Team:Cambridge/Project/CA01
From 2009.igem.org
MikeDavies (Talk | contribs) (→Carotenoids) |
|||
| (6 intermediate revisions not shown) | |||
| Line 8: | Line 8: | ||
[https://2009.igem.org/Team:Cambridge/Project/CA02 Design] | [https://2009.igem.org/Team:Cambridge/Project/CA02 Design] | ||
[https://2009.igem.org/Team:Cambridge/Project/CA03 Characterisation] | [https://2009.igem.org/Team:Cambridge/Project/CA03 Characterisation] | ||
| + | [https://2009.igem.org/Team:Cambridge/Project/CA05 Datasheet] | ||
| + | [https://2009.igem.org/Team:Cambridge/Project/CA04 Reference] | ||
{{Template:Cambridgetemplatebottom}} | {{Template:Cambridgetemplatebottom}} | ||
| - | == | + | == Background == |
Carotenoids are organic pigments naturally present in plants, algae and some bacteria. There are more than 600 carotenoids, which can be categorised into xanthophylls (hydrocarbons containing oxygen element) and carotenes (hydrocarbons containing no oxygen). Carotenoids perform a range of functions, including light energy absorption, protection against photo-damage, acting as antioxidants, and as precursor to other organic compounds. In human, for example, beta-carotene is the precursor to vitamin A. | Carotenoids are organic pigments naturally present in plants, algae and some bacteria. There are more than 600 carotenoids, which can be categorised into xanthophylls (hydrocarbons containing oxygen element) and carotenes (hydrocarbons containing no oxygen). Carotenoids perform a range of functions, including light energy absorption, protection against photo-damage, acting as antioxidants, and as precursor to other organic compounds. In human, for example, beta-carotene is the precursor to vitamin A. | ||
| Line 17: | Line 19: | ||
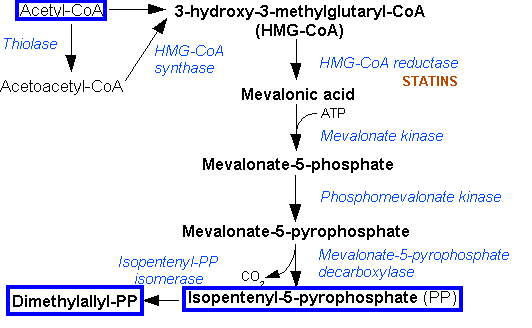
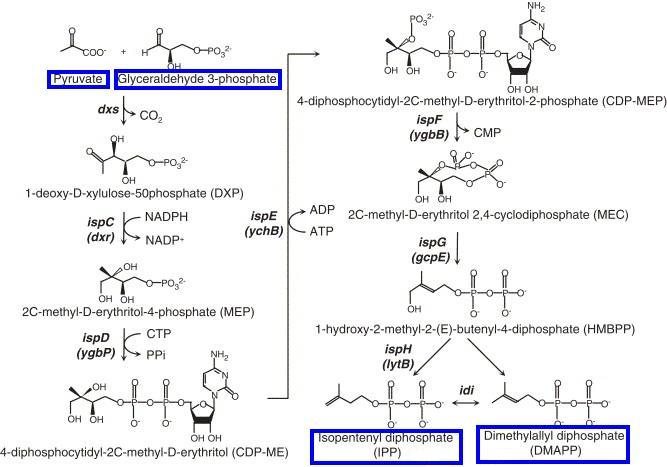
The common starting point for carotenoid synthesis is farnesyl pyrophosphate (FPP), which derives from two precursors, isopentenyl pyrophosphate (IPP) and dimethylallyl pyrophosphate (DMAPP). In general, there are two pathways for synthesising IPP and DMAPP: the Mevalonate Pathway (starting with acetyl CoA) and the Non-mevalonate Pathway (starting with pyruvate and glyceradehyde-3-phosphate). While the Mevalonate Pathway is present in all higher eukaryotes, the Non-mevalonate Pathway is present in ''E.coli''. | The common starting point for carotenoid synthesis is farnesyl pyrophosphate (FPP), which derives from two precursors, isopentenyl pyrophosphate (IPP) and dimethylallyl pyrophosphate (DMAPP). In general, there are two pathways for synthesising IPP and DMAPP: the Mevalonate Pathway (starting with acetyl CoA) and the Non-mevalonate Pathway (starting with pyruvate and glyceradehyde-3-phosphate). While the Mevalonate Pathway is present in all higher eukaryotes, the Non-mevalonate Pathway is present in ''E.coli''. | ||
| - | [[Image:Cam09 Mevalonate pathway.png|334px|thumb|left|'''Mevalonate Pathway''' (image adopted from Wikipedia, http://en.wikipedia.org/wiki/Mevalonate_pathway)]] | + | [[Image:Cam09 Mevalonate pathway.png|334px|thumb|left|'''Mevalonate Pathway''' (image adopted from Wikipedia, http://en.wikipedia.org/wiki/Mevalonate_pathway)]][[Image:Cam09 Non mevalonate.JPG|350px|thumb|right|'''Non-mevalonate Pathway''' (image adopted from Yuan et al. 2006)]] |
| - | [[Image:Cam09 Non mevalonate.JPG| | + | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
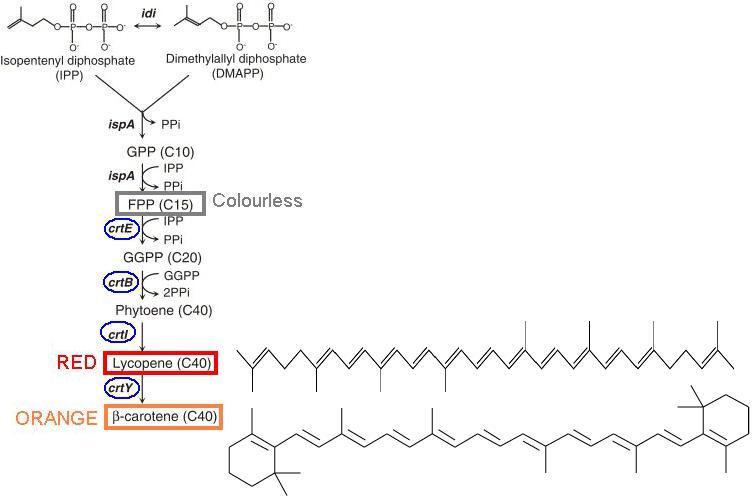
After IPP and DMAPP go on to form FPP, a series of enzymatic reactions convert the colourless FPP to coloured compounds: red lycopene and orange β-carotene. | After IPP and DMAPP go on to form FPP, a series of enzymatic reactions convert the colourless FPP to coloured compounds: red lycopene and orange β-carotene. | ||
| Line 30: | Line 29: | ||
By modulating the enzymes involved in this conversion pathway ("CrtE", "CrtB", "CrtI", and "CrtY"), we aim to produce and control different coloured pigments as signal output. | By modulating the enzymes involved in this conversion pathway ("CrtE", "CrtB", "CrtI", and "CrtY"), we aim to produce and control different coloured pigments as signal output. | ||
| + | |||
| + | |||
| + | {{Template:CambridgeBottom}} | ||
Latest revision as of 01:07, 22 October 2009
Categories :
Project :
-
Overview
Sensitivity Tuner
--- Characterisation
--- Modelling
Colour Generators
--- Carotenoids (Orange/Red)
--- Melanin (Brown)
--- Violacein (Purple/Green)
The Future
Safety
Notebook :
Team Logistics :
Carotenoids
Background
Carotenoids are organic pigments naturally present in plants, algae and some bacteria. There are more than 600 carotenoids, which can be categorised into xanthophylls (hydrocarbons containing oxygen element) and carotenes (hydrocarbons containing no oxygen). Carotenoids perform a range of functions, including light energy absorption, protection against photo-damage, acting as antioxidants, and as precursor to other organic compounds. In human, for example, beta-carotene is the precursor to vitamin A.
Biochemical pathway of carotenoid synthesis
The common starting point for carotenoid synthesis is farnesyl pyrophosphate (FPP), which derives from two precursors, isopentenyl pyrophosphate (IPP) and dimethylallyl pyrophosphate (DMAPP). In general, there are two pathways for synthesising IPP and DMAPP: the Mevalonate Pathway (starting with acetyl CoA) and the Non-mevalonate Pathway (starting with pyruvate and glyceradehyde-3-phosphate). While the Mevalonate Pathway is present in all higher eukaryotes, the Non-mevalonate Pathway is present in E.coli.
After IPP and DMAPP go on to form FPP, a series of enzymatic reactions convert the colourless FPP to coloured compounds: red lycopene and orange β-carotene.
By modulating the enzymes involved in this conversion pathway ("CrtE", "CrtB", "CrtI", and "CrtY"), we aim to produce and control different coloured pigments as signal output.
 "
"