Team:Groningen/Project/Accumulation
From 2009.igem.org
(→Zinc) |
(→Alternatives) |
||
| Line 93: | Line 93: | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
==Modelling== | ==Modelling== | ||
Revision as of 15:28, 21 October 2009
[http://2009.igem.org/Team:Groningen http://2009.igem.org/wiki/images/f/f1/Igemhomelogo.png]
|
|---|
- Transport
- Accumulation
- Metal-sensitive Promoters
- Gas Vesicles
AccumulationOnce heavy metals have entered the cell, it is crucial to keep them there. As these metals are toxic to cell survival in critical amounts, evolution has provided us with biological detoxicification proteins such as [http://en.wikipedia.org/wiki/Metallothionein metallothioneins]. These proteins can aid us in our quest to accumulate a variety of heavy metals as they bind to a wide range of metals including cadmium, zinc, mercury, copper, arsenic, silver, etc..
|
Metallothioneins
Metallothioneins are a class of low molecular-weight metal-binding proteins (<10kDa) rich in cysteines residues(~30%). The contain a conserved cys-x-cys or cys-x-his motif which coordinates metal binding, as can be seen in figure 1. They are capable of binding a variety of heavy metals (e.g. Zn, Cu, Cd, Hg, As) with high avidity (Kb), they are in vivo used as a defense against oxidative stress by chelating metals. This proteins do also have a function in storing, detoxify and distributing metals throughout the cell (Merrifield 2004, Gold 2008). These proteins have readily been used to create cell based systems for purification of contaminated water (Chen 1998, Brady 1994). In addition to their wide application possibilities, they also have the capacity to carry multiple metal ions at one time, in contrast to some other metalloproteins that carry them one-on-one (Chang 1998). Many forms of metallothioneins are known and their affinity for different metals has been investigated on several occasions, such as for cadmium (Deng 2007), arsenic (Ngu 2006, Kostal 2004, Singh 2008), mercury (Chen 1998, Chen 1997-2, Deng 2008), nickel (Deng 2003) or a combination of metals (Chang 1998, Kao 2008). Metal-protein complexes can be quantified using a fluorescent molecule (Cadosch 2008) but Cu(I) binding to metallothioneins in metal thiolates, was shown to cause a concentration dependant increase in luminescence. These Cu(I) binding metallothioneins were shown to give rise to a Stokes shift of approximately 300nm upon excitation at 280nm (Beltramini 1981, Gold 2008).
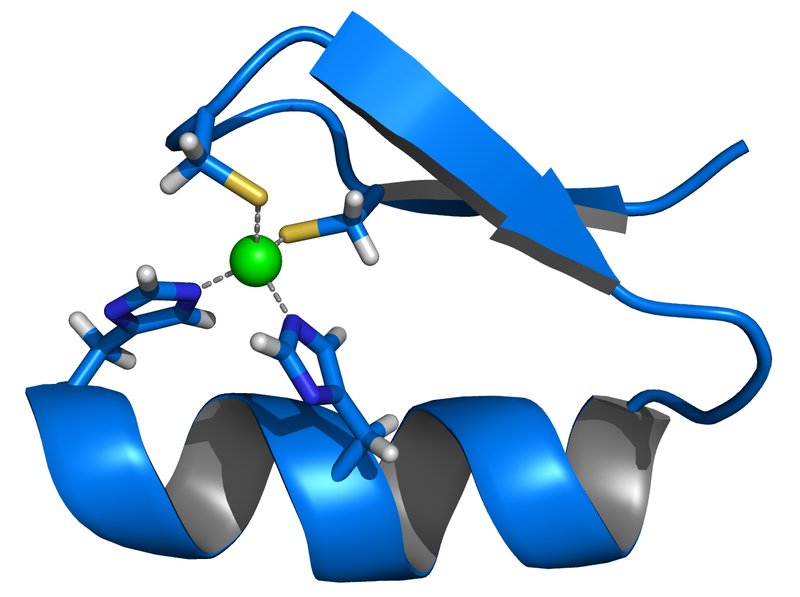
- Figure 1: Zinc finger protein, consisting of a α-helix and an anti-parallel β-sheet. The zinc atom (green) is bound by two histidines and two cysteins.
Cloning strategy
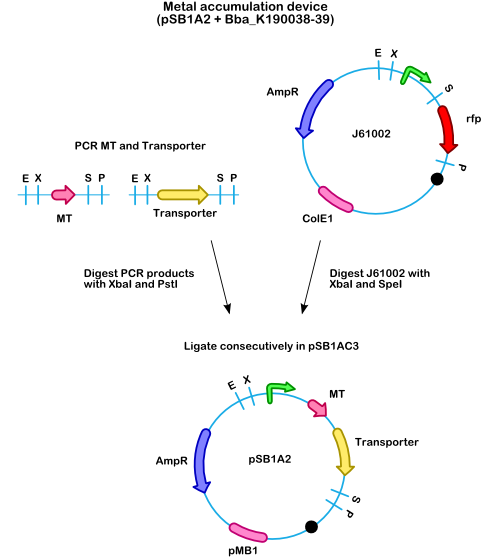
In order to have a functional accumulation device, the cDNA of a metallothionein (MT) will be amplified using [http://en.wikipedia.org/wiki/PCR PCR] and cloned into , also a corresponding metal-ion transporter was amplified by PCR and cloned behind the MT. Both will expressed by one promoter (constitutive or lactose inducible). In this way the bacterium will take up the metal-ion and consecutively the metal-ion will be sequestered by the MT. When this device is combined with the floating device, the bacteria will start floating when a certain threshold of intracellular metal concentration is reached, because the negative regulator of the buoyancy device will be released and the gas vesicle cluster can be transcribed.
- Figure 2: Cloning strategy for the metal accumulation device. A promoter taken from will be cloned in front of a metallothionein and a metal transporter in a vector. This device will be combined with the floating device.
Practical note
MTs are degraded intracellular inside lysozymes, especially when they are in the apo/non-bound state (Gold 2008), for bacteria the degradation rate is not known, but for mammalian MT this can be estimated around 0.8nmol apo-MT/mg protein/min (Klaassen 1994). This can be avoided by adding metal-salts (ZnCl, CuCl) to cells expressing the protein.
Metals
Arsenic
For the accumulation of arsenic some MTs are possible, like rh-MT (human MT) (Ngu 2006) and fMT (the seaweed species Fucus vesiculosis) both binding As(III). The oxidized version of arsenic (As (V)) can also be bound by the metallothioneins but with lower affinity (Singh 2008), another way As(V) is proposed to be accumulated is by conversion of As(V) to As(III) by the arsenate reductase and subsequent bound to the metallothionein or ArsR. rh-MT is known to bind 6x As(III) per molecule, fMT binds 5x As(III). No extra quantitative information is known from literature.
ArsR
ArsR is a trans-acting repressor that senses environmental As(III)and regulates the chromosomal ars operon. The ArsR protein has a specific binding site for As(III) and discriminates effectively against other metals like: phosphate, cadmium, sulfate and cobalt. The affinity of ArsR for As(III) is very high 10-15M of AS(III) can induce the promotor. The specific binding site spans 33 nucleotides in the promotor region including the putative -35 promotor element. When ArsR was purified, its size corresponded to that of a homodimer, bound to promoter DNA. Because of the high affinity of ArsR for As(III) the protein could be used for arsenic remediation. Chen and co-workers overexpressed ArsR in E. coli JM109 cells and found that the specific AS(III) content was 13-fold higher than the control without ArsR expression. High level expression of ArsR appeared to be toxic as a 3-fold reduction in cell density was observed. It has been shown that fusion partners reduce the toxicity of overexpression. Originally, Chen and co-workers made a fusion between ArsR and ELP (elastin protein), which is build out of VPGVG repeats. Because making a ArsR ELP153 fusion is very time consuming, we choose to make a fusion between MBP (maltose binding protein) and ArsR (Chen 1998).
Also see the sensitive promoters.
As ordering rh-MT was not successful, we try to use fMT for accumulation of As(III) and use ArsR to regulate the expression of the GVP cluster behind the ArsR regulated promoter.
Results
The fusion protein MBP-ArsR was built by creating giving the reverse primer of the MBP and the Forward primer of the ArsR a mutual restriction site SacI. The linker region was designed in such a way that it contained a Tev cleavage site, containing a SacI restriction site and a string of alanine residues to facilitate folding. The fusion protein has been succesfully cloned into the psb1AC3 vector creating biobrick [http://partsregistry.org/Part:BBa_K190027 BBa_K190027], but further attempts to add a promotor and rbs failed. Due to time the MBP-ArsR fusion protein has not been equipped with a promotor and so overexpression could not be established.
fMT
The Fucus [http://www.bioc.uzh.ch/mtpage/intro.html Metallothionein] (fMT) was isolated from the [http://en.wikipedia.org/wiki/Seaweed macroalgae] [http://en.wikipedia.org/wiki/Fucus_vesiculosus Fucus vesiculosus ](Morris 1999). It consists of 67 amino acid residues and has 16 cysteine residues, a high cysteine content is a key feature of MT. Another characteristic is the lack of aromatic residues is also seen in fMT where it only has one, tryptophan. Two domains containing cysteine residues are presumed to be involved in the metal binding function. Unusual in fMT is the presence of a 14 amino acid linker region between the two putative metal-binding domains which contains no cysteine residues. Plant MTs show this feature with about 40 residues, where vertebrate MTs only have three residues (Morris 1999). Being a MT fMT binds a multitude of metal ions, 6 Cd2+ ions or 5 As3+ ions in a sequential order, facilitated by the elongated linker domain BUSY
Results
Arsenite uptake assays were done to determine the As(III) accumulation of E. coli WT and fMT / GlpF overexpression strains. The concentration was measured by ICP-MS.
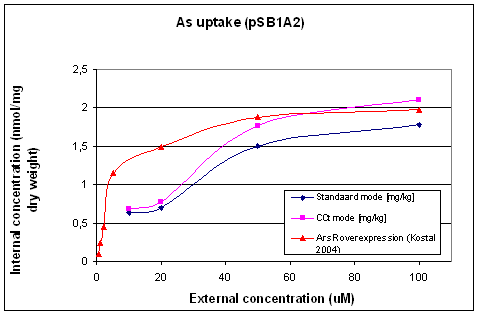
The arsenic uptake in E. coli WT (figure 3) as measured during this project (by [http://www.rikilt.wur.nl/NL/ RIKILT], Wageningen University), was compared with the uptake of E. coli with ArsR overexpression (described by Kostal 2004, see figure 3). This shows that the arsenic uptake in E. coli WT behaves similar but has lower final As(III) uptake yield. The difference is about 10% in the standard mode, but a higher extracellular arsenic concentration seems to be needed to saturate the uptake of arsenic in E. coli WT compared to E. coli with ArsR overexpression. This can be seen by comparing the transition point to saturation in figure 3, which are respectively around 50µM As(III) and around 20µM.
- Figure 3: Uptake of As(III) by E. coli WT (containing pSB1A2-pLac)
There is a relatively large difference between the data generated by measuring the arsenic concentration with ICP-MS in the standard mode and measuring in the collusion cell technology mode (CCT mode). The difference between these two techniques is that in the standard mode it is possible that multi-atomic compounds lead to interference with the arsenic (mw = 75) peak, like argon-chloride (Ar = 40 + Cl = 35 (75%) or 37(25%)). Because 25% of this compound is found in the mw = 77 peak, a correction factor may be calculated to correct for this, the ICP-MS software (Thermo) automatically corrects for Ar-Cl interference. It uses the amount of Krypton and Selenium for this correction. In the CCT all multi-atomic compounds are supposed to be decomposed, therefore no interference will be found in this mode. But a disadvantage of this mode is that the resolution is 10x lower than the standard mode, leading to a smaller signal-to-noise ratio. Because of this, we decided to use the standard mode (corrected for interference) to determine the arsenic accumulation by E. coli.
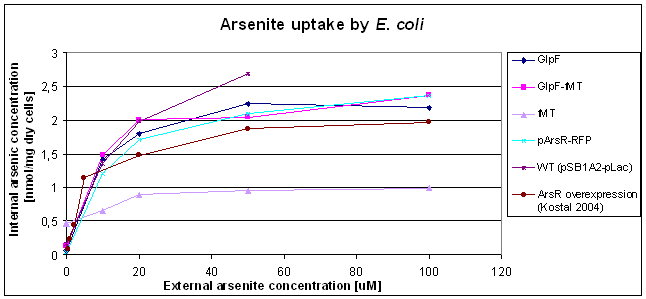
A second arsenic measurement was performed (by [http://www.vwa.nl/portal/page?_pageid=119,1639634&_dad=portal&_schema=PORTAL Food and Consumer Product Safety Authority], Groningen) using E. coli WT and E. coli containing the [accumulation device] () and the different parts (GlpF () and fMT ()). The data was measured in the standard mode and the calculated arsenic imported by the cells is shown in figure 4.
- Figure 4: Uptake of As(III) by E. coli WT, and the strains containing the different parts of the accumulation device. As a control the arsenic uptake of E. coli with ArsR overexpression (as described by Kostal 2004) is also shown.
The curves in this figure show that there is no difference between the arsenic uptake by E. coli WT and by E. coli plus (parts of) the accumulation device. As a second observation, it can be seen that the uptake of arsenic in measured here is higher than found before (figure 3). A ratio of 2-3x was found for the WT strains (pSB1A2 and pArsR-RFP). These two differences will be discussed below. fMT shows exceptionally low arsenite uptake, this may be caused by incidentally "burning" the already dried cells at ~100;deg&C.
The raw data can be found at downloads.
Discussion
Between the two data sets there are a few differences, first there seems to be no difference between arsenic uptake in WT and E. coli with the accumulation device (or parts of this). Secondly, the data of arsenic uptake by E. coli WT could was not reproducible and the last data set showed a arsenic uptake which was even higher for E. coli WT than the E. coli ArsR overexpression strain.
- Why is there no difference between the E. coli WT and the E. coli with accumulation device?
This can be caused by non-functional expression of one of the genes (fMT or GlpF) or both. For membrane proteins it is known that functional overexpression is harder than for cytoplasmic proteins (Lundstom 2006). This could be tested by doing As(III) uptake/binding experiments with purified proteins, but this requires protein purification which could be facilitated by the addition of a his-tag (not present yet). The function of the transporter can be tested by measuring the uptake in membrane vesicles and that of the accumulation protein can be tested by measuring metal binding for instance by isothermal titration calorimetry. Otherwise the proteins may not be produced at all, this should be tested by protein purification or sds-page. Another possibility is that these proteins cannot be produced by ‘’E. coli’’ at once, though functional expression was already proven by Singh et al. (Singh 2008).
- Non reproducible concentrations of arsenic, imported by E. coli WT, which can be seen as there is a large difference (2-3x) in arsenic uptake determined from the first and the second measurement. All data from the second ICP-MS arsenic determination, were also unexpectedly higher than was found in literature (Kostal 2004, Singh 2008). This discrepancy may be caused by one of the following reasons.
During the second arsenic uptake assay the time between the incubation and washing the cells was decreased to the minimum though during the first assay there was some time for the cells to export the As(III) via there exporter ArsB. This may have caused the lower uptake yield of arsenic in the first data set. Also there was a difference in cell concentration, in the second assay this was 2.5 times higher. It is presumably that with a higher cell concentration the uptake rate is slower but a saturating incubation time (>1hr) might cause that the equilibrium of arsenite concentration in/outside the cell is reached faster. After destruction of the samples of the first data set, the samples did not become a clear solution but a suspension containing white flakes. These were removed by centrifugation, but this seems to indicate incomplete destruction. This was not seen for the second samples, therefore an increased arsenite concentration may be measured as arsenite bound to the white flakes is not measured. It also might be, that during the second arsenite uptake assay the cells were washed less properly causing the concentration to become way higher than the first measurement. A more acidic buffer used for washing the cells is probably more efficient in removing metal ions than the TB74S buffer (pH 7.4), but as this protocol was the same as described by Kostal 2004, this should be a major problem. The expected increase in arsenic concentration should be linear with the external arsenite concentration, but this was not seen (figure 4), a clear saturation curve was seen. A plausible cause is that there was a mistake in the calculations, a correction factor which was forgotten to correct for. Another plausible cause is that the concentration is higher because the measured concentration was for some samples 5 times higher than the calibration range. It might be that linear extrapolation is not correct. This can cause the structural increased arsenic uptake.
- Other considerations:
-Metal buffer interactions, causing a lower free-As(III) concentration surrounding the cell suspension. -Arsenic oxidation in aerobic conditions to As(V), this equilibrium may change over hours, so if the stock solution is enriched with As(V) it may take hours before it is changed to As(III) again. - Binding of other metal ions to the metallothionein causing competition for arsenite binding to fMT. Possible metal ions can be: Copper(I) or other metal ions present in the undefined LB medium. A requiry is that the metal ion should bind stronger or as strong to the MT as arsenite, which binds less strongly to MT than Zn(II) for instance or Cu(I).
Conclusion
TODO
Modelling
Arsenic - ArsR
Below you can calculate how many grams of arsenic will be taken out of the water per cubic meter of cells. This extra weight raises the density of the cell and therefore lowers its capacity for buoyancy. Our preliminary results look very promising. Even under the assumption that the weight of the metal is added to the weight of the cells, without increasing their volume, we could add up to a hundred times the currently computed weight without having a large effect on the required fraction of gas vesicles (it will only go up from about 12.2% to 12.7%).
At this moment we use four different variables:
- Molecular weight of arsenic. Source: [http://en.wikipedia.org/wiki/Arsenic Arsenic page on Wikipedia]
- Millimol arsenic per kg of cell dryweight (note that this is equivalent to nmol/mg). Source: Kostal 2004
- The proportion between the weight of a dry cell and a wet cell. Source: [http://redpoll.pharmacy.ualberta.ca/CCDB/cgi-bin/STAT_NEW.cgi CCDB Database]
- Cell density. Source: see our gas vesicle page.
| As per cell volume = awAs * nAs(III) / Mcell(dry) * Mcelldrywet * rhocell mol As per cell volume = nAs(III) / Mcell(dry) * Mcelldrywet * rhocell |
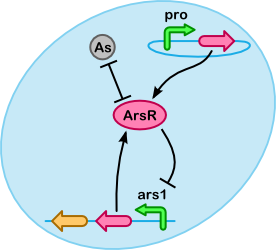
At a lower level arsenic accumulation can be described using reactions between ArsR, As(III) and the ars promoter. As shown in the figure on the right, a number of different substances(/complexes) are involved. For our purposes it is especially important to determine what fraction of As(III) is unbound, if more As(III) is bound we can accumulate more.
In addition to binding to As(III), ArsR can repress Ars, creating a negative feedback loop. In effect this regulates the production of ArsR based on the As(III) concentration (Chen 1997). In the E. coli top 10 there is only ars promoter present on the genome to produce ArsR (see BLAST results). There are plasmids which produce both ArsR and ArsD, but these are not used in this project. We intend to introduce instead a constitutive promoter (pro), which produces just ArsR, in order to bind as much As(III) as possible.
The calculator below tries to compute the ratio between bound and unbound arsenic, specifically As(III), in the cell. See our Modelling page for detailed information on the constants/variables used and a derivation of the formulas. Note that the computations currently involve slightly more variables/constants than strictly necessary.
|
In conclusion:
- Even at the accumulation levels of Koster et al. the amount of arsenic accumulated in E. coli is so little that it should not matter much for the buoyant density (which normally is about 1100kg/m3).
- If you substitute constitutive promotors for Ars promotors, you can see that it is clearly advantageous to use constitutive promotors as just adding ars promoters does not increase the accumulation factor. A plasmid containing an ars promoter and (just) a gene coding for ArsR behind it might contain more arsenic, but there would also be more unbound arsenic, increasing the toxicity.
- The model is not very sensitive to different values for K3d (with K3d=1mM the accumulation factor is 248.06 and with K3d=1nM it is 240.57).
- The accumulation factor is greatly affected by the product of the half-life of ArsR and the production rate.
 "
"