Notebook > AND Gate 1 > Core > Rencheng Gao's Note
2009.8.17
0:50
Bacteria Culture: 11N-pcrS6(1~4), 2K-pcrS6(1~4), pla2+SupD(1~4), pcr2+SupD(1~4)
12:00
Miniprep: 2GpcrS6(1~4), 5JpcrS6(1~4), 1HpcrS6(1~4)
17:00
Double-enzyme digestion assessment: 2GpcrS6(1~4), 5JpcrS6(1~4), 1HpcrS6(1~4)

24:00
Bacteria Culture: 1H-pcrS6(5~7), 11N-pcrS6(5~7), 2K-pcrS6(5~7), pla2+SupD(5~7), pcr2+SupD (5~7)
2009.8.18
11:00
Miniprep: plaSupD(5~7), pcrSupD(5~7)
14:35
Double-enzyme digestion assessment: plaSupD(5~7), pcrSupD(5~7)
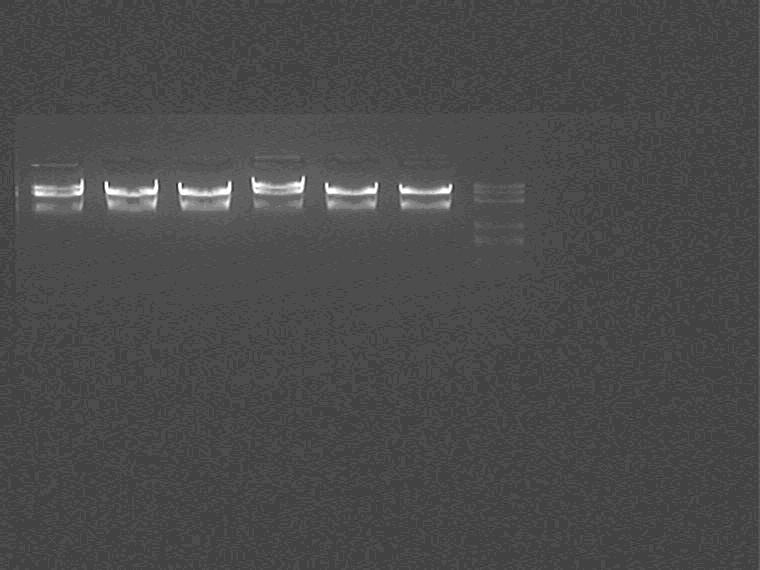
14:50
Ligation:
Insert: 11N, 2K, 1H
Vector: pcrS6
21:00
Transformation
2009.8.19
15:00
Double-enzyme digestion assessment
Plasmid: plaS6, pcrS6, 5JpcrS6, 2GpcrS6
22:00
Bacteria culture: 2K(1~5), 1H(1~5)
22:30
Gel Purification: plaS6, pcrS6, 5JpcrS6, 2GpcrS6
00:30
Ligation
Insert: plaS6, pcrS6, 5JpcrS6, 2GpcrS6
Vector: 1-7G
2009.8.20
12:00
Send samples for sequencing: plaS6, pcrS6, 5JpcrS6, 2GpcrS6
14:00
Miniprep: 2KpcrS6(1~5), 1HpcrS6(1~5), 11NpcrS6(1~5)
17:00
Double-enzyme digestion: 2KpcrS6(1~5), 1HpcrS6(1~5), 11NpcrS6(1~5)
22:00
Store the strain: 2KpcrS6(1), 1HpcrS6(1), 11NpcrS6(1)
2009.8.21
9:30
Send samples for sequencing: 2KpcrS6(1), 1HpcrS6(1), 11NpcrS6(1)
2009.8.22
11:00
Bacteria culture: pcrS6(1~4), plaS6(1~4), 2GpcrS6(1~4), 5JpcrS6(1~4)
15:30
Double-enzyme digestion: 2KpcrS6(1), 1HpcrS6(1), 11NpcrS6(1)
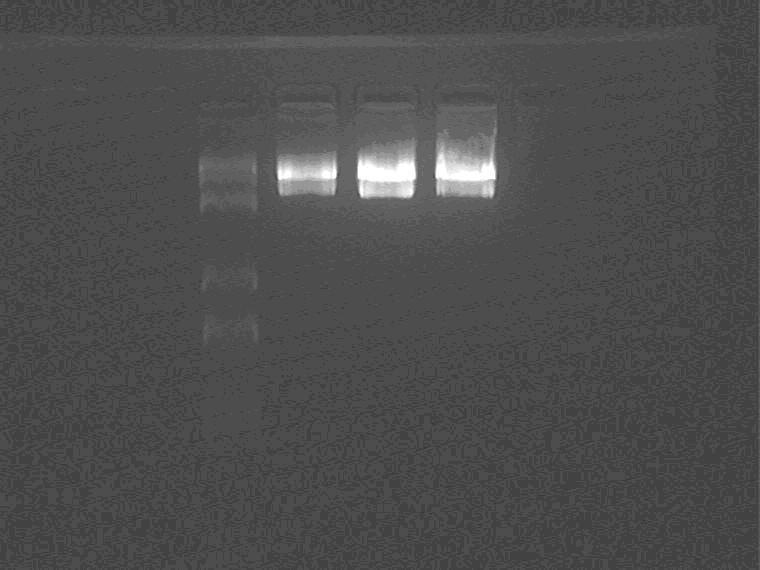
22:00
Gel purification
23:00
Miniprep: pcrS6(1~4), plaS6(1~4), 2GpcrS6(1~4), 5JpcrS6(1~4)
1:00
Ligation:
Insert: 2KpcrS6(1), 1HpcrS6(1), 11NpcrS6(1)
Vector:1-7G
2009.8.23
13:00
Double-enzyme digestion assessment: pcrS6(1~4), plaS6(1~4), 2GpcrS6(1~4), 5JpcrS6(1~4)
15:00
Transformation
0:00
Bacteria culture: 5JpcrS6-low-copy, 2GpcrS6-low-copy
2009.8.24
11:00
Bacteria culture: 2KpcrS-low-copy(1~4), 1HpcrS-low-copy(1~4), 11NpcrS-low-copy(1~4)
14:00
Double-enzyme digestion (back insert): T7pro-GFP
Double-enzyme digestion (back vector): 5JpcrS6-low-copy, 2GpcrS6-low-copy
21:00
Gel purification
22:00
Miniprep: 2KpcrS-low-copy(1~4), 1HpcrS-low-copy(1~4), 11NpcrS-low-copy(1~4)
23:30
Ligation
Insert: T7pro-GFP
Vector: 5JpcrS6-low-copy, 2GpcrS6-low-copy
24:00
Double-enzyme digestion: 2KpcrS-low-copy(1~4), 1HpcrS-low-copy(1~4), 11NpcrS-low-copy (1~4)
2009.8.25
13:30
Transformation: 2G-T7pro-GFP, 5J-T7pro-GFP
17:00
Strain storage: 2KpcrS-low-copy, 1HpcrS-low-copy, 11NpcrS-low-copy
2009.8.27
14:30
Inducement: AND gate(5J, 2G)
HSL: 10-4M
Arabinose: 10-4M
17:00
Double-enzyme digestion (front insert): 2KpcrS6(1), 1HpcrS6(1), 11NpcrS6(1)
2009.8.28
13:00
Inducement: AND gate(5J, 2G)
HSL: 10-4M
Arabinose: 10-4M
13:40
Double-enzyme digestion (front insert): T7pro-GFP
2009.8.29
13:00
Bacteria culture: 1HpcrS(1~3), 11NpcrS(1~3), 2KpcrS(1~3), 5JpcrS(1~3), 2GpcrS(1~3)
21:45
Ligation
Insert: 5NT7ptag, 2MT7ptag, 2IT7ptag, 1JT7ptag
Vector: pcrS6
2009.8.30
13:00
Miniprep: 1HpcrS(1~3), 11NpcrS(1~3), 2KpcrS(1~3), 5JpcrS(1~3), 2GpcrS(1~3)
14:00
Double-enzyme digestion assessment: 1HpcrS(1~3), 11NpcrS(1~3), 2KpcrS(1~3), 5JpcrS(1~3), 2GpcrS(1~3)
17:00
Bacteria culture: 5NT7ptag(1~3), 2MT7ptag(1~3), 2IT7ptag(1~3), 1JT7ptag(1~3)
21:30
Strain Storage: 2K(3), 5J(2), 1H(1), 11N(1), 2G(2)
22:00
Double-enzyme digestion (front vector): 2K(3), 5J(2), 1H(1), 11N(1), 2G(2)
2009.8.31
13:00
Miniprep: 5NT7ptag(1~3), 2MT7ptag(1~3), 2IT7ptag(1~3), 1JT7ptag(1~3)
14:00
Gel purification: 2K(3), 5J(2), 1H(1), 11N(1), 2G(2)
16:00
Send samples for sequencing: 2K(3), 5J(2), 1H(1), 11N(1), 2G(2)
17:00
Ligation:
Vector: 2K(3), 5J(2), 1H(1), 11N(1), 2G(2)
17:30
Double-enzyme digestion: 5NT7ptag(1~3), 2MT7ptag(1~3), 2IT7ptag(1~3), 1JT7ptag(1~3)
1:30
Strain Storage: 2I(1), 2M(2), 1J(2), 5N(1)
2009.9.1
13:30
Double-enzyme digestion (Front Vector)
Plasmid: 5N, 2M, 2I, 1J
21:30
Culture the bacteria: 5J, 2K, 1H, 2G-T7pro GFP
23:20
Ligation
Vector: 5N, 2M, 2I 2J
Insert: T7pro-GFP
2009.9.2
14:00
Inducement: 5J, 2K, 1H, 2G, 11N
22:00
Double-enzyme digestion
Plasmid: 1JpcrS, 11NpcrS, 2IpcrS, 5JpcrS, 2MpcrS, 1HpcrS, 5NpcrS, 2GpcrS, 2KpcrS
2009.9.4
15:30
Send samples for sequencing: 5N, 2M, 2I, 1J
15:45
Gel purification: 11N, 1H, 2M, 1J, 2G, 5N, 1J
16:00
Ligation:
Vector: 1-7G
Insert: 11N, 1H, 2M, 1J, 2G, 5N, 1J
2009.9.5
17:00
Double-enzyme digestion
Plasmid: 11N(1), 2M(2), 2K(2), 1H(1), 5J(1), 1J(2), 2G(2)
2009.9.6
16:00
Double-enzyme digestion (5J, 2I, 1J, 2K, 1H, 5N, 2M, 11N, 2G)
22:00
Culture the bacteria: 5N(3), 2I(1), 2I(3)
00:00
Ligation:
Insert: 5J, 2K, 1H, 2M, 2G
Vector: 1-7G
2009.9.7
14:00
Mini-prep: 2I(1), 5N(3), 2I(3)
16:30
Double-enzyme digestion: 1J
17:00
Prepare competent cells
21:00
Transformation: 2G, 2I(1), 2I(3), 5N(3)
2009.9.8
14:00
Culture the bacteria:
15:00
Ligation
Insert: 2M, 1H
Vector: 1-7G
2009.9.9
01:00
Double-enzyme digestion
Plasmid: 11N, 5J, 2K
20:30
Inducement: 2G, 5N, 2I
24:00
Mini-prep: 1J(1-3), 2M(1-3), 1H(1-3)
^Top
|
 "
"



