Team:Cambridge/Project
From 2009.igem.org
(→Project) |
(→Components) |
||
| Line 73: | Line 73: | ||
*'''Sensitivity Tuner''': These constructs are based on Cambridge 2007's amplifiers. The general construct is composed of a gene coding for a phage activator protein and a phage activator-sensitive promoter. With PoPS input, the phage activator gene is transcribed. After translation to produce the phage activator protein, the protein binds to the phage activator-sensitive promoter to activate transcription. Thus it is a device that generates a distinct PoPS out given a PoPS in. With an input-sensitive promoter alone, output generally varies linearly with input--this, at least, is the case with the arabinose-sensitive promoter pBad/Arac (BBa_I0500). However, when a sensitivity tuner is placed downstream of the input-sensitive promoter, the output versus input behavior is altered such that output increases dramatically at a certain input, resulting in sigmoidal behavior with a distinctive threshold. Because there are 3 different activator genes and 5 different activator-sensitive promoters in the registry, 15 combinations are possible, and each has a distinct threshold and peak rate of output. Potential exists to expand this kit of parts further using more phage activators and phage promoters that are not yet in the registry | *'''Sensitivity Tuner''': These constructs are based on Cambridge 2007's amplifiers. The general construct is composed of a gene coding for a phage activator protein and a phage activator-sensitive promoter. With PoPS input, the phage activator gene is transcribed. After translation to produce the phage activator protein, the protein binds to the phage activator-sensitive promoter to activate transcription. Thus it is a device that generates a distinct PoPS out given a PoPS in. With an input-sensitive promoter alone, output generally varies linearly with input--this, at least, is the case with the arabinose-sensitive promoter pBad/Arac (BBa_I0500). However, when a sensitivity tuner is placed downstream of the input-sensitive promoter, the output versus input behavior is altered such that output increases dramatically at a certain input, resulting in sigmoidal behavior with a distinctive threshold. Because there are 3 different activator genes and 5 different activator-sensitive promoters in the registry, 15 combinations are possible, and each has a distinct threshold and peak rate of output. Potential exists to expand this kit of parts further using more phage activators and phage promoters that are not yet in the registry | ||
| - | *'''Colour''': Though ''E. coli'' does not naturally produce pigment, several other bacterial species secrete | + | *'''Colour''': Though ''E. coli'' does not naturally produce pigment, several other bacterial species secrete pigmented antibiotics. We have used pigment-producing operons from other bacterial species and transformed them into ''E. coli''. In particular, we have devoted our summer to 3 different pigment systems: |
:*'''Carotenoids''': The enzymes required for carotenoid production originally come from ''Pantoea ananatis,'' and were available in the registry. We used them to produce orange and red. | :*'''Carotenoids''': The enzymes required for carotenoid production originally come from ''Pantoea ananatis,'' and were available in the registry. We used them to produce orange and red. | ||
:*'''Melanin''': The tyrosinase required for melanin production originally comes from ''Rhizobium etli'' and produces brown. | :*'''Melanin''': The tyrosinase required for melanin production originally comes from ''Rhizobium etli'' and produces brown. | ||
Revision as of 11:31, 20 October 2009
Categories :
Project :
-
Overview
Sensitivity Tuner
--- Characterisation
--- Modelling
Colour Generators
--- Carotenoids (Orange/Red)
--- Melanin (Brown)
--- Violacein (Purple/Green)
The Future
Safety
Notebook :
Team Logistics :
Project
Introduction
Improving Biosensors
The Parts Registry's repertoire of input-sensitive devices is incredibly varied. Teams have engineered E. coli to be sensitive to a wide range of environmentally significant compounds, including arsenic, mercury, lead, cyanide, etc., to genetically engineer biosensors as an alternative to other technologies. The Cambridge 2009 iGEM team identified two stumbling blocks to biosensor design.
- Output: Previous iGEM biosensor projects have used pH, electrical conductance, and fluorescence as output. However, these reporter mechanisms require further steps to read the output. While this is acceptable for First World applications, for biosensors to have true Third World applications, a simpler output is necessary.
- Response to Input: By utilizing an input-sensitive promoter, the biosensor is limited by the sensitivity of the promoter. For example, the promoter might be sensitive to input concentrations which have no real world meaning. The promoter's sensitivity could be too high, so it reports concentrations below levels of real-world interest. Alternately, the promoter's sensitivity could be too low, so it reports concentrations above those which mark the boundary between "safe" and "dangerous." A second limitation is the the behavior of the PoPS output from the promoter; for example, output may vary linearly with input. This type of response is incompatible with a digital "safe" or "dangerous" output.
Our Solutions
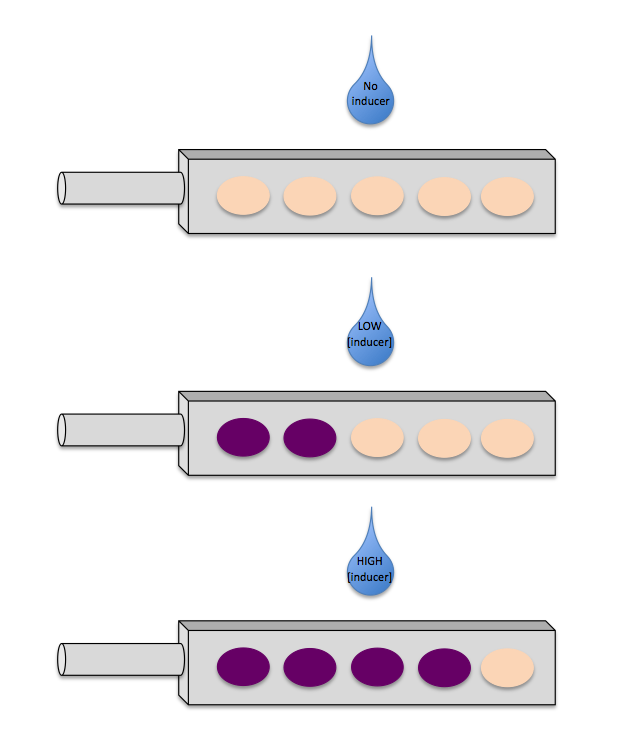
- A Sensitivity Tuner: To avoid being limited to the sensitivity of the promoter and in order to be able to detect distinct concentrations of an inducer using just one promoter, we see the need for a set of sensitivity tuners. These devices allow you to "tune" your biosensor, such that it reports meaningful concentrations of the inducer appropriate to the biosensor's application. The sensitivity tuner also modifies the PoPS output from the promoter's native behavior to a sigmoidal "on" or "off" response pattern.
- Colour Output: What if we could "see" the concentration of an inducer in a sample by a change in colour of the biosensor? Many prey species are brightly coloured, showing off clearly the fact that they are poisonous or otherwise harmful to potential predators, a phenomenon known as aposematism or warning colouration. Humans use colour as a means of conveying information as well - we can "see" if a child has a fever using a thermometer strip and we can "see" the pH of a solution using a pH indicator. Colour can be a meaningful but simple output solution for biosensors, adapting nature's idea of warning colouration.
Project Details
Design
The Product
We envisioned a marketable product that reports the concentration of an inducer by colour. Imagine a dipstick with wells, each of which contain pigment-expressing bacteria in response to an inducer. However, each strain is sensitive to a different concentration of the inducer. The concentration of the inducer in the test solution can be determined by reading the pattern of pigmentation.
The Genetically Engineered Machine
Each bacterial strain is a machine built from a three part system.
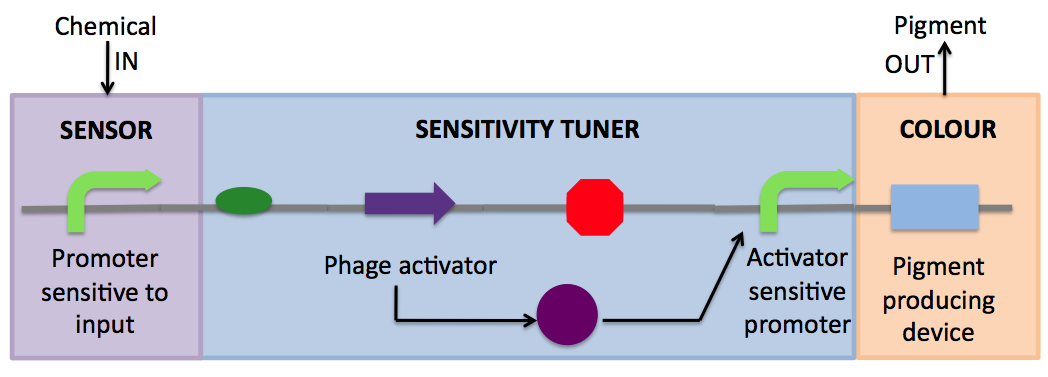
- Sensor: The sensor system is sensitive to different concentrations of an inducer.
- Sensitivity Tuner: This device is responsible for the setting the sensitivity to the inducer, and acts as an "on" switch to activate pigment production once the inducer has reached a threshold.
- Colour: Responsible for pigment production.
Components
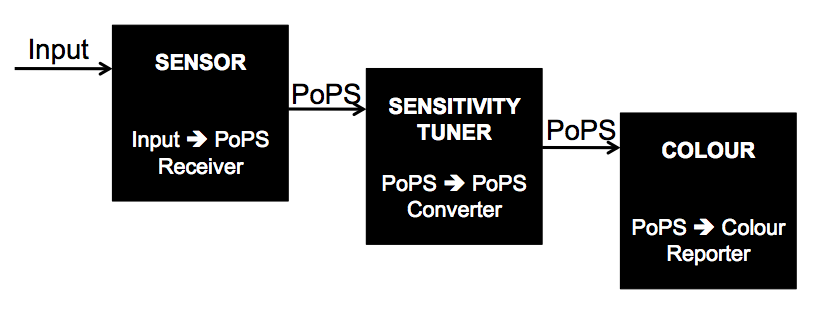
The three part system can be abstracted by the diagram below:
The components of each of the black boxes is as follows:
The sensor, sensitivity tuner, and colour should be viewed as separate parts that are pieced together to build a tunable biosensor that utilizes colour as output. The Parts Registry already has an impressive selection of parts that can be used as sensors. The Cambridge 2009 iGEM team focused on developing a selection of different sensitivity tuner parts and a selection of different pigment producing parts.
- Sensitivity Tuner: These constructs are based on Cambridge 2007's amplifiers. The general construct is composed of a gene coding for a phage activator protein and a phage activator-sensitive promoter. With PoPS input, the phage activator gene is transcribed. After translation to produce the phage activator protein, the protein binds to the phage activator-sensitive promoter to activate transcription. Thus it is a device that generates a distinct PoPS out given a PoPS in. With an input-sensitive promoter alone, output generally varies linearly with input--this, at least, is the case with the arabinose-sensitive promoter pBad/Arac (BBa_I0500). However, when a sensitivity tuner is placed downstream of the input-sensitive promoter, the output versus input behavior is altered such that output increases dramatically at a certain input, resulting in sigmoidal behavior with a distinctive threshold. Because there are 3 different activator genes and 5 different activator-sensitive promoters in the registry, 15 combinations are possible, and each has a distinct threshold and peak rate of output. Potential exists to expand this kit of parts further using more phage activators and phage promoters that are not yet in the registry
- Colour: Though E. coli does not naturally produce pigment, several other bacterial species secrete pigmented antibiotics. We have used pigment-producing operons from other bacterial species and transformed them into E. coli. In particular, we have devoted our summer to 3 different pigment systems:
- Carotenoids: The enzymes required for carotenoid production originally come from Pantoea ananatis, and were available in the registry. We used them to produce orange and red.
- Melanin: The tyrosinase required for melanin production originally comes from Rhizobium etli and produces brown.
- Violacein: The enzymes required for voilacein production originally come from Chromobacterium violacein. The operon can be manipulated to produce voilet, green, and blue.
 "
"