Team:Newcastle/Labwork/8 September 2009
From 2009.igem.org
(→Experiment procedure) |
Babyneurone (Talk | contribs) (→Midi-prep of transformant E. coli cells) |
||
| (36 intermediate revisions not shown) | |||
| Line 3: | Line 3: | ||
{{:Team:Newcastle/Left}} | {{:Team:Newcastle/Left}} | ||
__NOTOC__ | __NOTOC__ | ||
| - | =Lab | + | [[Image:Team Newcastle 2009 iGEM ProbationaryP-Sign.PNG|50px|right]] |
| - | ==<u>Metal | + | =Formal Lab Session - 8th September 2009= |
| + | [[Image:Team Newcastle 2009 iGEM 08-09-09 IMG 0925.JPG|400px|center]] | ||
| + | <br> | ||
| + | =<font color="Orange"><u>Overview</u></font>= | ||
| + | <font color="Orange"> | ||
| + | *[[#Metal Sensor Team|Metal Sensor Team]] '''- ''' | ||
| + | <br> | ||
| + | *[[#Stochastic Switch Team|Stochastic Switch Team]] '''- ''' | ||
| + | <br> | ||
| + | *[[#Sporulation Tuning/Chassis Team|Sporulation Tuning/Chassis Team]] '''- ''' | ||
| + | <br> | ||
| + | </font> | ||
| + | <br> | ||
| + | ==<u>Metal Sensor Team</u>== | ||
===Introduction=== | ===Introduction=== | ||
Yesterday's lab session saw the Metal Sensing team innoculate 6 tubes of 5ml LB (plus ampicillin) with six cultures of ''DH5-alpha'' transformed with ''BBa_J33206'' in plasmid ''pSB1A2'' in the morning. By the time afternoon had arrived, the bacteria had grown to sufficient numbers; three of these cultures were then used to further inoculate 3 flasks of 50ml LB+amp. The three flasks of 50ml LB+amp + transformant ''E. coli'' cells were then placed in the orbital incubator and will take part in today's midi-prep. Meanwhile the six tubes were used in a mini-prep attempt but alas, the procedure went wrong and had to be abandoned. | Yesterday's lab session saw the Metal Sensing team innoculate 6 tubes of 5ml LB (plus ampicillin) with six cultures of ''DH5-alpha'' transformed with ''BBa_J33206'' in plasmid ''pSB1A2'' in the morning. By the time afternoon had arrived, the bacteria had grown to sufficient numbers; three of these cultures were then used to further inoculate 3 flasks of 50ml LB+amp. The three flasks of 50ml LB+amp + transformant ''E. coli'' cells were then placed in the orbital incubator and will take part in today's midi-prep. Meanwhile the six tubes were used in a mini-prep attempt but alas, the procedure went wrong and had to be abandoned. | ||
| Line 12: | Line 25: | ||
===Procedure=== | ===Procedure=== | ||
====Midi-prep of transformant ''E. coli'' cells==== | ====Midi-prep of transformant ''E. coli'' cells==== | ||
| + | |||
To carry out the midi-prep we used Sigma-Aldrich's GenElute midi-prep kit and with it, [https://2009.igem.org/Team:Newcastle/Project/Labwork/MoreProtocols#Midiprep the protocol]. There were no changes to the protocol except for an additional step after step 10b - an additional 2 minute spin to ensure ethanol removal. The route we took was the '''Spin method'''. | To carry out the midi-prep we used Sigma-Aldrich's GenElute midi-prep kit and with it, [https://2009.igem.org/Team:Newcastle/Project/Labwork/MoreProtocols#Midiprep the protocol]. There were no changes to the protocol except for an additional step after step 10b - an additional 2 minute spin to ensure ethanol removal. The route we took was the '''Spin method'''. | ||
| Line 68: | Line 82: | ||
<br> | <br> | ||
===Results=== | ===Results=== | ||
| - | [[Image:Newcastle Geldoc 2009-09-08 17hr 50min.PNG| | + | [[Image:Newcastle Geldoc 2009-09-08 17hr 50min.PNG|250px]] |
| - | [[Image:Newcastle Geldoc 2009-09-08 17hr 52min.PNG| | + | [[Image:Newcastle Geldoc 2009-09-08 17hr 52min.PNG|250px]] |
<br> | <br> | ||
The two gel photographs above show the restriction enzyme digests of the ''pSB1A2'' plasmid, which contains the ''BBa_J33206'' BioBrick. The wells in the gel are as follows: | The two gel photographs above show the restriction enzyme digests of the ''pSB1A2'' plasmid, which contains the ''BBa_J33206'' BioBrick. The wells in the gel are as follows: | ||
| Line 81: | Line 95: | ||
* Lane 7 = ''HindIII'' ladder | * Lane 7 = ''HindIII'' ladder | ||
<br> | <br> | ||
| + | [[Image:Team Newcastle 2009 iGEM 08-09-09 IMG 0961.JPG|200px|right]] | ||
It can be seen that the ''pSB1A2'' in lanes 4 and 5 have been successfully linearised by ''EcoRI'' and ''PstI'' respectively as they appear to have taken longer in the gel than the uncut plasmid (lane 3). It can also be seen that after 2 restriction enzymes have been applied, the plasmid in lane 6 has been cut into the proportions predicted in the Parts Registry. | It can be seen that the ''pSB1A2'' in lanes 4 and 5 have been successfully linearised by ''EcoRI'' and ''PstI'' respectively as they appear to have taken longer in the gel than the uncut plasmid (lane 3). It can also be seen that after 2 restriction enzymes have been applied, the plasmid in lane 6 has been cut into the proportions predicted in the Parts Registry. | ||
| Line 94: | Line 109: | ||
==<u>Stochastic Switch Team</u>== | ==<u>Stochastic Switch Team</u>== | ||
| + | [[Image:Team Newcastle 2009 iGEM 08-09-09 IMG 0915.JPG|300px|center]] | ||
| + | <br> | ||
We tried the minipreps for the transformed colonies with the ligation product of pSB1AT3 + sspB however we were not successfull. On the gel we could not see any band. We will repeat the same process tomorrow. This time we will double the spinning time after adding isopropanol. | We tried the minipreps for the transformed colonies with the ligation product of pSB1AT3 + sspB however we were not successfull. On the gel we could not see any band. We will repeat the same process tomorrow. This time we will double the spinning time after adding isopropanol. | ||
We again prepared overnight cultures for the minipreps | We again prepared overnight cultures for the minipreps | ||
| + | [[Image:Team Newcastle 2009 iGEM 08-09-09 IMG 0924.JPG|200px|right]] | ||
Also today we did a transformation using JM109 cells and the ligations from yesterday; sac and ara in pSB1AT3. | Also today we did a transformation using JM109 cells and the ligations from yesterday; sac and ara in pSB1AT3. | ||
We used the promega protocol in the end plating out 400ul and 50ul of each culture making sure we also plated out the ligation controls: | We used the promega protocol in the end plating out 400ul and 50ul of each culture making sure we also plated out the ligation controls: | ||
| - | + | <br> | |
*plasmid+insert+ligase | *plasmid+insert+ligase | ||
*plasmid+insert | *plasmid+insert | ||
*plasmid+ligase | *plasmid+ligase | ||
| + | <br> | ||
| + | In the waiting periods for this procedure we also did a miniprep of the three plasmids gfp-rrnb pSB1AT3 and PMUTIN4 for each we had 3 cultures.The minipreps were put in the -20 freeer ready to be digested tomorrow. | ||
| - | + | ==<u>Sporulation Tuning/Chassis Team</u>== | |
| - | + | ||
| - | ==<u> Chassis | + | |
===Introduction=== | ===Introduction=== | ||
| - | To get clone of sleB, we use plasmid pSB1AT3 which contains restriction sites:EcoRI and speI. Also, our sleB primers were added with these two restriction sites when they were designed.After we digest sleB and pSB1AT3 with same enzymes,we can ligate sleB gene with pSB1AT3 backbone using T4 ligase. This ligased plasmid would trasformated into E.coli. | + | To get clone of ''sleB'', we use plasmid pSB1AT3 which contains restriction sites:EcoRI and speI. Also, our ''sleB'' primers were added with these two restriction sites when they were designed.After we digest ''sleB'' and pSB1AT3 with same enzymes,we can ligate sleB gene with pSB1AT3 backbone using T4 ligase. This ligased plasmid would trasformated into E.coli. |
| + | |||
| + | [[Image:Team_newcastle_2009_jhgeldoc090908_1.JPG.JPG|200px|thumb|''sleB'' gene after PCR clean up]] | ||
| + | [[Image:Team newcastle 2009 jhgeldoc 2.JPG|thumb|''sleB'' gene after gel extraction]] | ||
===Experiment procedure=== | ===Experiment procedure=== | ||
| - | ====Digest sleB PCR product==== | + | ====Digest sleB PCR product and ligate with plasmid==== |
* Prepare the digest reaction. | * Prepare the digest reaction. | ||
dd H2O 7ul | dd H2O 7ul | ||
| Line 136: | Line 157: | ||
22C 10min | 22C 10min | ||
| + | ===Conclusion=== | ||
| + | * The concentration of sleB gene is 13.3ng/ul after gel extraction. | ||
| + | |||
| + | |||
| + | <br> | ||
| + | |||
| + | ===Further plan=== | ||
| + | * Transform ligated sleB and pSB1AT3 backbone into E.coli cells | ||
| + | <br> | ||
| + | |||
| + | <br> | ||
| + | {{:Team:Newcastle/Project/Labwork/CalTemplate}} | ||
| - | |||
| - | |||
{{:Team:Newcastle/Footer}} | {{:Team:Newcastle/Footer}} | ||
{{:Team:Newcastle/Right}} | {{:Team:Newcastle/Right}} | ||
Latest revision as of 12:13, 20 August 2010
Formal Lab Session - 8th September 2009
Overview
Metal Sensor Team
Introduction
Yesterday's lab session saw the Metal Sensing team innoculate 6 tubes of 5ml LB (plus ampicillin) with six cultures of DH5-alpha transformed with BBa_J33206 in plasmid pSB1A2 in the morning. By the time afternoon had arrived, the bacteria had grown to sufficient numbers; three of these cultures were then used to further inoculate 3 flasks of 50ml LB+amp. The three flasks of 50ml LB+amp + transformant E. coli cells were then placed in the orbital incubator and will take part in today's midi-prep. Meanwhile the six tubes were used in a mini-prep attempt but alas, the procedure went wrong and had to be abandoned.
Today's lab session will focus on creating midi-prepped plasmids from the inoculated 50ml LB+amp flasks as well as carry out restriction enzyme digest analysis on a sample. The samples will then be run on agarose gel by DNA gel electrophoresis.
Procedure
Midi-prep of transformant E. coli cells
To carry out the midi-prep we used Sigma-Aldrich's GenElute midi-prep kit and with it, the protocol. There were no changes to the protocol except for an additional step after step 10b - an additional 2 minute spin to ensure ethanol removal. The route we took was the Spin method.
The only thing to note is that we did not midi-prep all of the samples placed in the flask; cultures 1 and 2 (both deriving from the 200ul LB+amp plates) were both spun down with supernatants removed (culture 2 from the 500ul plate was discarded) with culture 1 being placed in the freezer for storage and culture 2 used for the midi-prep process. Here's a summary of the process:
- 50ml of culture 1 was spun down for 10 minutes at 5,000g. The supernatant was aspirated off and the pellet was resuspended in 4ml Resuspension/RNAse solution.
- 4ml of lysis solution was immediately applied and after inverting the falcon tube 6 times, the solution was allowed to sit on the bench for 4 minutes. 4ml neutralisation solution was added once the time had expired and the tube inverted 6 times again.
- 3ml of binding solution was added and after 2 inversions the solution was placed into the filter syringe. With the plunger removed from the syringe, the lysate remained in the syringe for 5 minutes. During this time, 4ml of Column Preparation Solution was allowed to be run through the midiprep binding column (by centrifugation).
- The plunger was then applied to the syringe and half of the lysate (which had been sitting in the syringe barrel for 5 minutes) was injected into the midiprep binding column. The binding column was placed in the centrifuge at 3,000g for 2 minutes and once the eluate had filtered through the remaining half of the lysate was added and treated in the same way.
- 4ml of Wash Solution 1 was then added to the binding column and centrifuged for 2 minutes before 4ml of Wash Solution 2 was added and then placed in the centrifuge for 2 minutes again. The eluate was discarded before the tubes were placed under a further 2 minute spin to ensure all of the ethanol had gone.
- 1 ml of Elution solution was then added to the midiprep binding column before the column was spun for 5 minutes at 3,000g. The eluate was then harvested at the bottom of the binding column and transferred to the freezer for storage.
Restriction Enzyme digests
Samples were taken from the midi-prepped plasmid from culture 2 (from 200ul plate) and placed into 4 Eppendorf tubes with the following contents:
- Eppendorf tube 1 = pSB1A2 with BBa_J332306 + NO ENZYMES
- Eppendorf tube 2 = pSB1A2 with BBa_J332306 + EcoRI
- Eppendorf tube 3 = pSB1A2 with BBa_J332306 + PstI
- Eppendorf tube 4 = pSB1A2 with BBa_J332306 + EcoRI + PstI
The table below shows the contents of each Eppendorf tube:
| pSB1A2 alone | pSB1A2 + EcoRI | pSB1A2 + PstI | pSB1A2 + EcoRI + PstI | |
|---|---|---|---|---|
| DNA (ul) | 2 | 2 | 2 | 2 |
| Restriction Buffer (ul) | 2 | 2 | 2 | 2 |
| Restriction Enzyme(ul) | 0 | 1 | 1 | 2 |
| Distilled water(ul) | 16 | 15 | 15 | 14 |
The digests were allowed to progress for 1 hour in the waterbath, which was set to 37C.
DNA gel electrophoresis analysis
We loaded the gel in the same way suggested by the DNA gel electrophoresis protocol except we loaded 6ul of DNA samples (5ul of DNA and 1ul of loading buffer/dye). After about 30 minutes of electrophoresis under 90v, the gel was taken to the dark room and analysed using GelDoc. The photograph can be seen in the 'Results' section below.
Results
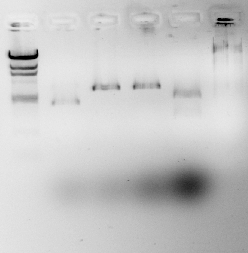
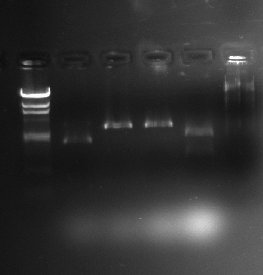
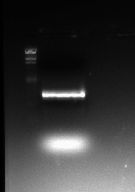
The two gel photographs above show the restriction enzyme digests of the pSB1A2 plasmid, which contains the BBa_J33206 BioBrick. The wells in the gel are as follows:
- Lane 1 = blank
- Lane 2 = HindIII ladder
- Lane 3 = pSB1A2 alone
- Lane 4 = pSB1A2 + EcoRI
- Lane 5 = pSB1A2 + PstI
- Lane 6 = pSB1A2 + EcoRI + PstI
- Lane 7 = HindIII ladder
It can be seen that the pSB1A2 in lanes 4 and 5 have been successfully linearised by EcoRI and PstI respectively as they appear to have taken longer in the gel than the uncut plasmid (lane 3). It can also be seen that after 2 restriction enzymes have been applied, the plasmid in lane 6 has been cut into the proportions predicted in the Parts Registry.
The backbone fragment (the clearest fragment in lane 6) lies between the 2322bp and the 4361bp band which is expected (2079bp is the predicted plasmid backbone size) whereas the BioBrick fragment (fainter and just below the plasmid backbone) lies between the 564bp and the 2027bp bands which is also expected (956bp is the expected BioBrick size).
So we know that Chris French's version of the BBa_J33206 BioBrick in pSB1A2 plasmid DNA is the correct version. This DNA will probably be used further.
There are a few things that can be improved the next time we carry out DNA gel electrophoresis:
- Make up some more HindIII DNA ladder - the DNA in lane 1 is the remainder of the ladder we have used for a while, the ladder in lane 7 has been made quite recently and has proven useless.
- Add more DNA to the samples - the bands are quite faint (especially the BioBrick fragement) - in future we should add more
Stochastic Switch Team
We tried the minipreps for the transformed colonies with the ligation product of pSB1AT3 + sspB however we were not successfull. On the gel we could not see any band. We will repeat the same process tomorrow. This time we will double the spinning time after adding isopropanol.
We again prepared overnight cultures for the minipreps
Also today we did a transformation using JM109 cells and the ligations from yesterday; sac and ara in pSB1AT3.
We used the promega protocol in the end plating out 400ul and 50ul of each culture making sure we also plated out the ligation controls:
- plasmid+insert+ligase
- plasmid+insert
- plasmid+ligase
In the waiting periods for this procedure we also did a miniprep of the three plasmids gfp-rrnb pSB1AT3 and PMUTIN4 for each we had 3 cultures.The minipreps were put in the -20 freeer ready to be digested tomorrow.
Sporulation Tuning/Chassis Team
Introduction
To get clone of sleB, we use plasmid pSB1AT3 which contains restriction sites:EcoRI and speI. Also, our sleB primers were added with these two restriction sites when they were designed.After we digest sleB and pSB1AT3 with same enzymes,we can ligate sleB gene with pSB1AT3 backbone using T4 ligase. This ligased plasmid would trasformated into E.coli.
Experiment procedure
Digest sleB PCR product and ligate with plasmid
- Prepare the digest reaction.
dd H2O 7ul
10X fast digest buffer 7ul
Fast EcoRI 3ul
Fast speI 3ul
sleB DNA 50ul
------------------------------
70ul
- Incubate 1 hour at 37 degree.
- Run the digested product on 0.8% agarose gel.
- Using Gel Extraction Kit to purify digested sleB fragments.
- Ligate sleB fragements with liner pSB1AT3 backbone(already digested with EcoRI and speI)
pSB1AT3 backbone(12ng/ul) 10ul
sleB fragement 10ul
5x Rapid ligation buffer 4ul
dd H2O 15ul
T4 ligase 1ul
-------------------------------------------
30ul
Votex the mixture few seconds 22C 10min
Conclusion
- The concentration of sleB gene is 13.3ng/ul after gel extraction.
Further plan
- Transform ligated sleB and pSB1AT3 backbone into E.coli cells
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
News
Events
- 20 – 21 June 2009 - Europe workshop (London)
- 23 – 24 June 2009 - UK iGEM meetup (Edinburgh)
- 23 October Practice Presentation (Newcastle)
- 23 October T-shirts are ready
- 27 October Practice Presentation (Sunderland)
- 27 October Poster is ready
- 30 October – 2 November 2009 - Jamboree (Boston)
Social Net
- Newcastle iGEM Twitter
- [http://www.facebook.com/home.php#/group.php?gid=131709337641 Newcastle on Facebook]
- [http://www.youtube.com/user/newcastle2009igem Newcastle Youtube Channel]
 "
"