Team:Brown/Project S.epidermidis
From 2009.igem.org
Sscheung918 (Talk | contribs) |
|||
| (16 intermediate revisions not shown) | |||
| Line 6: | Line 6: | ||
| - | + | <html><img src="http://img26.imageshack.us/img26/6156/thechassis.png"></html> | |
| - | + | ||
| - | + | ||
| - | = | + | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
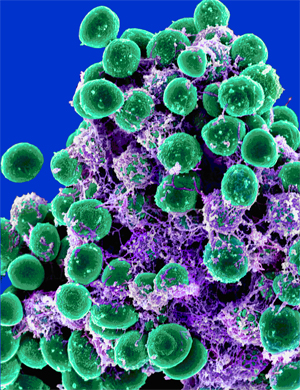
[[Image:Staphylococcus epidermidis.jpg|250 px|thumb|right|Staphylococcus epidermidis biofilm]]Staphylococcus epidermidis is an ideal vehicle for our genetic machine for several reasons: it is a native organism in the human nasal flora, it is generally non-pathogenic, and its sequenced genome is used with some frequency in lab work. | [[Image:Staphylococcus epidermidis.jpg|250 px|thumb|right|Staphylococcus epidermidis biofilm]]Staphylococcus epidermidis is an ideal vehicle for our genetic machine for several reasons: it is a native organism in the human nasal flora, it is generally non-pathogenic, and its sequenced genome is used with some frequency in lab work. | ||
| Line 23: | Line 12: | ||
[[Image:Plasmid_with_HBP+secretion_tag.jpg|200 px|thumb|left|Final construct with secretion tag attached to HBP]] | [[Image:Plasmid_with_HBP+secretion_tag.jpg|200 px|thumb|left|Final construct with secretion tag attached to HBP]] | ||
| - | Since our gene of interest, rEV131, can only bind histamine if it is outside the cell, we have to engineer its secretion from S. epidermidis. We decided to use a signal peptide motif normally found on ß-lactamase, the S. epidermidis gene for ampicillin resistance, which uses the native Sec pathway for its secretion. | + | Since our gene of interest, rEV131, can only bind histamine if it is outside the cell, we have to engineer its secretion from S. epidermidis. We decided to use a signal peptide motif normally found on ß-lactamase, the S. epidermidis gene for ampicillin resistance, which uses the native Sec pathway for its secretion. |
| - | We had the DNA sequence of this peptide synthesized and ligated it N-terminally to GFP as a test construct. This reporter would allow us to visually ascertain | + | We had the DNA sequence of this peptide synthesized by GeneArt AG and ligated it N-terminally to GFP as a test construct (<partinfo>BBa_K212004</partinfo>). This reporter would allow us to visually ascertain |
| - | whether the produced protein was secreted. | + | whether the produced protein was secreted using differential centrifugation. An additional advantage is that GFP is the approximately the same size as rEV131, increasing the likelihood that the two proteins behave similarly in terms of secretion. If the secretion tag works properly, the GFP will be secreted through the Sec pathway into the surrounding environment. If we grow S. epi in solution, the secreted GFP will remain in the solution. We can then use differential centrifugation to separate the cells from whatever proteins are in the solution. Once we have obtained the cell-free supernatant, we will use our fluorospectrophotometer to determine the fluorescence of GFP in the supernatant, which can be compared to the fluorescence of the supernatant from similarly prepared cells expressing GFP without a secretion tag to determine whether the secretion tag is functional. |
| + | In our final genetic construct, the GFP reporter is replaced by rEV131. | ||
| + | =Quorum Sensor= | ||
| - | |||
| + | Although Staphylococcus epidermidis is one of the more benign species of Staphylococcus, it can form infectious biofilms that are impervious to antibiotic treatment if its cell | ||
| + | density becomes too great. This change in phenotype is accomplished by the S. epidermidis agr operon, which upregulates pathogenicity genes in response to a quorum. | ||
| + | We reasoned that we could incorporate a safety mechanism into our bacteria by putting a death gene under the regulation of the agr promoter. This way, whenever the bacteria would reach a high enough density to become dangerous, they would simply begin to die until they reach a safer, lower density. In order to first test that our cassette works, we ligated the agr promoter upstream of GFP, so that whenever the cells reached a quorum, they would fluoresce green. Later, the GFP would be switched out for a CCDB, a DNA gyrase poison <partinfo>BBa_P1010</partinfo>. | ||
| + | Staphylococcus epidermidis is not readily made chemically competent due to its thick cell wall. Most researchers use electroporation to induce cells to take up their target DNA. Throughout the course of our research, we have tweaked the electroporation protocol many times but there are always a very small number of transformants. | ||
| + | [https://2009.igem.org/Team:Brown/Project_Implications Read more about human practices and safety issues here] | ||
| + | ---- | ||
| + | ''<big>References</big>'' | ||
| + | * Götz, Friedrich. "Staphylococcus and Biofilms." Molecular Microbiology 43.6 (2002): 1367-368. Print. | ||
| + | * Lina, Gerard, Florent Boutitte, Anne Tristan, Michele Bes, Jerome Etienne, and Francois Vandenesch. "Bacterial Competition for Human Nasal Cavity Colonization: Role of Staphylococcal agr Alleles." Applied and Environmental Microbiology 69.1 (2003): 18-23. Print. | ||
| - | + | * Novick, Richard P., and Edward Geisinger. "Quorum Sensing in Staphylococci." Annual Review of Genetics 42 (2008): 541-64. Print. | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| + | You, L., R. S. Cox, R. Weiss, and F. H. Arnold. "Programmed population control by cell-cell communication and regulated killing." Nature 428.6985 (2004): 868-71. Print. | ||
| - | + | <html> | |
| - | + | <a href="https://2009.igem.org/Team:Brown/Project_All_Together"> | |
| + | <img src="https://static.igem.org/mediawiki/2009/9/91/Brown_system_schematic_button.png"> | ||
| + | </html> | ||
Latest revision as of 02:56, 22 October 2009

Since our gene of interest, rEV131, can only bind histamine if it is outside the cell, we have to engineer its secretion from S. epidermidis. We decided to use a signal peptide motif normally found on ß-lactamase, the S. epidermidis gene for ampicillin resistance, which uses the native Sec pathway for its secretion. We had the DNA sequence of this peptide synthesized by GeneArt AG and ligated it N-terminally to GFP as a test construct (). This reporter would allow us to visually ascertain whether the produced protein was secreted using differential centrifugation. An additional advantage is that GFP is the approximately the same size as rEV131, increasing the likelihood that the two proteins behave similarly in terms of secretion. If the secretion tag works properly, the GFP will be secreted through the Sec pathway into the surrounding environment. If we grow S. epi in solution, the secreted GFP will remain in the solution. We can then use differential centrifugation to separate the cells from whatever proteins are in the solution. Once we have obtained the cell-free supernatant, we will use our fluorospectrophotometer to determine the fluorescence of GFP in the supernatant, which can be compared to the fluorescence of the supernatant from similarly prepared cells expressing GFP without a secretion tag to determine whether the secretion tag is functional.
In our final genetic construct, the GFP reporter is replaced by rEV131.
Quorum Sensor
Although Staphylococcus epidermidis is one of the more benign species of Staphylococcus, it can form infectious biofilms that are impervious to antibiotic treatment if its cell density becomes too great. This change in phenotype is accomplished by the S. epidermidis agr operon, which upregulates pathogenicity genes in response to a quorum. We reasoned that we could incorporate a safety mechanism into our bacteria by putting a death gene under the regulation of the agr promoter. This way, whenever the bacteria would reach a high enough density to become dangerous, they would simply begin to die until they reach a safer, lower density. In order to first test that our cassette works, we ligated the agr promoter upstream of GFP, so that whenever the cells reached a quorum, they would fluoresce green. Later, the GFP would be switched out for a CCDB, a DNA gyrase poison .
Staphylococcus epidermidis is not readily made chemically competent due to its thick cell wall. Most researchers use electroporation to induce cells to take up their target DNA. Throughout the course of our research, we have tweaked the electroporation protocol many times but there are always a very small number of transformants.
Read more about human practices and safety issues here
References
- Götz, Friedrich. "Staphylococcus and Biofilms." Molecular Microbiology 43.6 (2002): 1367-368. Print.
- Lina, Gerard, Florent Boutitte, Anne Tristan, Michele Bes, Jerome Etienne, and Francois Vandenesch. "Bacterial Competition for Human Nasal Cavity Colonization: Role of Staphylococcal agr Alleles." Applied and Environmental Microbiology 69.1 (2003): 18-23. Print.
- Novick, Richard P., and Edward Geisinger. "Quorum Sensing in Staphylococci." Annual Review of Genetics 42 (2008): 541-64. Print.
You, L., R. S. Cox, R. Weiss, and F. H. Arnold. "Programmed population control by cell-cell communication and regulated killing." Nature 428.6985 (2004): 868-71. Print.
 "
"