Team:KU Seoul/Result
From 2009.igem.org
(→Results) |
(→Results) |
||
| Line 13: | Line 13: | ||
</head> | </head> | ||
</html> | </html> | ||
| - | |||
| - | |||
{|style= "background:#FFCC33;" align="center" | {|style= "background:#FFCC33;" align="center" | ||
| - | |<b>1. Synthetic Circuit - I : pSB3C5/Pars-gfp-Pznt-rfp</b> | + | |== Results == |
| + | |||
| + | <b>1. Synthetic Circuit - I : pSB3C5/Pars-gfp-Pznt-rfp</b> | ||
1)Pznt-rfp | 1)Pznt-rfp | ||
Revision as of 16:37, 21 October 2009

| == Results ==
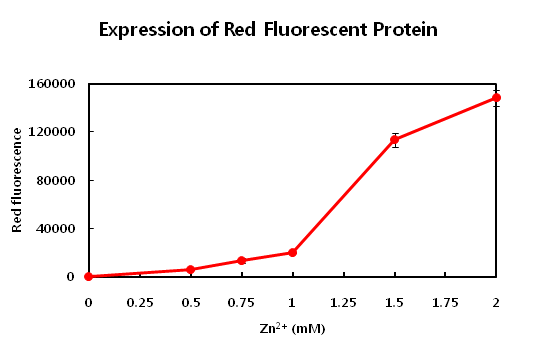
1. Synthetic Circuit - I : pSB3C5/Pars-gfp-Pznt-rfp 1)Pznt-rfp
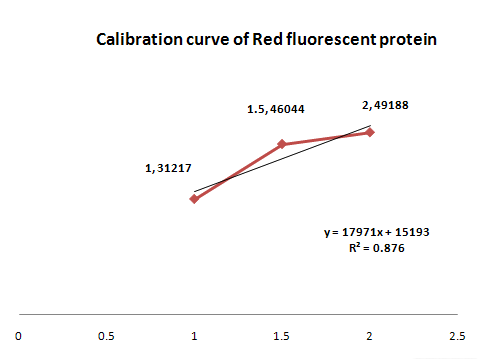
This parts produces red fluorosence based on the zinc ion level. The zinc detecting promoter can be induced beyond 1mM concentration of zinc ion. This range from 1mM to 2mM is enough to detect zinc level in common waste water. We need to detect the upper concentration limit of zinc ion so that we know when dilution is required. If the level curve does not change proportionally according to concentraion, you may have to dilute the concentration of sample. The value of R square is 0.876. Although it is not a satisfying value for confidence, we can make a new formula; Absorbance(fluorsence) = 17971x + 15193. x indicates the concentration of zinc ion(unit:mM). If you have a sample showing specific absorbance, you can calculate the concentration of zinc ion in an indirect way. If you presume that the sample contains large amount of zinc ion, you can try another way to measure the concentration of zinc ion. First, dilute the sample by serial dilution (ex, 10^-1, 10^-2, 10^-3 ...). Second, find the absorbance value between 1,31217 and 2,49188. Then calculate the concentration inversely.
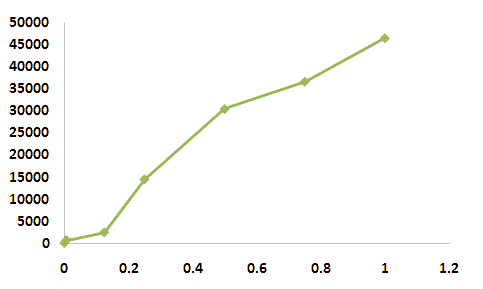
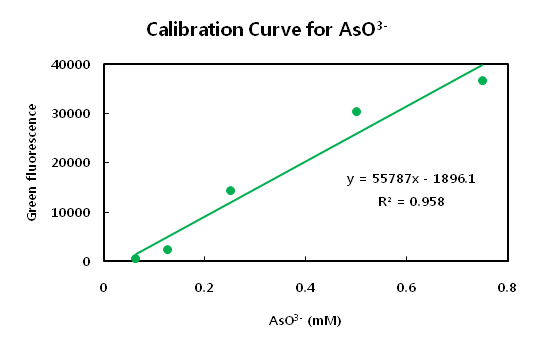
It seems that the the cost of expression of arsenic ion binder is very expensive for microbial cells. In lower levels of concentraion, the fluorosence curve did not show regression curve at the initiation point. Strong inhibitor might exist at the initation of expression state. The main point of issue is to define the possible measure range. Although the overall results show a sigmoid curve, the range from 0.15 to 0.5 mM concentration of arsenic ion shows regression. We should utilize the midpoint of this curve to measure arsenic ions instead of the endpoint. We can also use the dilution method if the sample contains too much amount of arsenic ions. Because the population size of E.coli reflects the intensity of fluorosence, we belive that the hypothesis we have postulated is correct. Compared to zinc ion, the arsenic ion has more harmful effect on the growth of bacteria. This is a drwaback to our detecting system cosnidering that the bacteria can be killed while measuring concentrations. It is hard to distinguish between the effect of concentration of sample or death rate of bacteria.
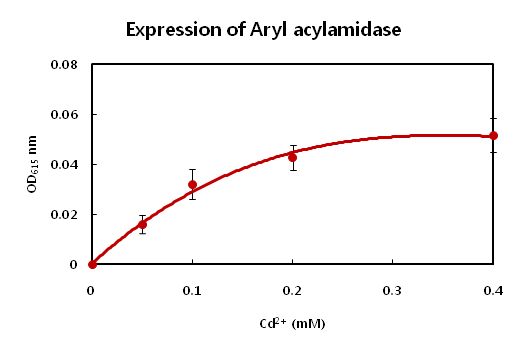
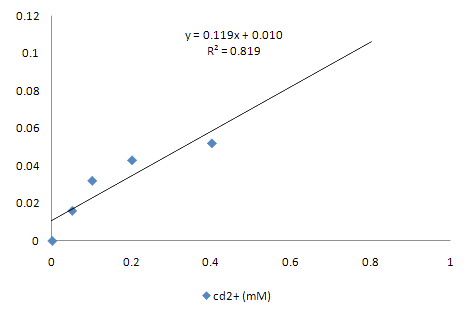
2.Synthetic circuit – II : PyodA-AMD
Cadimum detector has a similar problem related to toxicity, as discussed above. Like arsenic ion, cadmium has harmful effects on bacteria. 0.2mM of concentration of cadimum is enough to inhibt cell growth of E.coli. The E.coli did not show big difference between 0.2mM and 0.4mM concentration of cadimum. We wanted to show the change in concentration with naked eye, but the difference was too subtle to be acknowledged with our eyes.Amd gene, which turns brown in the presence of aceteaminopen, demonstrates only the possiblity. We conisdered using a stronger promoter to detect heavy metals, but we did not use them due to standards issues. Some parts registered on the registry did not work, not to mention inaccurate sequencing. GFP and RFP fluorsence can be seen only with UV irritation. |
 "
"