Team:UNIPV-Pavia/Notebook/Week1Jun
From 2009.igem.org
| Line 43: | Line 43: | ||
</td> | </td> | ||
<td> | <td> | ||
| - | + | <font class='didascalia'> | |
| - | + | [[Image:pv_matteo_resusp.jpg|thumb|300px|left|Matteo and Lorenzo resuspending BioBricks from iGEM 2009 plates]] | |
| - | + | </font> | |
</td> | </td> | ||
</tr> | </tr> | ||
| Line 114: | Line 114: | ||
</td> | </td> | ||
<td> | <td> | ||
| - | + | <font class='didascalia'> | |
| - | + | [[Image:pv_susanna_colonypcr.jpg|thumb|220px|left|Susanna preparing samples for colony PCR]] | |
| - | + | </font> | |
</td> | </td> | ||
</tr> | </tr> | ||
Revision as of 13:32, 22 June 2009

|
|
|
|
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
Week from June 1st, to June 7th, 2009
 Previous Week
Previous Week
|
Next Week

|
June, 3rd
- We received lactose monohydrate, M9 minimal salts and Thiamine Hydrochloride from Sigma.
- DNA resuspension from iGEM 2009 plates. We resuspended the two parts we needed to re-built the inconsistent BioBrick Q04400, the five commonly used RBSs and a 3OC6-HSL inducible measurement system for GFP test at Tecan F200:
|
|
- Transformation of resuspended DNA (2 ul) in TOP10 E. coli, plated transformed bacteria and incubated the plates overnight at 37°C.
- We streaked a plate with a 2008 glycerol stock containing E0240 under the control of a constitutive promoter (we call this construct "01") in order to use it in the fluorescence test at Tecan F200 on June 5th.
June, 4th
- All the overnight plates showed colonies. 01 plate showed green fluorescence under UV rays, as we expected because GFP was expressed constitutively!
Unfortunately, B0034 and B0032 plates also showed a very small amount of unexpected red colonies...(@_@!?) These two parts had been resuspended from iGEM 2009 plate 1, just like Berkeley constitutive promoters, but it is not clear how they could be contaminated...
- Anyway, we decided to continue our work and we picked one colony from the following plates:
|
and infected 1 ml of LB + Amp. We incubated the inocula at 37°C, 220 rpm for 5 and 1/2 hours. Of course, we carefully avoided to pick red colonies from B0034 and B0032 plates:) Sequencing checks will tell us if our parts are correct.
- Glycerol stocks for all the grown cultures (except for 01, because we already had it).
- NOTE: we re-picked colonies from J23100, J23101 and J23118 plates because data analysis of experiment 1 at Tecan F200 showed that J23101 promoter seemed stronger than J23100. This is not in accordance to the ranking of the promoters (http://partsregistry.org/Promoters/Catalog/Anderson), so we decided to repeat the test at Tecan F200 with brand new colonies and to store these new glycerol stocks at -80°C.
|
|
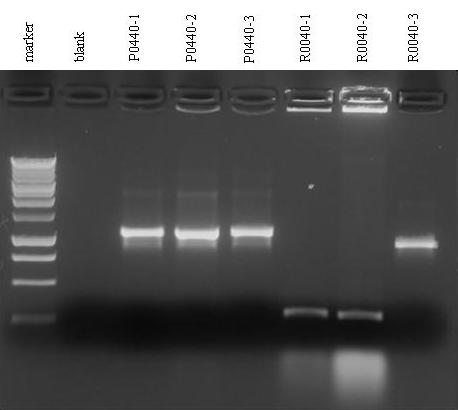
- Gel results: the 3 colonies of P0440 all have the plasmid with the correct length of the insert (1078 bp), while there have been problems with R0040 colonies. R0040-1 and R0040-2 show the correct length of the insert (292 bp) with a high weight contaminant and R0040-3 show an unexpected wrong length. Next week we are going to digest P0440 and R0040 to perform their assembly, so we will check the actual plasmid and insert length again.
<td> </td>
June, 5th
Preparation of the second experiment with Tecan F200
|
|
|
Experiment with Tecan F200
- Description
- Purpose:
- Materials & Methods
- Protocol
- Results
June, 6th
Experiment with Tecan F200
- Results check
 Previous Week
Previous Week
|
Next Week

|
 "
"