Team:Newcastle/Characterisation
From 2009.igem.org
| Line 2: | Line 2: | ||
{{:Team:Newcastle/Header}} | {{:Team:Newcastle/Header}} | ||
{{:Team:Newcastle/Left}} | {{:Team:Newcastle/Left}} | ||
| + | |||
| + | We successfully characterized IPTG inducable KinA sporulation trigger device. Although Spo0A is the master regulator of sporulation in 'B. subtilis'', only a gradual increase in phosphorylated Spo0A can trigger the sporulation(1). We achieved this scenario by articially inducing KinA by IPTG to participate in a multicomponent phosphorelay. | ||
| + | |||
| + | We used microscopy to verify our results. Our synthesied device was cloned into pgfp-rrnb integration vector. By placing our device just before the Gfp CDS in the integration vector, we hoped to see GFP when we induce teh device with IPTG. However for IPTG to work we needed LacI expressed in the cells. | ||
| + | |||
| + | When we first did our transformation for B. subtilis we forgot this link and we got similar results with cells induced with IPTG and not induced with IPTG. Obviously LacI was not present in the cells and adding IPTG did not make any difference. | ||
| + | |||
| + | We then used a ''Bacillus subtilis'' mutant, BFS687, which has pmutin4 integrated into the chromosomal DNA (2). We selected this mutant especially since it does not have any phenotype. Since pmutin4 has LacI gene, we were able to get LacI expressed in the cells. We successfully transformed the mutant with pgfp-rrnb into AmyE region of B. subtilis with a double crossover. As a result of this crossover, cells loose amyE gene and canot break starch as a result. When transformed colonies are plated into starch plates, the transformed colonies can be seen without any halos around them. | ||
| + | |||
| + | We then selected two transformed colonies from the starch plate and used to induce sporulation by adding IPTG. | ||
| + | |||
| + | To select the transformed colonies we prepared our plates with LB + Erythromycin + Chloramphenicol. Chloramphenicol was used to select pgf-rrnb transformations and Erythromycin was used to select the colonies with pmutin4. Hence by adding two antibiotics we made sure that we were using BFS867 mutants transformed with pgfp-rrnb integration vector. | ||
| + | |||
| + | Concentrations used to characterize our device | ||
| + | |||
| + | IPTG : 1mM | ||
| + | Erythromycin :0.3ug/ml | ||
| + | Chloramphenicol: 5ug/ml | ||
{| | {| | ||
| Line 32: | Line 50: | ||
|} | |} | ||
| + | |||
| + | #Fujita, M. and R. Losick (2005). "Evidence that entry into sporulation in Bacillus subtilis is governed by a gradual increase in the level and activity of the master regulator Spo0A." Genes & Development 19(18): 2236-2244. | ||
| + | # Retrieved 20/10/2009, from http://bacillus.genome.jp/bsorf_mutant_list/Page12.htm | ||
{{:Team:Newcastle/Footer}} | {{:Team:Newcastle/Footer}} | ||
{{:Team:Newcastle/Right}} | {{:Team:Newcastle/Right}} | ||
Revision as of 14:12, 20 October 2009
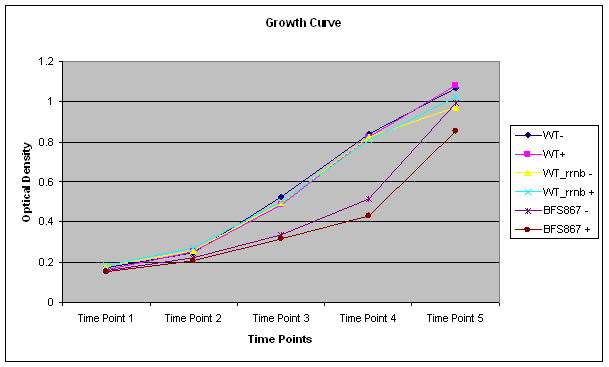
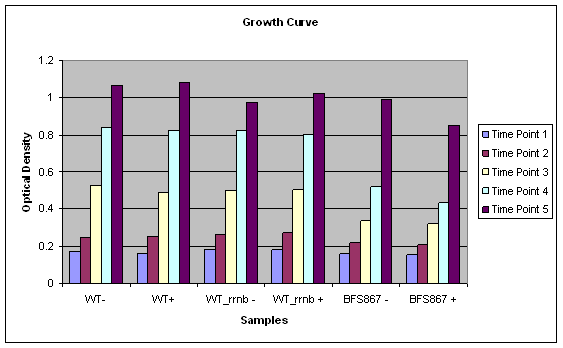
We successfully characterized IPTG inducable KinA sporulation trigger device. Although Spo0A is the master regulator of sporulation in 'B. subtilis, only a gradual increase in phosphorylated Spo0A can trigger the sporulation(1). We achieved this scenario by articially inducing KinA by IPTG to participate in a multicomponent phosphorelay.
We used microscopy to verify our results. Our synthesied device was cloned into pgfp-rrnb integration vector. By placing our device just before the Gfp CDS in the integration vector, we hoped to see GFP when we induce teh device with IPTG. However for IPTG to work we needed LacI expressed in the cells.
When we first did our transformation for B. subtilis we forgot this link and we got similar results with cells induced with IPTG and not induced with IPTG. Obviously LacI was not present in the cells and adding IPTG did not make any difference.
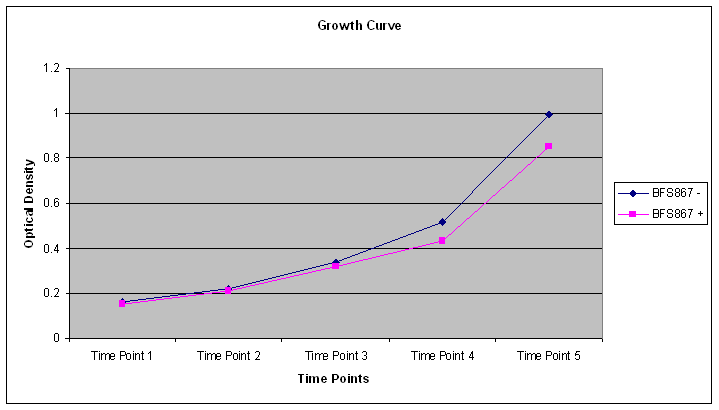
We then used a Bacillus subtilis mutant, BFS687, which has pmutin4 integrated into the chromosomal DNA (2). We selected this mutant especially since it does not have any phenotype. Since pmutin4 has LacI gene, we were able to get LacI expressed in the cells. We successfully transformed the mutant with pgfp-rrnb into AmyE region of B. subtilis with a double crossover. As a result of this crossover, cells loose amyE gene and canot break starch as a result. When transformed colonies are plated into starch plates, the transformed colonies can be seen without any halos around them.
We then selected two transformed colonies from the starch plate and used to induce sporulation by adding IPTG.
To select the transformed colonies we prepared our plates with LB + Erythromycin + Chloramphenicol. Chloramphenicol was used to select pgf-rrnb transformations and Erythromycin was used to select the colonies with pmutin4. Hence by adding two antibiotics we made sure that we were using BFS867 mutants transformed with pgfp-rrnb integration vector.
Concentrations used to characterize our device
IPTG : 1mM Erythromycin :0.3ug/ml Chloramphenicol: 5ug/ml
| |
| |
| |
- Fujita, M. and R. Losick (2005). "Evidence that entry into sporulation in Bacillus subtilis is governed by a gradual increase in the level and activity of the master regulator Spo0A." Genes & Development 19(18): 2236-2244.
- Retrieved 20/10/2009, from http://bacillus.genome.jp/bsorf_mutant_list/Page12.htm
News
Events
- 20 – 21 June 2009 - Europe workshop (London)
- 23 – 24 June 2009 - UK iGEM meetup (Edinburgh)
- 23 October Practice Presentation (Newcastle)
- 23 October T-shirts are ready
- 27 October Practice Presentation (Sunderland)
- 27 October Poster is ready
- 30 October – 2 November 2009 - Jamboree (Boston)
Social Net
- Newcastle iGEM Twitter
- [http://www.facebook.com/home.php#/group.php?gid=131709337641 Newcastle on Facebook]
- [http://www.youtube.com/user/newcastle2009igem Newcastle Youtube Channel]
 "
"
