Team:Cambridge/Notebook/Week7
From 2009.igem.org
(→Dry Work) |
MikeDavies (Talk | contribs) (→Tuesday) |
||
| (30 intermediate revisions not shown) | |||
| Line 9: | Line 9: | ||
===Wet Work=== | ===Wet Work=== | ||
| - | ==== | + | ====Threshold Device==== |
Colony PCR of 8 colonies picked from 391 in pSB3K3 transformants, 8 colonies picked from 374 in pSB3K3 transformants, 8 colonies picked from 384 in pSB3K3, and 1 colony picked from 381 in pSB3K3 transformants confirmed to contain an insert of the right length. Overnight cultures of each of those colonies as well. No 374 constructs were found to contain inserts of the right length, however 4 384 constructs were of the correct length. The 381 construct was confirmed once again to be the correct length. | Colony PCR of 8 colonies picked from 391 in pSB3K3 transformants, 8 colonies picked from 374 in pSB3K3 transformants, 8 colonies picked from 384 in pSB3K3, and 1 colony picked from 381 in pSB3K3 transformants confirmed to contain an insert of the right length. Overnight cultures of each of those colonies as well. No 374 constructs were found to contain inserts of the right length, however 4 384 constructs were of the correct length. The 381 construct was confirmed once again to be the correct length. | ||
| Line 19: | Line 19: | ||
====MelA BioBrick==== | ====MelA BioBrick==== | ||
| - | PCR done of the | + | PCR done of the original MelA and the first MelA intermediate (without the second PstI site), to create the DNA for a PstI restriction digest. After PCR the DNA was run on a CYBRsafe-stained gel and then extracted with the QuiGen kit. A Restriction digest was carried out using the fastdigest buffer and PstI and left for three hours. |
We also tested the Primer C again, as it didn't work last time. Three PCR's were done: | We also tested the Primer C again, as it didn't work last time. Three PCR's were done: | ||
| Line 25: | Line 25: | ||
*Primer F and a new aliquot of Primer C | *Primer F and a new aliquot of Primer C | ||
*Primer F and revortexed and re-aliquoted primer C from the origonal vial | *Primer F and revortexed and re-aliquoted primer C from the origonal vial | ||
| - | None of these produced any result - there is | + | None of these produced any result - there is likely to be a problem with the primer. |
| + | |||
| + | ====Pyocyanin *NEW*==== | ||
| + | |||
| + | The following biobricks were transformed into TOP10 and plated out in preparation for pyocyanin biosynthesis: | ||
| + | |||
| + | *I746910 (pSB1A2) - pBAD + B0034 | ||
| + | *I723024 (pSB3K3) - PhzM | ||
| + | *I723025 (pSB1A2) - PhzS | ||
| + | *B0015 (pSB1AK3) - double terminator | ||
| + | *B0034 (pSB1A2) - RBS | ||
===Dry Work=== | ===Dry Work=== | ||
| Line 36: | Line 46: | ||
Development of Matlab programs to automatically process plate reader data was continued. | Development of Matlab programs to automatically process plate reader data was continued. | ||
| + | |||
| + | {{Template:CambridgeNewPage}} | ||
| + | |||
| + | == Tuesday == | ||
| + | |||
| + | ===Wet Work=== | ||
| + | |||
| + | ====Threshold Device==== | ||
| + | |||
| + | Colony PCR of 391 in pSB3K3 showed none had the correct insert. Ligations of 380, 382, 385, 390 into pSB3K3, transformed into Top10. | ||
| + | |||
| + | 381 and 384 were tranformed into the arabinose strain in preparation for assaying using the plate reader. | ||
| + | |||
| + | ====Violacein==== | ||
| + | |||
| + | James has a plasmid that produces cuiR which when stimulated with C6-HSL acts activates the vioA promoter. We put the vio+ cells into the shaking incubator, preparing to make them competent tomorrow. | ||
| + | |||
| + | ====Pyocyanin==== | ||
| + | |||
| + | Transformations of I746910, I723025 and B0034 worked, and were put in overnight culture in preparation for miniprepping. | ||
| + | |||
| + | I723024 and B0015 were transformed again into TOP10. | ||
| + | |||
| + | |||
| + | |||
| + | {{Template:CambridgeNewPage}} | ||
| + | |||
| + | ==Wednesday== | ||
| + | |||
| + | ===Wet Work=== | ||
| + | |||
| + | ====Threshold Device==== | ||
| + | |||
| + | *Overnight colony PCR of 380, 382, 385, 390 in pSB3K3. | ||
| + | *Moved 375, 392, 394, 395 into pSB3K3, transformed into TOP10. | ||
| + | *Overnight colonies of 381 in pSBK3K in arabinose strain and 384 in pSB3K3 in arabinose strain in preparation for plate reader run tomorrow, as well as the appropriate controls. | ||
| + | |||
| + | ====MelA BioBrick==== | ||
| + | |||
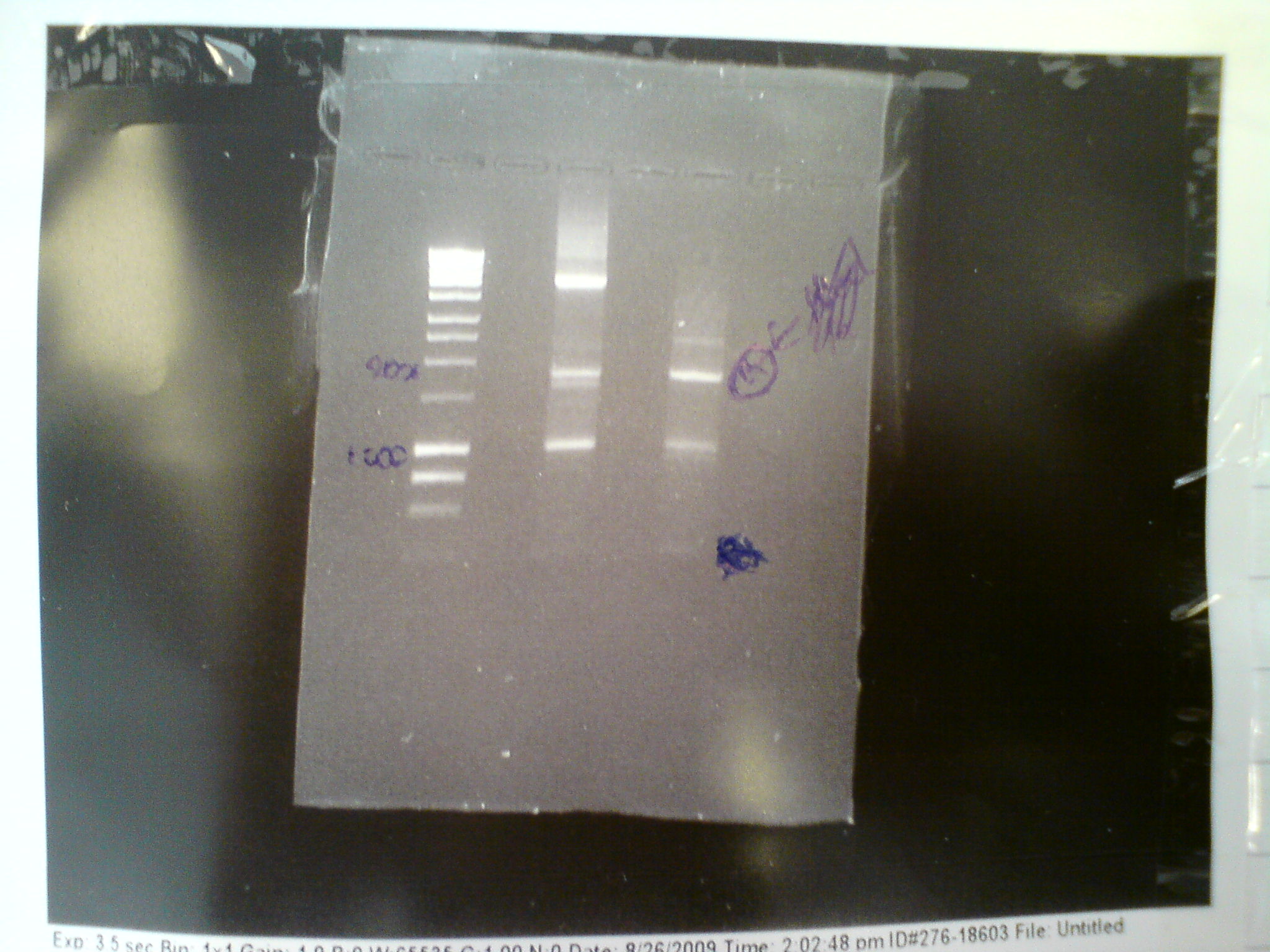
| + | In a final attempt to get Primer C to work, we tried touchdown PCR with the lowest temperatuire 45 degrees C. Primer C and Primer F were used with the origonal melA gene and the end product of the PCR was run on an ethidium bromide gel at 80V for 45 mins: | ||
| + | |||
| + | [[Image:Igem 1 003.jpg | 400px]] | ||
| + | |||
| + | Product is shown starred in the above gel. The MelA protocol can now be completed. | ||
| + | |||
| + | ====Violacein==== | ||
| + | |||
| + | Made competent vio+ cells and transformed them with James's plasmid. Transformed cells were plated on trimethoprim/ampicillin plates and placed in the 37 degree incubator overnight. | ||
| + | |||
| + | ===Dry Work=== | ||
| + | |||
| + | Planned standard assemblies for the next month | ||
| + | |||
| + | {{Template:CambridgeNewPage}} | ||
| + | |||
| + | ==Thursday== | ||
| + | |||
| + | ===Wet Work=== | ||
| + | |||
| + | ====Threshold Device==== | ||
| + | |||
| + | *Successfully moved 380, 382, 385, 390 into pSB3K3, cultured colonies overnight in preparation for miniprep tomorrow | ||
| + | *Overnight colony PCR of 375, 392, 394, 395 into pSB3K3. | ||
| + | *Overnight plate reader run of arabinose strain colonies transformed with 382 in pSB3K3 and arabinose strain colonies transformed with 384 in pSB3K3 exposed to varying concentrations of arabinose | ||
| + | |||
| + | ====Violacein==== | ||
| + | |||
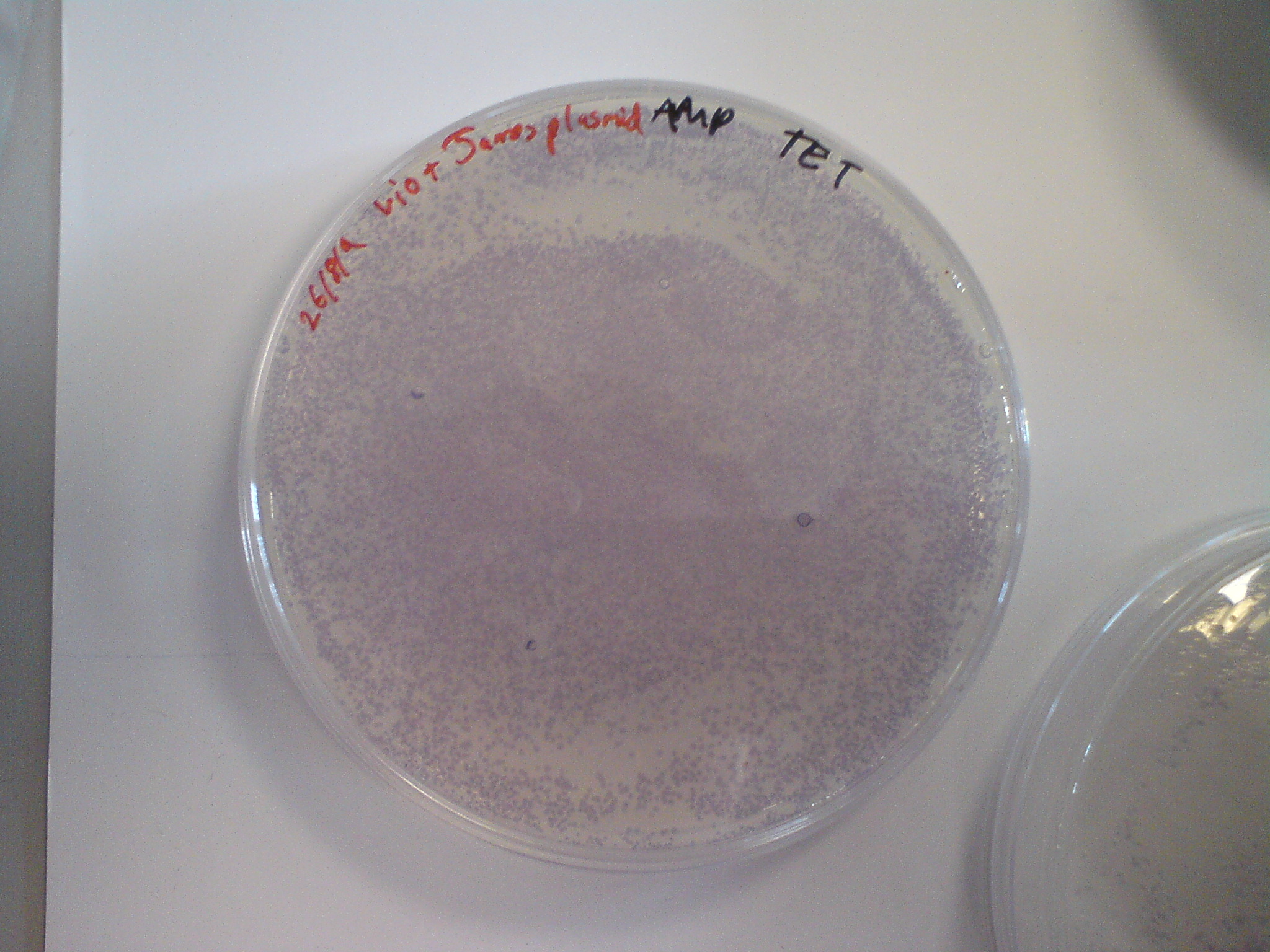
| + | Observed the transformed cells. Violet pigment was seen, after only one night of growth! | ||
| + | |||
| + | [[Image:Igem_1_001.jpg | 400px]] | ||
| + | |||
| + | In order to collect liquid culture colour for characterisation with the plate-reader, two cultures were grown. One contained 10mM of 3-oxo-C6-HSL in order to increase the level of pigment, hopefully such that it can be seen, and therefore characterised, in liquid culture. | ||
| + | |||
| + | ====MelA BioBrick==== | ||
| + | |||
| + | Two PCR's were run, using the MelA-biobrick intermediate created last week (minus one PstI site) as the template: | ||
| + | :*Primer C and Primer F | ||
| + | :*Primer A and Primer B | ||
| + | These two will remove the second PstI site. | ||
| + | |||
| + | After running the gel, the correct products were excised and purified using the QuiGen gel purification kit. The products were then used for a further PCR run (overnight) using primers A and F. | ||
| + | |||
| + | If the correct products were removed, then this final run should produce the MelA BioBrick. | ||
| + | |||
| + | ===Dry Work=== | ||
| + | |||
| + | Prepared iGEM-style practise presentation for the meeting on Friday. | ||
| + | |||
| + | {{Template:CambridgeNewPage}} | ||
| + | |||
| + | ==Friday== | ||
| + | |||
| + | ===Wet Work=== | ||
| + | |||
| + | ====Threshold Device==== | ||
| + | |||
| + | Successfully moved 375, 392, 394, 395 into pSB3K3. | ||
| + | |||
| + | ====Violacein==== | ||
| + | |||
| + | Observed the liquid cultures made yesterday: | ||
| + | |||
| + | [[Image:Igem_2.jpg | 400px]] | ||
| + | |||
| + | The tube on the left contains the 3-oxo-C6-HSL while the one on the right just contains the vio bacteria. Violacein in liquid culture! This can now be used with the plate reader to characterise the pigment. | ||
| + | |||
| + | ===Dry Work=== | ||
| + | |||
| + | ====Practise Presentation==== | ||
| + | |||
| + | We prepared an iGEM-style presentation for the afternoon, and presented it for the faculty. The following comments and suggestions were made regarding it: | ||
| + | |||
| + | :*Pictures need to be close-up of the coloured colonies, with no satilites and with a control for comparison | ||
| + | :*Slide design should be looked at - in particular the coloured sidebar, which created dead space | ||
| + | :*Forward rather than a backward explanation (Sensor - threshold - colour) and with a navigation bar (perhaps at the side?) to show the audience where we are in the talk | ||
| + | :*Ways to characterise the different strains and their effects were discussed, such as using a bar graph with coloured pictures of the cells in the boxes - an effective way to show a large amount of information on one slide. | ||
| + | :*Testing the threshold. It was suggested that as well as attaching the different threshold levels to different colours, we first attach them all to the same colour as proof they respond to different input levels | ||
| + | :*The actual physical product should be constructed in a prototype form, such as by using 5-well microscope slides. We were also encouraged to think more about this final product and how we would practically produce it. | ||
| + | :*BioBrick numbers need to be included | ||
| + | :*The pigment production requires a little more science background | ||
| + | |||
| + | We also decided not too concentrate on the phzS and phzM pigments, as these have already been made into Biobricks. Instead, we will just concentrate on the proper characterisation of the carotenoid pigments, creating the MelA and Violacein biobricks, and attaching them too the threshold level amplifiers. | ||
<!--Do not remove the first and last lines in this page!-->{{Template:CambridgeBottom}} | <!--Do not remove the first and last lines in this page!-->{{Template:CambridgeBottom}} | ||
Latest revision as of 16:27, 7 October 2009
Categories :
Project :
-
Overview
Sensitivity Tuner
--- Characterisation
--- Modelling
Colour Generators
--- Carotenoids (Orange/Red)
--- Melanin (Brown)
--- Violacein (Purple/Green)
The Future
Safety
Notebook :
Team Logistics :
Week 7
Monday
Wet Work
Threshold Device
Colony PCR of 8 colonies picked from 391 in pSB3K3 transformants, 8 colonies picked from 374 in pSB3K3 transformants, 8 colonies picked from 384 in pSB3K3, and 1 colony picked from 381 in pSB3K3 transformants confirmed to contain an insert of the right length. Overnight cultures of each of those colonies as well. No 374 constructs were found to contain inserts of the right length, however 4 384 constructs were of the correct length. The 381 construct was confirmed once again to be the correct length.
Overnight,
- Restirction digests of pSB3K3, 380, 382, 385, and 390.
- Colony PCR of 10 new 391 in pSB3K3 transformants, another 374 in pSB3K3 transformants
MelA BioBrick
PCR done of the original MelA and the first MelA intermediate (without the second PstI site), to create the DNA for a PstI restriction digest. After PCR the DNA was run on a CYBRsafe-stained gel and then extracted with the QuiGen kit. A Restriction digest was carried out using the fastdigest buffer and PstI and left for three hours.
We also tested the Primer C again, as it didn't work last time. Three PCR's were done:
- Primer F and Primer C (same aliquot as used last time, to check it wasn't just an experimental error on my part)
- Primer F and a new aliquot of Primer C
- Primer F and revortexed and re-aliquoted primer C from the origonal vial
None of these produced any result - there is likely to be a problem with the primer.
Pyocyanin *NEW*
The following biobricks were transformed into TOP10 and plated out in preparation for pyocyanin biosynthesis:
- I746910 (pSB1A2) - pBAD + B0034
- I723024 (pSB3K3) - PhzM
- I723025 (pSB1A2) - PhzS
- B0015 (pSB1AK3) - double terminator
- B0034 (pSB1A2) - RBS
Dry Work
We found the phzM and phzS pigments in the registry!
- BBa_I723024 (phzM)
- BBa_I723025 (phzS)
This means we can now start working on these pigments, connecting them to processing and logic systems.
Development of Matlab programs to automatically process plate reader data was continued.
Tuesday
Wet Work
Threshold Device
Colony PCR of 391 in pSB3K3 showed none had the correct insert. Ligations of 380, 382, 385, 390 into pSB3K3, transformed into Top10.
381 and 384 were tranformed into the arabinose strain in preparation for assaying using the plate reader.
Violacein
James has a plasmid that produces cuiR which when stimulated with C6-HSL acts activates the vioA promoter. We put the vio+ cells into the shaking incubator, preparing to make them competent tomorrow.
Pyocyanin
Transformations of I746910, I723025 and B0034 worked, and were put in overnight culture in preparation for miniprepping.
I723024 and B0015 were transformed again into TOP10.
Wednesday
Wet Work
Threshold Device
- Overnight colony PCR of 380, 382, 385, 390 in pSB3K3.
- Moved 375, 392, 394, 395 into pSB3K3, transformed into TOP10.
- Overnight colonies of 381 in pSBK3K in arabinose strain and 384 in pSB3K3 in arabinose strain in preparation for plate reader run tomorrow, as well as the appropriate controls.
MelA BioBrick
In a final attempt to get Primer C to work, we tried touchdown PCR with the lowest temperatuire 45 degrees C. Primer C and Primer F were used with the origonal melA gene and the end product of the PCR was run on an ethidium bromide gel at 80V for 45 mins:
Product is shown starred in the above gel. The MelA protocol can now be completed.
Violacein
Made competent vio+ cells and transformed them with James's plasmid. Transformed cells were plated on trimethoprim/ampicillin plates and placed in the 37 degree incubator overnight.
Dry Work
Planned standard assemblies for the next month
Thursday
Wet Work
Threshold Device
- Successfully moved 380, 382, 385, 390 into pSB3K3, cultured colonies overnight in preparation for miniprep tomorrow
- Overnight colony PCR of 375, 392, 394, 395 into pSB3K3.
- Overnight plate reader run of arabinose strain colonies transformed with 382 in pSB3K3 and arabinose strain colonies transformed with 384 in pSB3K3 exposed to varying concentrations of arabinose
Violacein
Observed the transformed cells. Violet pigment was seen, after only one night of growth!
In order to collect liquid culture colour for characterisation with the plate-reader, two cultures were grown. One contained 10mM of 3-oxo-C6-HSL in order to increase the level of pigment, hopefully such that it can be seen, and therefore characterised, in liquid culture.
MelA BioBrick
Two PCR's were run, using the MelA-biobrick intermediate created last week (minus one PstI site) as the template:
- Primer C and Primer F
- Primer A and Primer B
These two will remove the second PstI site.
After running the gel, the correct products were excised and purified using the QuiGen gel purification kit. The products were then used for a further PCR run (overnight) using primers A and F.
If the correct products were removed, then this final run should produce the MelA BioBrick.
Dry Work
Prepared iGEM-style practise presentation for the meeting on Friday.
Friday
Wet Work
Threshold Device
Successfully moved 375, 392, 394, 395 into pSB3K3.
Violacein
Observed the liquid cultures made yesterday:
The tube on the left contains the 3-oxo-C6-HSL while the one on the right just contains the vio bacteria. Violacein in liquid culture! This can now be used with the plate reader to characterise the pigment.
Dry Work
Practise Presentation
We prepared an iGEM-style presentation for the afternoon, and presented it for the faculty. The following comments and suggestions were made regarding it:
- Pictures need to be close-up of the coloured colonies, with no satilites and with a control for comparison
- Slide design should be looked at - in particular the coloured sidebar, which created dead space
- Forward rather than a backward explanation (Sensor - threshold - colour) and with a navigation bar (perhaps at the side?) to show the audience where we are in the talk
- Ways to characterise the different strains and their effects were discussed, such as using a bar graph with coloured pictures of the cells in the boxes - an effective way to show a large amount of information on one slide.
- Testing the threshold. It was suggested that as well as attaching the different threshold levels to different colours, we first attach them all to the same colour as proof they respond to different input levels
- The actual physical product should be constructed in a prototype form, such as by using 5-well microscope slides. We were also encouraged to think more about this final product and how we would practically produce it.
- BioBrick numbers need to be included
- The pigment production requires a little more science background
We also decided not too concentrate on the phzS and phzM pigments, as these have already been made into Biobricks. Instead, we will just concentrate on the proper characterisation of the carotenoid pigments, creating the MelA and Violacein biobricks, and attaching them too the threshold level amplifiers.
 "
"