Team:Cambridge/Project/Amplification
From 2009.igem.org
(→Further Work - Cambridge 2009) |
(→The Sensitivity Tuner) |
||
| Line 42: | Line 42: | ||
[[Image:thresholddevice3.jpg]] = [[Image:converter.jpg]] | [[Image:thresholddevice3.jpg]] = [[Image:converter.jpg]] | ||
| - | Our hope is that this | + | Our hope is that this kit of sensitivity tuners, characterised using the Cambridge 2007 contructs, can be used by future iGEM teams to fit the needs of their projects. Further, this kit of parts can be expanded in the future to include more phage activators and promoters that have yet to be added to the registry. |
== Recreating Previous Work == | == Recreating Previous Work == | ||
| Line 85: | Line 85: | ||
[[Image:Cambridge_maxrates9.jpg | 600px]] | [[Image:Cambridge_maxrates9.jpg | 600px]] | ||
| - | == | + | == Reconstruction == |
| - | + | ||
| + | Below is a table listing the PoPS converter | ||
<!--Do not remove the first and last lines in this page!-->{{Template:CambridgeBottom}} | <!--Do not remove the first and last lines in this page!-->{{Template:CambridgeBottom}} | ||
Revision as of 06:48, 19 October 2009
Categories :
Project :
-
Overview
Sensitivity Tuner
--- Characterisation
--- Modelling
Colour Generators
--- Carotenoids (Orange/Red)
--- Melanin (Brown)
--- Violacein (Purple/Green)
The Future
Safety
Notebook :
Team Logistics :
The Sensitivity Tuner
Introduction
Cambridge 2007
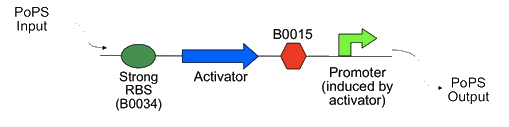
The Cambridge 2007 iGEM team developed a PoPS amplifier system using phage activators and promoters. The system works by using a PoPS input to make an activator protein, as shown in the diagram from their wiki below, which then binds to a promoter and generates a PoPS output.
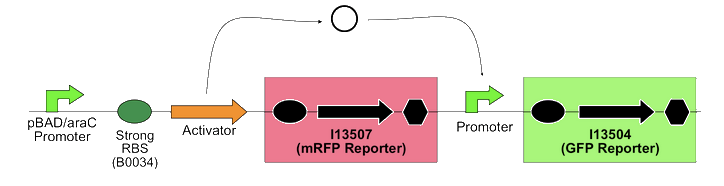
In order to quantify the ratio between PoPS in and PoPS out, the team built the following construction on the high copy plasmid pSB1A2, with mRFP and GFP as PoPS reporter. They genenerated 15 total combinations of different activators and promoters.
They successfully quantified the PoPS amplification factors for each activator/promoter combination after arabinose induction.
Further Work - Cambridge 2009
We have two major goals - characterisation and redisign.
Characterisation
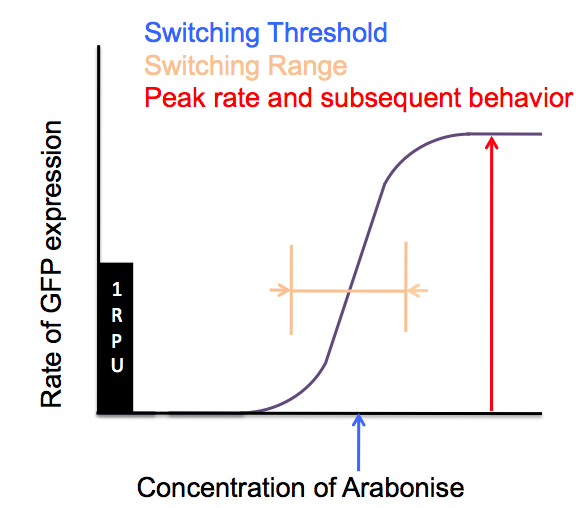
First, we hope to characterise the Cambridge 2007 activator constructs with RFP and GFP reporters on low copy plasmids, looking at three major characteristics relating input (arabinose) to output (GFP) and how they are modified compared to pBad/AraC on its own.
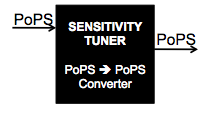
Reconstruction
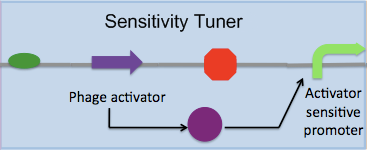
Our second goal is to build a library of devices following the pattern in the figure below, which can also be abstracted as a PoPS converter:
Our hope is that this kit of sensitivity tuners, characterised using the Cambridge 2007 contructs, can be used by future iGEM teams to fit the needs of their projects. Further, this kit of parts can be expanded in the future to include more phage activators and promoters that have yet to be added to the registry.
Recreating Previous Work
We began by recreating the 2007 team's data with some select amplifier constructs. We have the advantage over the 2007 team in that we have a better plate reader that is able to take OD600 absorbance readings at the same time as taking RFP and GFP output readings. For our transformations, we used the E. coli host strain BW27783. This host strain constitutively expresses arabinose transporters and is unable to metabolise arabinose, making it an ideal host for arabinose titration experiments.
Results: The 2007 team's hopes for future work included investigating a problem they attributed to the toxicity of high levels of activator in the cell. Overnight OD600 readings of cells transformed with their amplifier constructs indicated cell death. However, these OD readings were conducted separately from their RFP and GFP output measurements. The 2009 team gathered data on the plate reader capable of taking OD600 absorbance readings as well as RFP and GFP output readings; no OD600 readings suggested cell death due to toxicity.
- graphs*
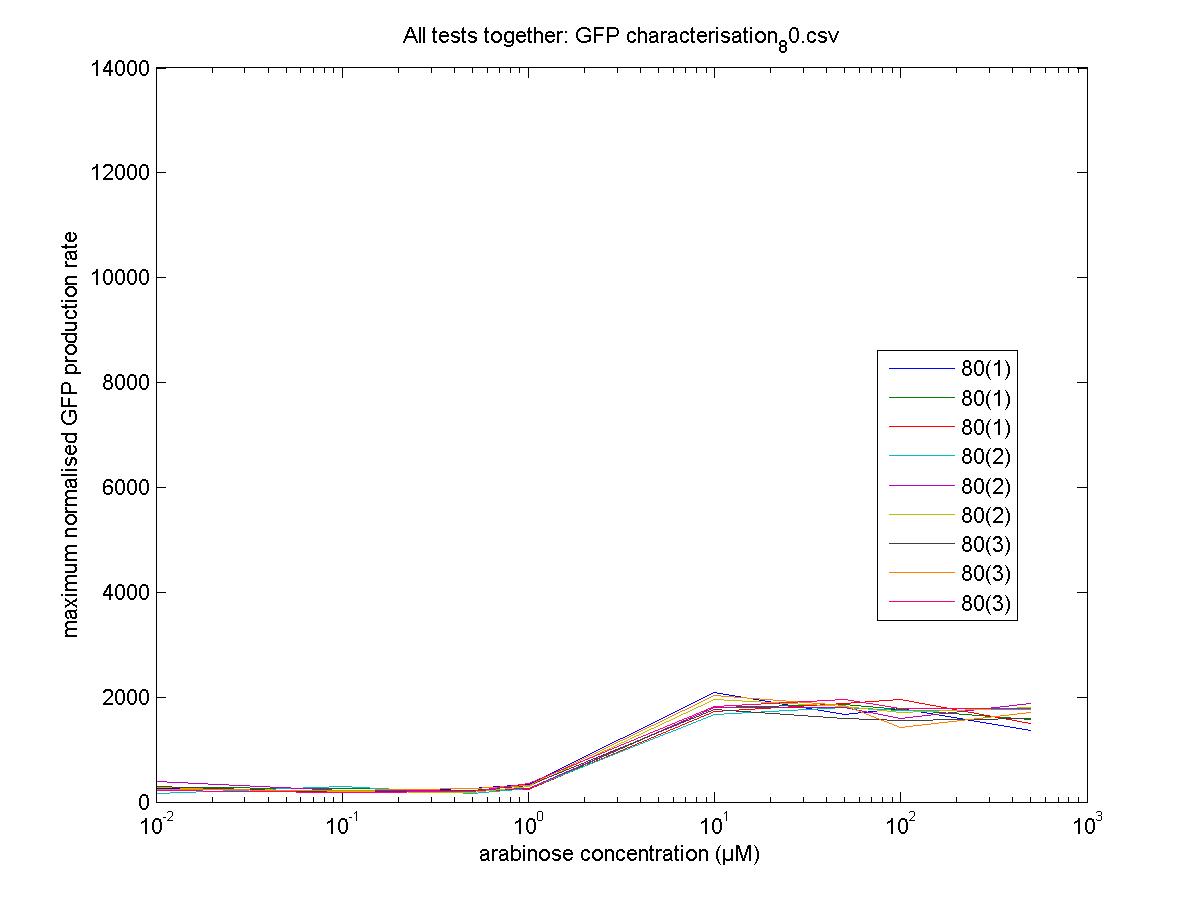
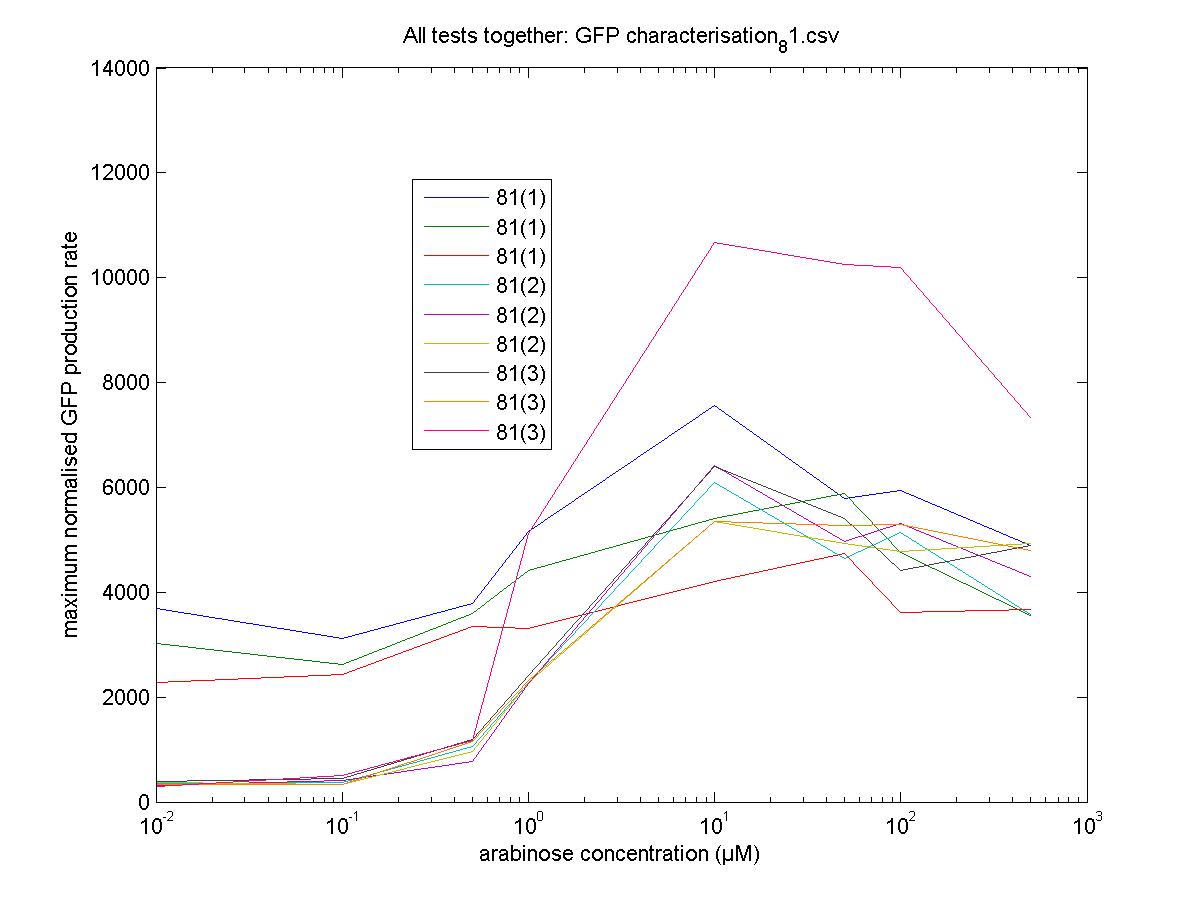
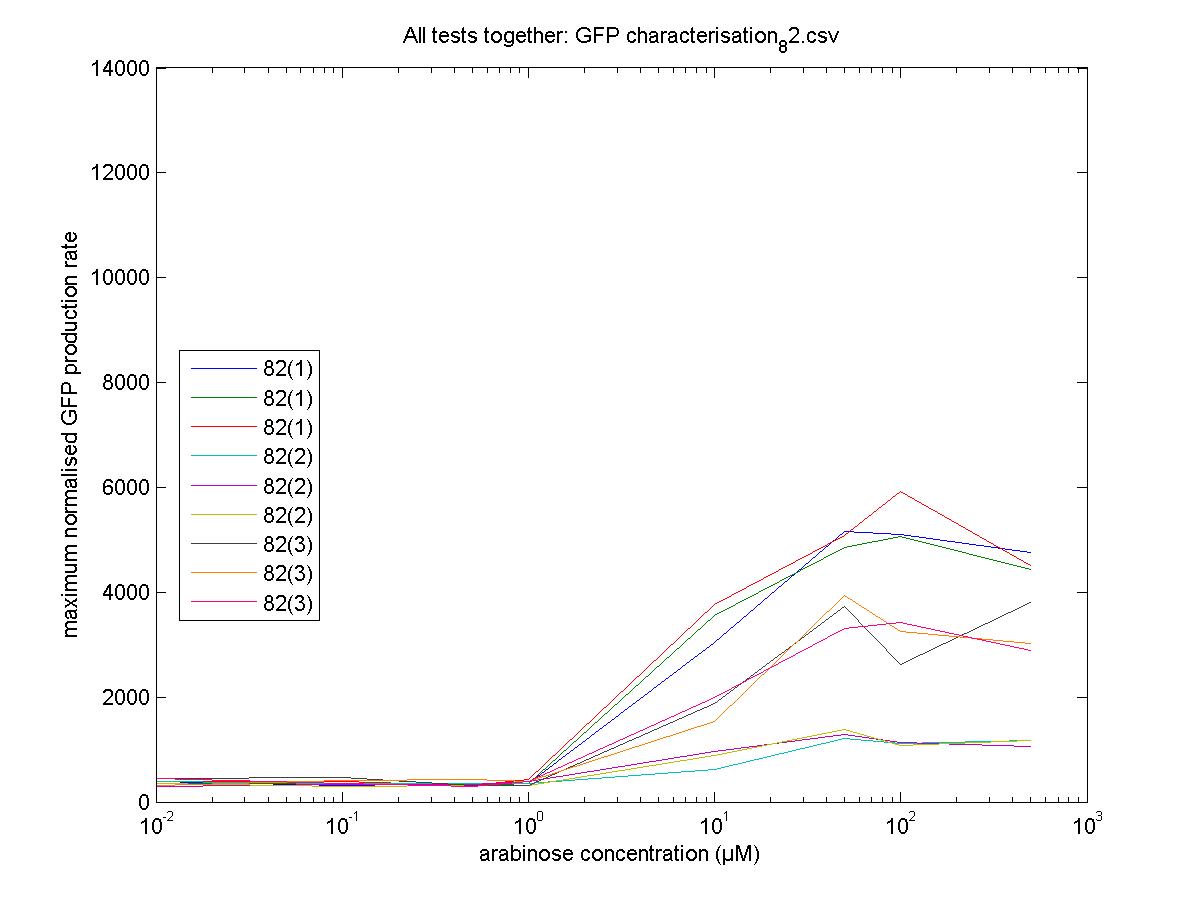
Characterisation
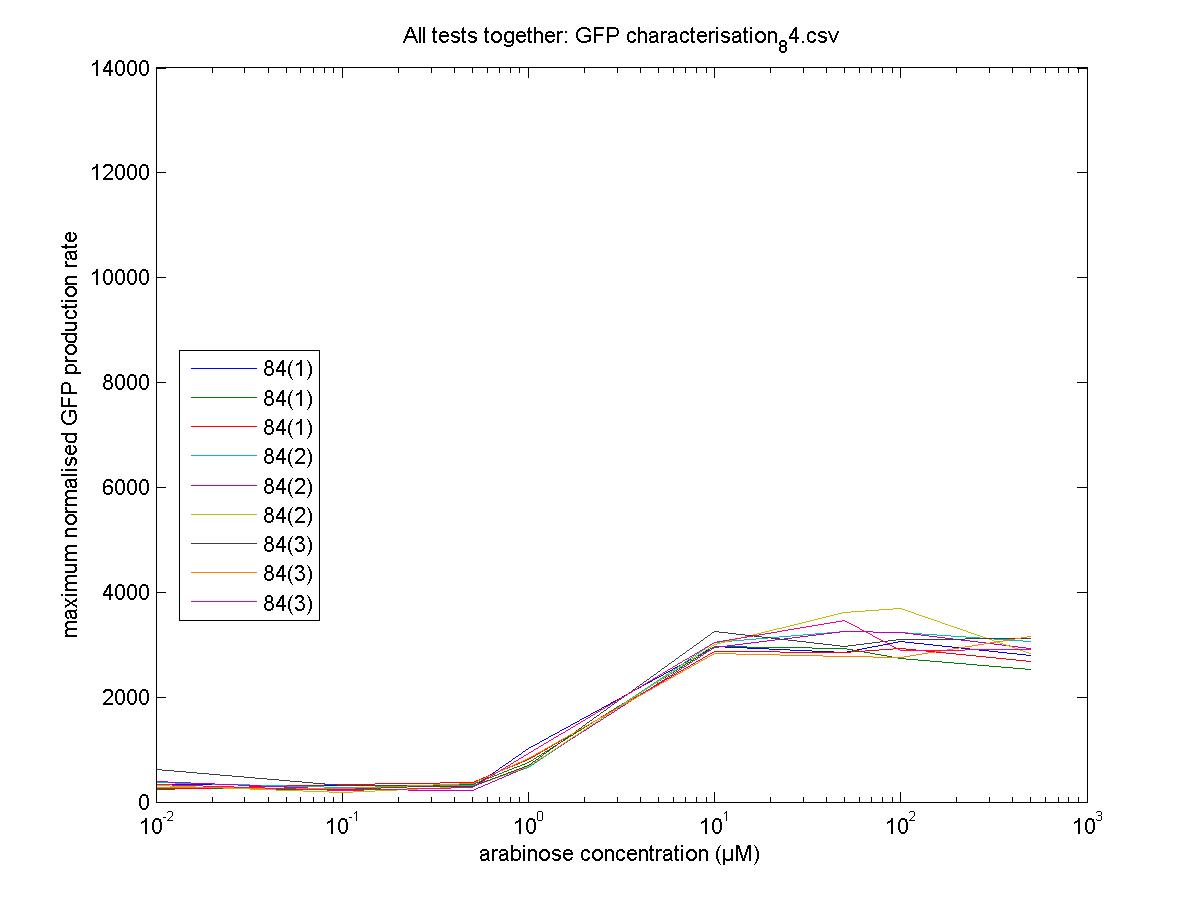
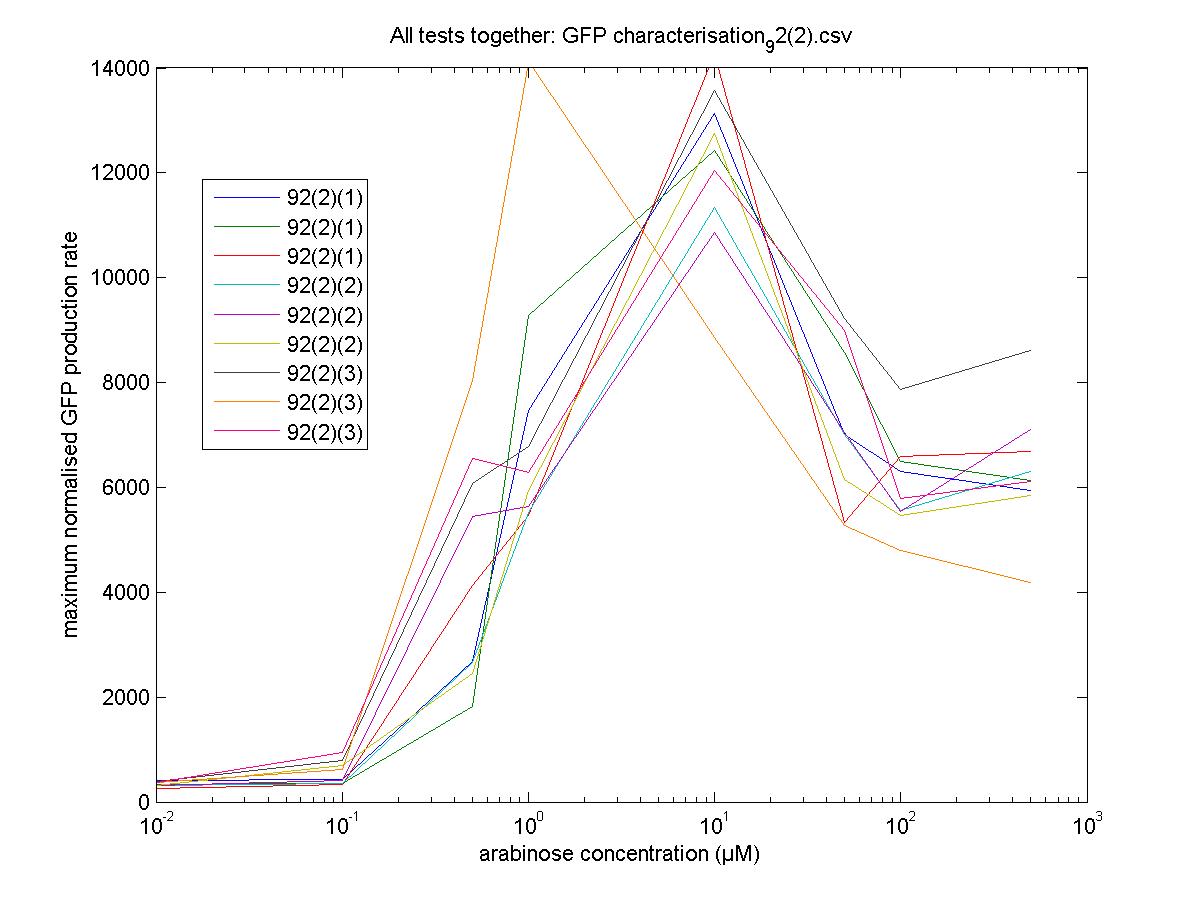
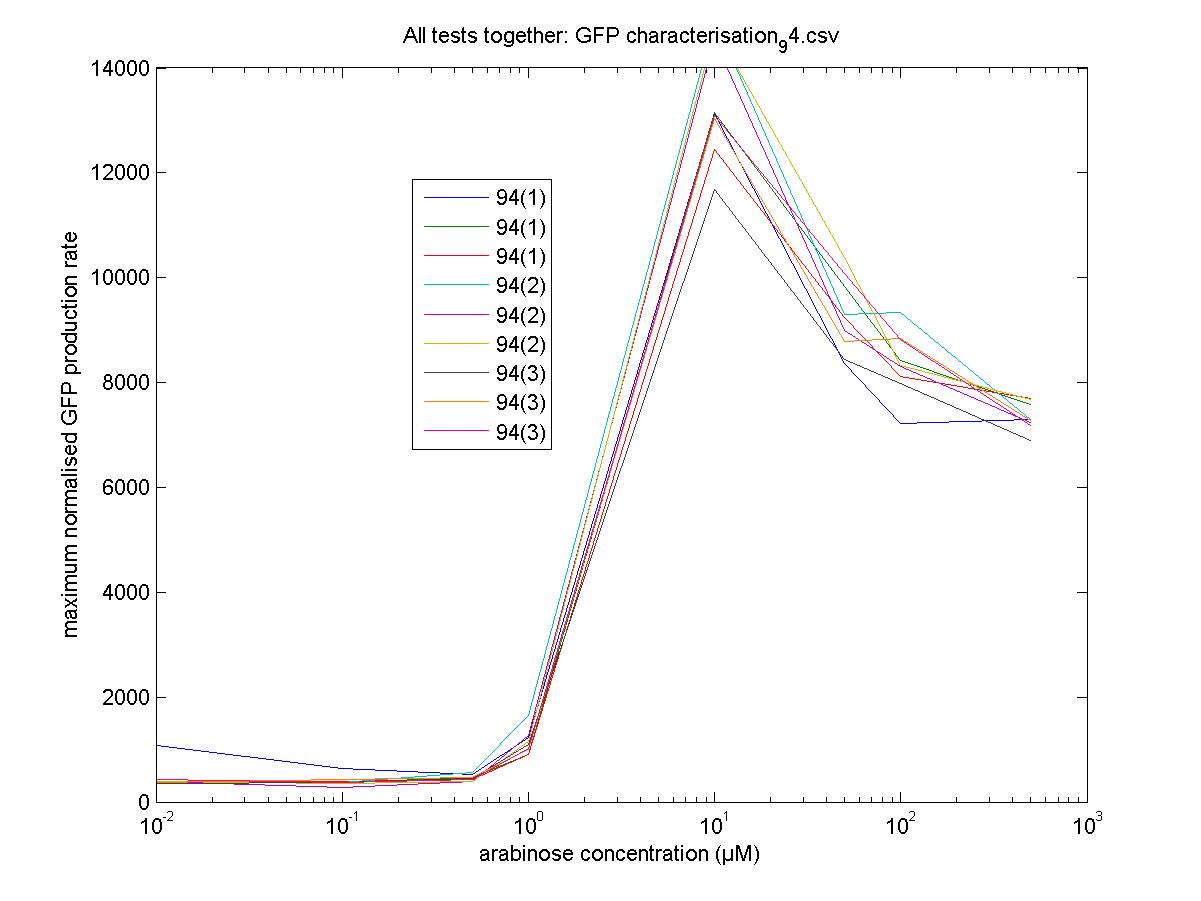
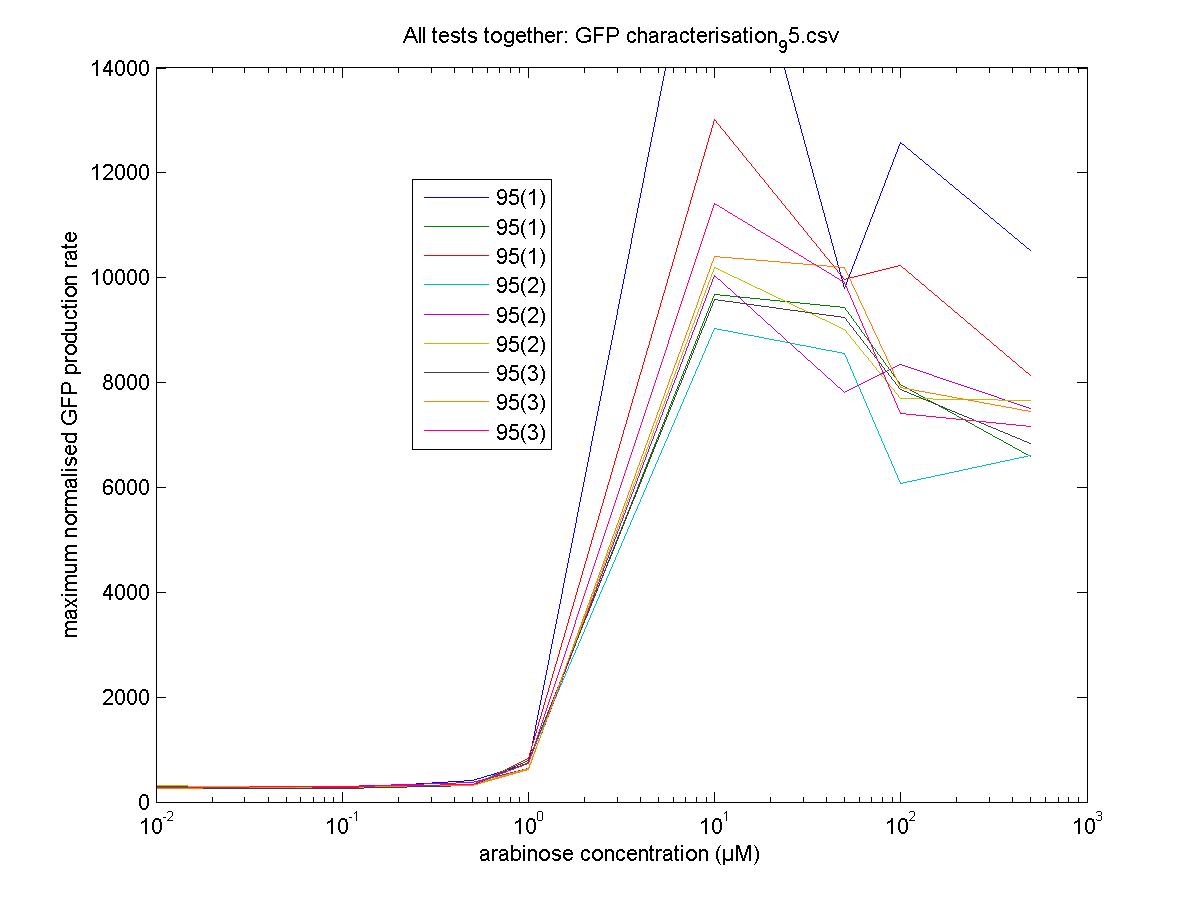
We moved all 15 activator constructs onto pSB3K3, a low copy plasmid. The standard promoter for 1 RPU, J69591, is also on pSB3K3 and has a GFP reporter, so we can make meaningful comparisons on the plate reader.
Maximum Rates against Arabinose Concentrations
80
81
82
84
85
90
92
94
95
Reconstruction
Below is a table listing the PoPS converter
 "
"