Team:Brown/Project HBP
From 2009.igem.org
(→rEV131: High-Affinity Histamine Binding Protein) |
(→rEV131: High-Affinity Histamine Binding Protein) |
||
| Line 40: | Line 40: | ||
Another component of the binding affinity test includes the running of free histamine along the histamine-protein-bound column. We will be able to determine the rate of dissociation of the EV131 based upon the interaction of which histamine the EV131 will competitively bind to: the stationary histamine column or the poured free histamine. We will observe the time the protein takes to dissociate from the column and confirm the dissociation constant for the protein described in the literature. | Another component of the binding affinity test includes the running of free histamine along the histamine-protein-bound column. We will be able to determine the rate of dissociation of the EV131 based upon the interaction of which histamine the EV131 will competitively bind to: the stationary histamine column or the poured free histamine. We will observe the time the protein takes to dissociate from the column and confirm the dissociation constant for the protein described in the literature. | ||
| + | |||
''Test 2: Testing EV131 with Histamine Competitor Molecules'' | ''Test 2: Testing EV131 with Histamine Competitor Molecules'' | ||
Revision as of 16:23, 21 October 2009
rEV131: High-Affinity Histamine Binding Protein
rEV131 effectively sequesters histamine without disrupting histamine receptors, hence this treatment may prove to be a viable alternative to current antihistamines in combating allergic symptoms without causing negative side effects. rEV131 is a high affinity histamine binding protein secreted by the tick Rhipicephalus appendiculatus. This protein allows the tick to overcome the host’s inflammatory response by sequestering histamine at the site of feeding, outcompeting the host's histamine receptors for the ligand, thereby effectively combating the allergic response. rEV131 has the highest affinity (lowest dissociation constant) among all histamine binding proteins secreted by R. appendiculatus (Paesen, 1999).In our genetic construct, rEV131 will be placed under the OmpC promoter so that histamine binding to our engineered receptor triggers the transcription of rEV131.
Though the protein has been purified, cloned, expressed and well characterized, we will further characterize the protein by conducting binding assays in which vary levels of free histamine to mimic the extracellular fluid in the nasal cavity. We will also demonstrate protein-ligand specificity by exposing the protein to molecules similar to histamine, such as histidine and imidazole. Further research goals include investigating modifications to the EV131 protein that could enhance histamine-binding affinity, including modifying or introducing novel binding pockets and honing protein residues.
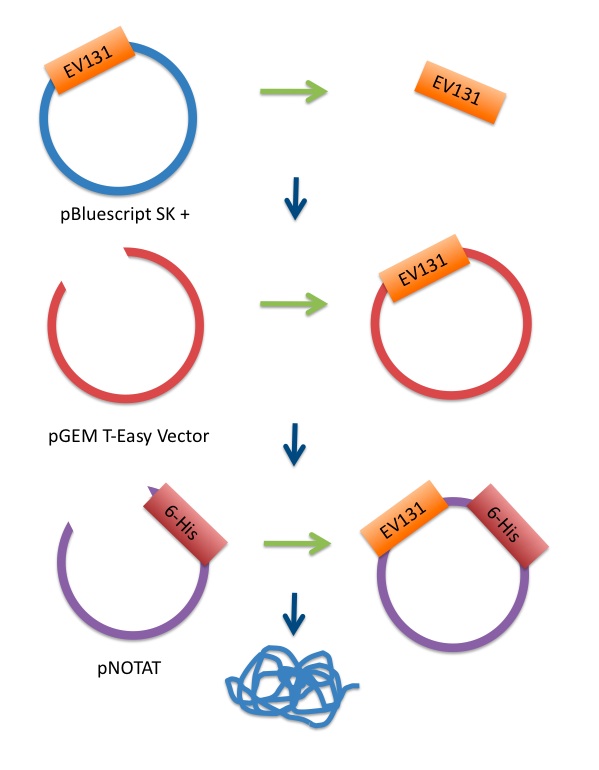
Cloning the EV131 Sequence into an Expression Plasmid, Transformation of E. coli, and Cell Selection
Expression of rEV131 Protein in BL21 E.coli
Efficient and controlled expression of protein in E. coli cells is regulated by the presence of the lac repressor protein. During normal cell growth, this protein binds to the operator sequences in the plasmid and prevents recombinant protein expression. Expression of recombinant proteins encoded by the pNOTAT expression vector is induced by the addition of IPTG, which binds to the lac repressor protein and inactivates it. Once the lac repressor is inactivated, the host cell’s RNA polymerase can transcribe the sequences downstream from the promoter. The transcripts produced are then translated into the recombinant protein. Inducing protein expression with IPTG means that the cellular metabolism concentrates almost exclusively on the production of recombinant protein under tight control. 5-mL liquid cultures of cell colonies isolated from ampicillin plates were grown up and then induced with IPTG. The amount of time allowed for protein expression was be varied in a range from two to five hours for different cultures. Thereafter, we began sample preparations for SDS-PAGE and Nickel Column Chromatography.
Protein Purification Under Native or Denaturing Conditions
In order to retain the biological activity of the protein, we wish to purify 6-His-tagged proteins under native conditions. For this, the 6-His-tagged protein must be soluble. In this case, however, there is greater potential for nonspecific proteins to interact with the Nickel-NTA resin. Also, since there is a chance of the 6-His tag being hidden by the tertiary structure of the native protein, the soluble proteins require denaturation before they can be purified on the Ni- NTA column. As a control, a parallel purification under denaturing conditions will be carried out. If purification is only possible under denaturing conditions, the tag can be made generally accessible by moving it to the opposite terminus of the protein. The QIAgen QIAexpress® Ni-NTA Fast Start Kit is an appropriate kit for the purification and detection of recombinant 6-His-tagged proteins in high yield.
Histamine and Competing Molecule Binding Assays
Test 1: Testing for Histamine Binding Affinity
As a model of the free histamine-EV131 interaction, we will be testing the binding affinity strength of a free poured solution of EV131 being poured into a column that contains histamine beads. The threshold level for eliciting a significant allergic response is 12 ng/mL. The binding capacity of EV131 is two molecules of histamine for one molecule of protein. We will test the amount of protein necessary to sequester the threshold level of histamine by running protein solutions of varying concentrations along the histamine bead column. We will measure the initial amount of protein added to the column and then measure the remaining solution eluted from the column. This will determine the amount of protein needed to sequester histamine at threshold levels.
Another component of the binding affinity test includes the running of free histamine along the histamine-protein-bound column. We will be able to determine the rate of dissociation of the EV131 based upon the interaction of which histamine the EV131 will competitively bind to: the stationary histamine column or the poured free histamine. We will observe the time the protein takes to dissociate from the column and confirm the dissociation constant for the protein described in the literature.
Test 2: Testing EV131 with Histamine Competitor Molecules
After having investigated the histamine-EV131 interaction, we will test the binding affinity that molecules similar in structure to histamine will have with EV131. Such molecules include histadine, imidazole, and aspartate. The procedure will be similar to that of testing free histamine versus stationary histamine binding with the recombinant EV131 protein. We will pour solutions of free histadine, imidazole, or aspartate into a stationary histamine bead column with EV131 protein bound to it. Thereafter, we will observe how much histamine, imidazole, or aspartate elutes with the protein bound to it. Ideally, the results of this experiment will suggest that EV131 has a much higher binding affinity for histamine than do other molecules of a similar structure.
Further Improvements and Insights Upon EV131 Recombinant Protein
EV131 is a novel protein because it has a high affinity for histamine and it outcompetes histamine receptors located throughout the body for histamine. For these reasons, EV131 has tremendous potential as a therapeutic drug for allergies and other inflammatory diseases. We hope to improve the existing functions of EV131 and to broaden the scope of protein functions by modifying the protein. We could potentially improve the binding affinity of the protein for histamine by increasing the number of binding pockets available, which would optimize the protein to histamine ratio. We could also look at the possibility of constructing a fusion protein that includes cell receptor signals, such as ApoE, that may signal faster uptake and degradation of saturated histamine binding proteins or histamine sensors. This can be accomplished by structurally changing the protein, which will involve single amino acid modifications via site directed mutagenesis. Forthcoming research into these methods will help determine their feasibility and practicality.
 "
"