||
=== Quantitative measurement of pigment production === | === Quantitative measurement of pigment production === | ||
| Line 81: | Line 84: | ||
|- style="color:#333; background-color:#FFFFFF;" cellpadding="6" cellspacing="0" border="1" | |- style="color:#333; background-color:#FFFFFF;" cellpadding="6" cellspacing="0" border="1" | ||
|} | |} | ||
| + | |||
'''Rough Estimation''' | '''Rough Estimation''' | ||
Latest revision as of 23:16, 21 October 2009
Categories :
Project :
-
Overview
Sensitivity Tuner
--- Characterisation
--- Modelling
Colour Generators
--- Carotenoids (Orange/Red)
--- Melanin (Brown)
--- Violacein (Purple/Green)
The Future
Safety
Notebook :
Team Logistics :
Carotenoids
Testing and Characterising
In vivo expression of our devices in E.coli MG1655
Our preliminary research indicated that carotenoids production in E.coli TOP10 was very weak. We decided to use another strain, E.coli MG1655, for in vivo expression of our devices. The amount of carotenoids produced became visibly higher. Generally, the colour became visible after incubation for about 12 hours.
Lycopene-producing device (constitutive)
β -carotene-producing device (constitutive)
Quantitative measurement of pigment production
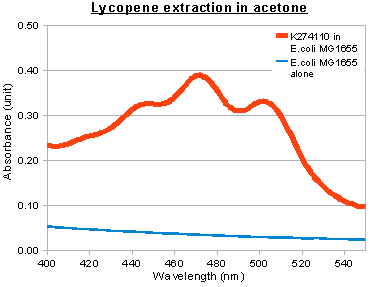
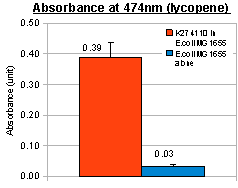
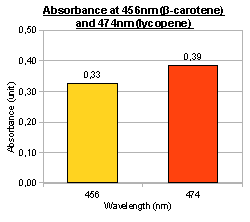
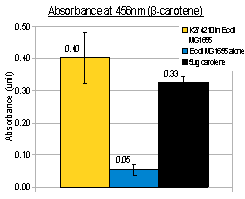
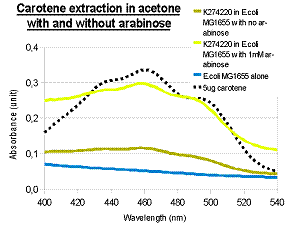
In addition to visual inspection of coloured colonies, we hope to quantitatively measure the amount of lycopene or β-carotene produced by our devices. We grew transformed E.coli MG1655 in LB culture at 37°C for 24 hours and collected cell pellet by centrifugation. Acetone was added to the cell pellet and warmed at 50°C for 10 minutes, allowing carotenoids (lycopene or β-carotene) in the cells to dissolve. The acetone extracts were then put into MicroPlate Reader for a full photospectrum scan (wavelengths between 300nm and 800nm). The absorbance curves showed peaks characteristics of the respective carotenoids: at wavelength 474nm for lycopene, and 456nm for β-carotene. In the case of β-carotene, we used pure β-carotene of known amount as the standard reference and included it in the graph below.
Lycopene-producing device (constitutive)
β -carotene-producing device (constitutive)
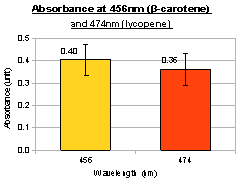
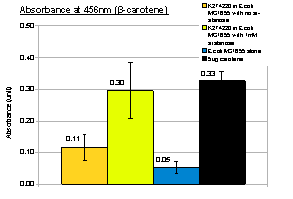
To investigate the induction of pigment production, we tested our device K274220 in E.coli MG1655 and 1mM arabinose was added to LB culture. Cell culture that was induced with arabinose showed greater β-carotene production than cell culture without arabinose, indicating the feasibility of inducing pigment production. Using known amount of pure β-carotene to construct a standard curve, we were able to estimate the amount of β-carotene produced per unit volume of cell culture by our device.
Rough Estimation
| Samples | Absorbance (unit) | Carotene (μg per 100μL Acetone extract) | Total mass of carotene (μg per mL-culture) |
|---|---|---|---|
| K274210 in E.coli MG1655 | 0.4 | ~6 | 6*5/20=1.5 |
| K274220, with 1mM arabinose in E.coli MG1655 | 0.3 | ~5 | 5*5/20=1.25 |
Discussion of results
Despite relatively poor performance in E.coli TOP10, our pigment-producing devices gave encouraging results in E.coli MG1655. Simply by overnight incubation, the colonies were able to show visible colour changes. Since the precursors were already present in normal E.coli, no additional growth medium or special treatment was needed. This paves the way to developing a signalling system that is quick to produce visual outputs.
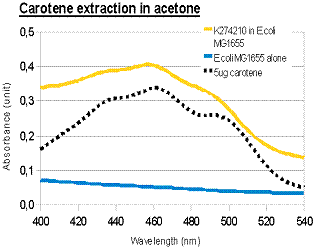
The acetone extraction of carotenoid provides a low-cost method for quantitative measurement. In the above experiments, the results of photo-spectrometry were consistent with characteristic absorbance wavelengths of lycopene (around 475nm) and β-carotene (around 450nm), as noted in literature. In the case of β-carotene, the absorbance profile of our β-carotene-producing device (K274210) matched that of pure carotene standards, confirming that our device was functional in the cells.
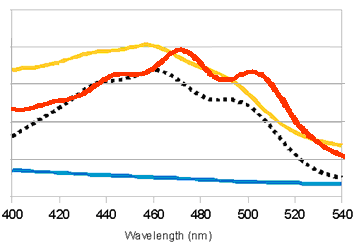
When we overlay the graphs of our two devices (constitutive), we obtained the following result:
red: K274110/lycopene; orange: K274210/β-carotene; black: pure β-carotene; blue: untransformed E.coli MG1655 (negative control).
Lycopene and β-carotene showed characteristic absorbance profiles, with different maximum absorbance wavelengths. It was tempted to think that by converting lycopene to β-carotene (e.g. by expressing CrtY gene), we would be able to see a “shift” in the peaks of maximum absorbance. However, in the bar charts above it was noted that lycopene and β-carotene had significant absorbance at both 456nm and 474nm, albeit with different ratios (i.e. lycopene absorbed more at 474nm; β-carotene absorbed more at 456nm). Therefore, measurement at a single wavelength was insufficient to determine the identity of the carotenoid; we might need to use “456nm:474nm” ratio instead. It would be interesting to see the effect of mixing different amounts of lycopene and β-carotene, and whether the “456nm:474nm” ratio can indicate the relative percentage of lycopene and β-carotene. In the current project, we were unable to obtain pure lycopene due to high cost of purchase.
In terms of inducing β-carotene production, 1mM arabinose, acting on Pbad promoter, was able to produce a level of synthesis close to that under constitutive promoter. In the non-induced population, there was probably a small degree of “leakiness” and basal level of gene expression (leading to some β-carotene synthesis). In future experiments, it might be helpful to move the pigment-producing devices to low copy plasmids and reduce the effect of such basal expression.
One possible design using our devices is to construct a system where lycopene is constitutively produced and CrtY, which converts red lycopene to orange β-carotene, is controlled by external stimulus. Hence, the red cells will turn orange upon sensing the specific external stimulus, and this change in colour serves as the signal output that can be detected visually and measured quantitatively at low cost.
 "
"