Team:Newcastle/Metalsensing
From 2009.igem.org
(Difference between revisions)
(→Lab Work done) |
(→Lab Work done) |
||
| Line 88: | Line 88: | ||
|'''[https://2009.igem.org/Team:Newcastle/Labwork/15_September_2009#Metal_Sensor_Team 15th September 2009]''' | |'''[https://2009.igem.org/Team:Newcastle/Labwork/15_September_2009#Metal_Sensor_Team 15th September 2009]''' | ||
|No transformant colonies spotted on plates - either ligations or transformations didn't work. Reattempted ligation and transformation in ''E. coli'' cells. | |No transformant colonies spotted on plates - either ligations or transformations didn't work. Reattempted ligation and transformation in ''E. coli'' cells. | ||
| + | |- | ||
| + | |'''[https://2009.igem.org/Team:Newcastle/Labwork/16_September_2009#Metal_Sensor_Team 16th September 2009]''' | ||
| + | |Transformations failed! Reattempted digesting the ''BBa_J33206'' (with no promoter) and ''cadA'' promoter with ''BamHI'' and ''NheI'' and also reattempted ligations | ||
|- | |- | ||
|} | |} | ||
Revision as of 22:32, 21 October 2009
Metal Sensing
Introduction
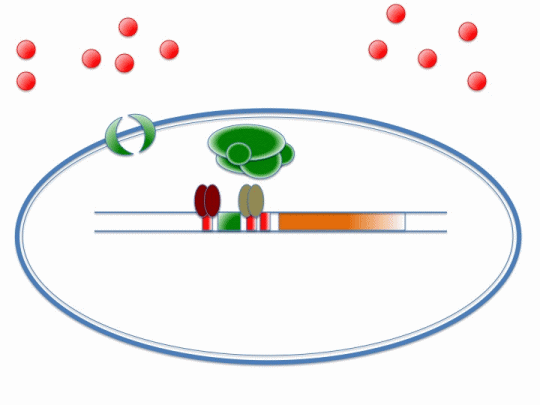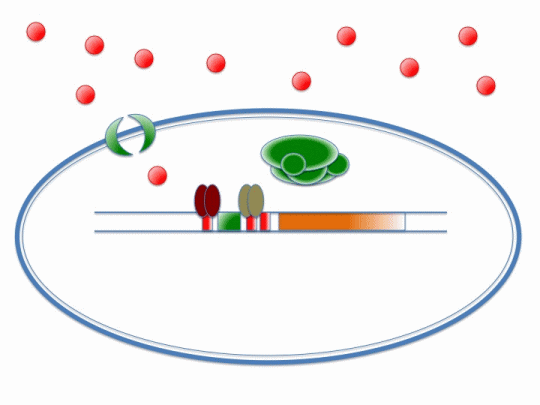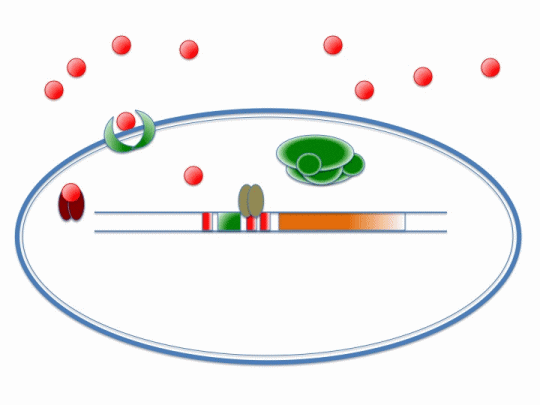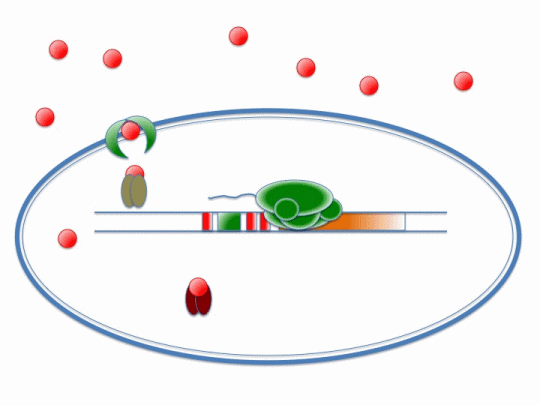
If our project is to process cadmium and not other metals, we need to genetically engineer Bacillus subtilis to carry out a set of cellular processes based on the action of metal sensors. These metal sensors will detect cadmium through a system known as AND Gating.
There are two metal sensing repressors, which are known to respond to cadmium: arsR and czrA. By placing binding sites to these two metal sensing repressors next to each other in a promoter region, the gene regulated by that promoter will be synthesized only when a combination of metals that bind to both sensors are present; this is a combinatorial approach for gene expression regulation.
Modelling
BioBrick constructs
Lab Work Strategies
Other Presentations and Diagrams
Lab Work done
| Summary of Lab Sessions for Cadmium Sensing | |
|---|---|
| | |
| 18th August 2009 | Transformed DH5-alpha E. coli cells with BBa_J33206 from the Spring Distribution |
| 19th August 2009 | Inoculated 3 tubes of LB with 3 colonies of potential transformant E.coli cells |
| 20th August 2009 | Conduct mini-preps on the three overnight-grown cultures of potential BBa_J33206 transformants and digested them with restriction enzymes. |
| 21st August 2009 | Analysed digested BBa_J33206 mini-prep DNA by DNA gel electrophoresis and prepared midi-preps also. |
| 25th August 2009 | Concentrated (and ethanol precipitated) the BBa_J33206 Midi-prep sample. |
| 26th August 2009 | Digested BBa_J33206 midi-prep DNA with EcoRI and PstI and analysed through DNA gel electrophoresis - digest reaction not successful (a second attempt at digests needed) |
| 27th August 2009 | Digested pSB1A2 (containing BBa_J33206 BioBrick), ran DNA though gel and excised band. Also analysed digested pSB1A2 (BBa_J33206 BioBrick) in gel - bands erroneous |
| 28th August 2009 | Cleaned gel band using Gel extraction kit |
| 1st September 2009 | Attempted to PCR amplify the needed czrA gene from the genome of Bacillus subtilis |
| 2nd September 2009 | PCR amplification of czrA gene failed - conducted B. subtilis genome prep and carried out PCR reaction on this DNA |
| 3rd September 2009 | Second attempt at czrA PCR amplification analysed on gel - also unsuccessful. Reattempted PCR amplification of czrA on B. subtilis genomic DNA at different annealing temperatures. |
| 4th September 2009 | In light of 27/08/09 lab session (i.e. anomalous bands with digested BBa_J33206 BioBrick), we have sent away the BioBrick for sequencing and are now using BBa_J33206 sent to us by Chris French. Used Chris's BBa_J33206 to transform E. coli cells. Also carried out PCR reactions |
| 7th September 2009 | Inoculated LB media with colonies of BBa_J33206 (sent from Chris French) E. coli transformants for mini preps. By afternoon, cultures had grown sufficiently to further inoculate flasks of 50ml LB + amp for midi-preps. Mini-prep attempted but abandoned |
| 8th September 2009 | Skipped mini-prep re-attempt and immediately carried out midi-prep of BBa_J33206 BioBrick sent by Chris French. Digested sample with EcoRI and PstI and analysed through gel. Successful! |
| 10th September 2009 | PCR amplification of the cadA promoter and BBa_J33206 BioBrick (missing promoter) |
| 11th September 2009 | Gel analysis shows PCR reactions worked. Tried to proceed with running the cleaned-up PCR products through gel to excise band but ethanol presence stopped us. Will have to reattempt PCR reactions and other subsequent steps. |
| 14th September 2009 | Both cadA promoter and BBa_J33206 (with promoter missing) PCR reactions worked! Subsequently excised from gel and cleaned up. Both fragments cut with BamHI and NheI and ligated together. Attempted to transform E. coli with ligated cadmium-sensor. |
| 15th September 2009 | No transformant colonies spotted on plates - either ligations or transformations didn't work. Reattempted ligation and transformation in E. coli cells. |
| 16th September 2009 | Transformations failed! Reattempted digesting the BBa_J33206 (with no promoter) and cadA promoter with BamHI and NheI and also reattempted ligations |
News
Events
- 20 – 21 June 2009 - Europe workshop (London)
- 23 – 24 June 2009 - UK iGEM meetup (Edinburgh)
- 23 October Practice Presentation (Newcastle)
- 23 October T-shirts are ready
- 27 October Practice Presentation (Sunderland)
- 27 October Poster is ready
- 30 October – 2 November 2009 - Jamboree (Boston)
Social Net
- Newcastle iGEM Twitter
- [http://www.facebook.com/home.php#/group.php?gid=131709337641 Newcastle on Facebook]
- [http://www.youtube.com/user/newcastle2009igem Newcastle Youtube Channel]
 "
"