Team:Newcastle/Labwork/11 September 2009
From 2009.igem.org
Babyneurone (Talk | contribs) (→Procedure) |
Babyneurone (Talk | contribs) (→Conclusions) |
||
| Line 149: | Line 149: | ||
===Conclusions=== | ===Conclusions=== | ||
| - | + | ||
<br> | <br> | ||
Today's lab session has been centred around the following list of objectives: | Today's lab session has been centred around the following list of objectives: | ||
Latest revision as of 12:15, 20 August 2010
Formal Lab Session - 11th September 2009
Overview
Metal Sensor Team
Introduction and Outline
So far we have transformed DH5-alpha E. coli cells with two synthesized BioBricks, which are the cotC-GFP-smtA and the kinA BioBricks. The cotC-GFP-smtA BioBrick is involved within the metal sensing sub-project whereas the kinA BioBrick is involved with the Sporulation Team's sub-project. The Metal Sensing team have since taken these transformant colonies and used them to set up LB+kanamycin cultures for mini-preps and midi-preps; these were then left overnight in the orbital shaking incubator and are awaiting processing today. However there are a range of tasks which need to be accomplished today; these are listed below:
- Prepare Mini-preps of the kinA and cotC-GFP-smtA transformants
- Carry out the mini-prep processes
- Analyse the mini-prep DNA by treating with restriction enzymes.
- PCR the cotC-GFP-smtA BioBrick with pMK15 and pMK16
- Set up the PCR
- Run the PCR for the correct duration
- Analyse PCR products through DNA gel electrophoresis
- Clean up PCR products if PCR reaction proves to be successful
- Midi-prep cotC-GFP-smtA, kinA, pGFP-rrnB, pMUTIN4 and pSB1AT3
- Carry out the five midi-preps.
- Quantify the midi-prep samples
- Run the midi-prep samples on agarose gel through DNA gel electrophoresis
- Run products from a PCR reaction involving arsR and cadA DNA on gel
- Run PCR products on gel
- If successful, clean up fragments, run on gel again, cut with restriction enzymes NheI and BamHI, clean and then ligate together fragments.
Procedure
The following procedures were carried out:
Freeze the kinA and cotC-GFP-smtA transformant cells
Instead of preparing mini-prep samples from the two groups of transformant E.coli cells (i.e. kinA transformed E.coli cells and cotC-GFP-smtA transformed cells), we instead took the decision to use the inoculated 5ml LB tubes in freezing the cells. Analysis of the midi-prep samples will be used to determine whether the BioBricks have been truly transformed - this decision will both save time and also provide a back-up stock of cells for future work. This procedure was carried out by James using the protocol
PCR the cotC-GFP-smtA BioBrick with pMK15 and pMK16
Midi-prep cotC-GFP-smtA, kinA, pGFP-rrnB, pMUTIN4 and pSB1AT3
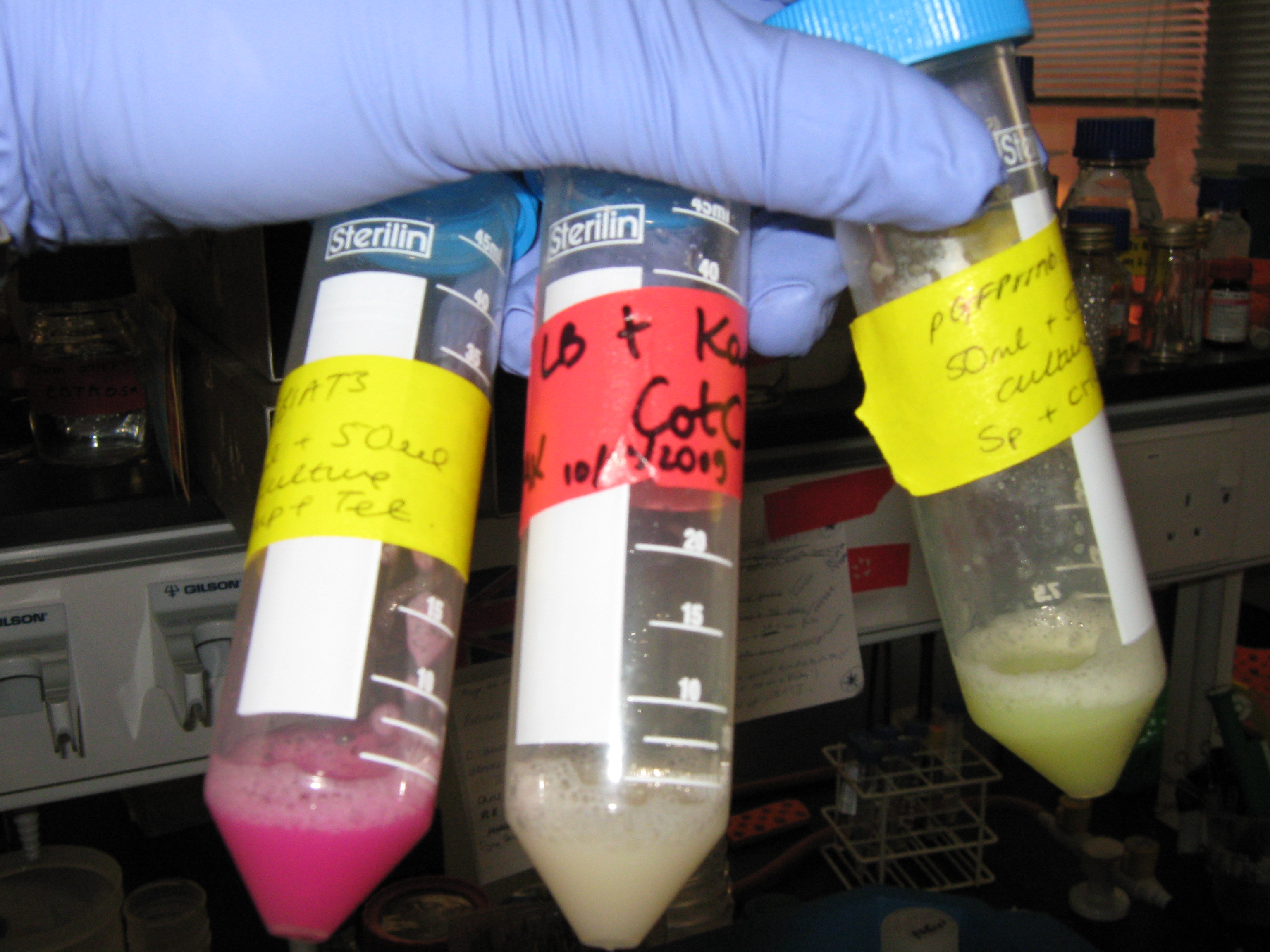
The midi-preps were conducted, as usual, using the Sigma-Aldrich Midi-prep kit and protocol. There were no changes to the protocol and 10ul samples of the five midi-prepped plasmids were loaded onto 0.8% agarose gel (see the 'Results' section below). The midi-prepped DNA samples were also assessed for DNA concentration using the NanoDrop spectrophotometer.
cotC-GFP-smtA
Whilst the plasmid + BioBrick is 3,663bp the plasmid was uncut so it appears as a shorter fragment in the gel. To prove that the midi-prepped DNA is 3,663bp, we should carry out restriction enzyme digests on another day.
kinA
Whilst the plasmid + BioBrick is 4,327bp the plasmid was uncut so it appears as a shorter fragment in the gel. Likewise, to prove that the midi-prepped DNA is 4,327bp, we should carry out restriction enzyme digests on another day.
pGFP-rrnB
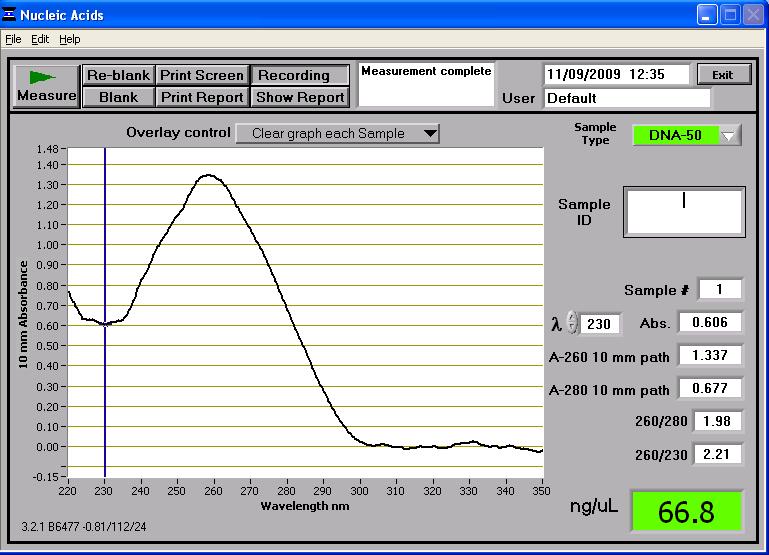
The true length of the plasmid is 8399bp but seen as the plasmid is uncut the gel again doesn't reveal its true length. Like said before, we will need to linearise the fragment with appropriate enzymes to prove it's length.
pMUTIN4
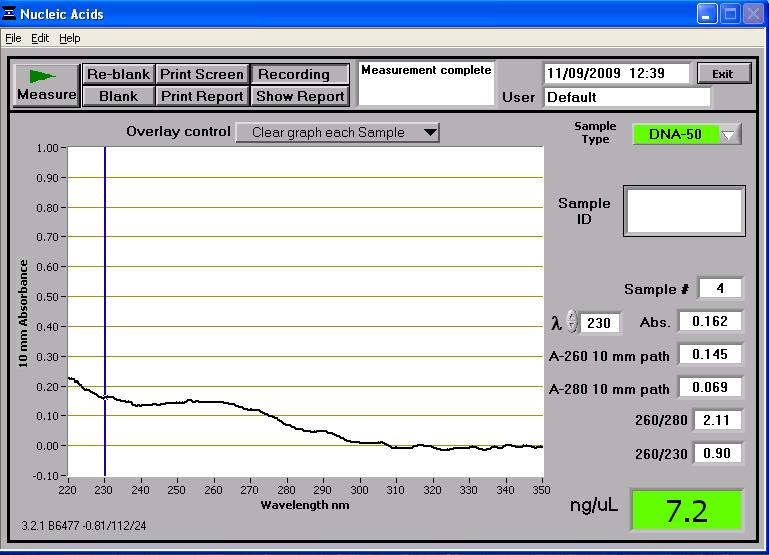
To deal with this problem, a second midi-prep should be attempted of the pMUTIN4 plasmid.
pSB1AT3
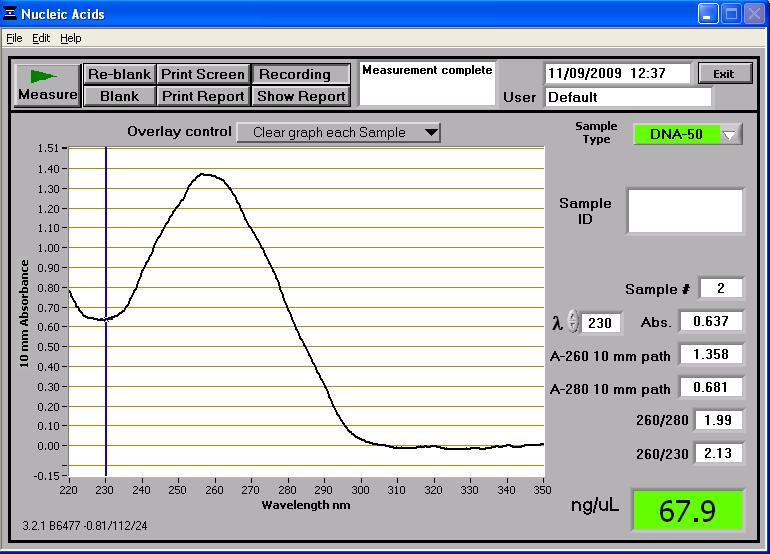
The plasmid size is 3,446bp but seen as the plasmid is uncut the gel again doesn't reveal its true length. Like said before, we will need to linearise the fragment with appropriate enzymes to prove it's length.
Second attempt at pMUTIN4 midi-preps
In a lab session on the 24th July 2009, we took the decision to prepare some E.coli cells containing the pMUTIN4 plasmid by taking 2 flasks of inoculated 100ml LB (inoculated with these E. coli cells and grown overnight in the previous lab session), splitting them into four 50ml falcon tubes and then spinning them down. The supernatants were then decanted and the pellets stored in the -20C freezer. Today, the Metal Sensing team will use these cells to attempt a second midi-prep.
Two of the four tubes were taken and each was treated using the Sigma-Aldrich Midi-prep kit and protocol. There were no changes to the protocol but there are two important notes to make:
- This midi-prep attempt started at step 2 (titled 'Resuspend Cells) in the protocol
- The Spin method was the preferred route (as opposed to the Vacuum method).
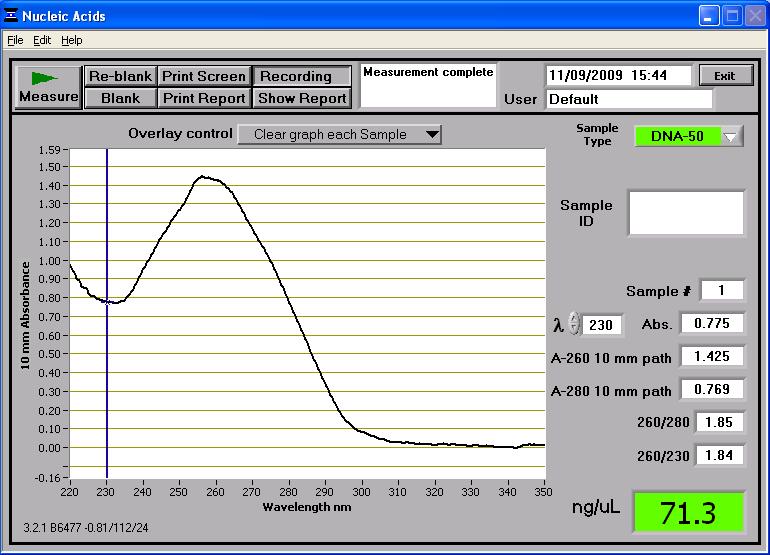
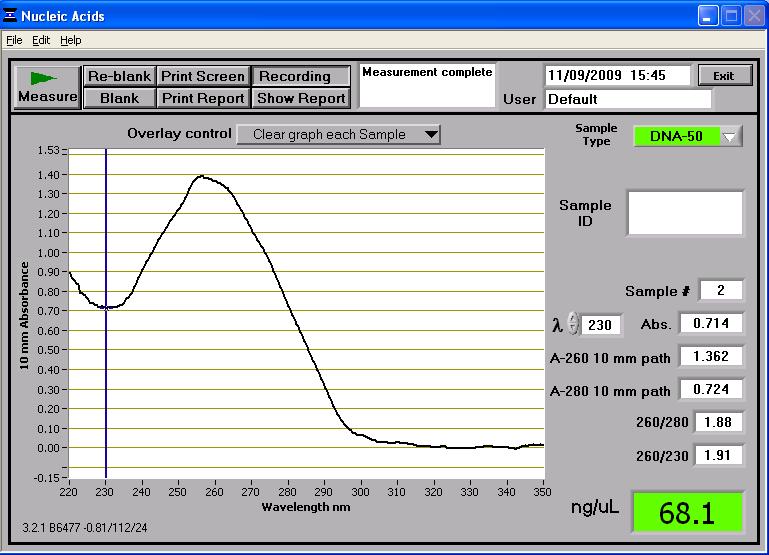
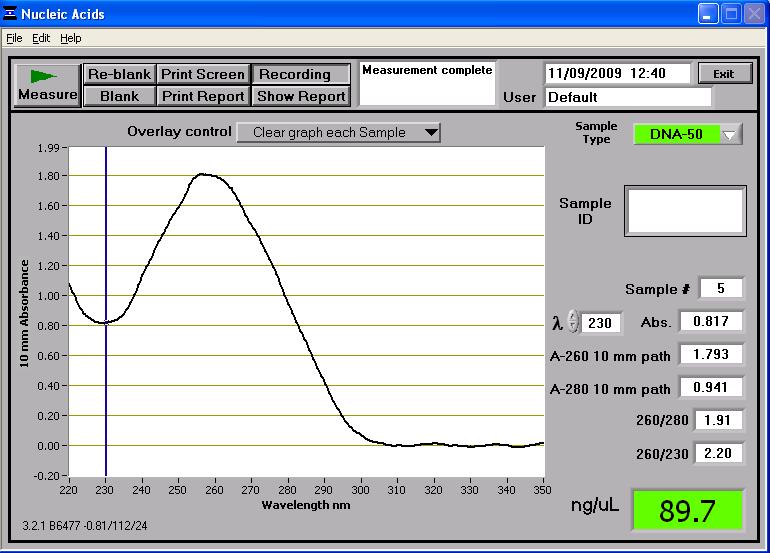
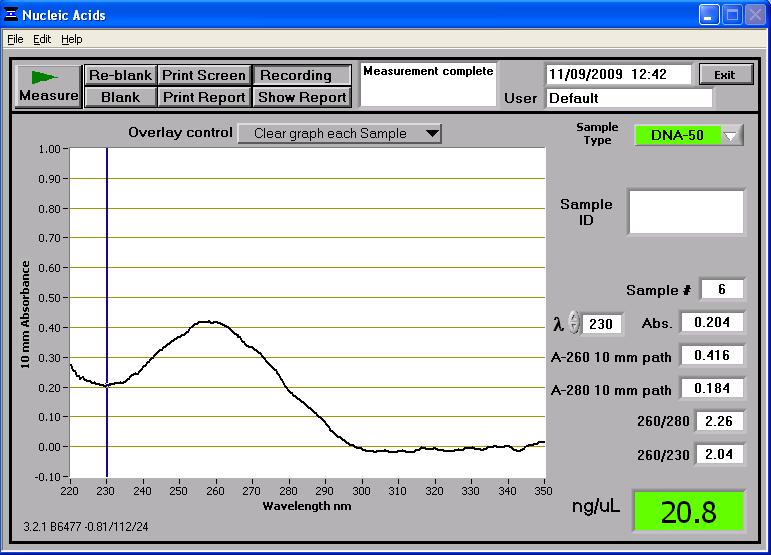
Once two midi-prep samples of pMUTIN4 were successfully prepared from the 2 cultures of E. coli, they were taken to the NanoDrop spectrophotometer. The following readings were produced:
These two midi-prep samples of pMUTIN4 contain a higher concentration of DNA than the first attempt. These concentrations are sufficient for further processing. These two samples were also run on the gel and can be found in lanes 14 and 16 in the first well of rows (see gel photograph in 'Results' section below). Whilst the two samples are found near the 4,361bp band of the HindIII DNA ladder, it has to be remembered that they are uncut so they don't reflect their true length (which is roughly bp) - the gel just shows that there is DNA present there. However the two samples are consistent in size which suggests no contamination.
Process products from a PCR reaction involving arsR BioBrick and cadA Promoter DNA
In the previous lab session, PCR reactions were carried out in an attempt to amplify a promoter-less arsR BioBrick (the BBa_J33206 BioBrick minus the promoter region) and cadA promoter region - these will form the cadmium-sensing AND gate. The plan is to run these products on a gel and if we get results, carry out the following:
- Clean up product
- Run in agarose gel in electrophoresis
- Cut with NheI and BamHI
- Clean digested product
- Ligate cadA promoter region and a promoter-less BBa_J33206 arsR BioBrick (in pSB1A2 plasmid)
The gel photograph produced after the PCR products were run through 0.8% agarose gel showed that the PCR reactions did in fact work. The Metal Sensing team proceded to clean up the two products however not all of the ethanol was removed! This error became apparent when trying to load the gel with the cleaned up product as the sample immediately dissipated into the buffer surrounding the gel.
This means we will have to carry out another PCR reaction to obtain the arsR BioBrick (without promoter) in pSB1A2 backbone and the cadA promoter region; we shall then continue with the steps listed above. The PCR reaction was prepared and initiated in the afternoon so the products should be ready for tomorrow's lab session.
Results
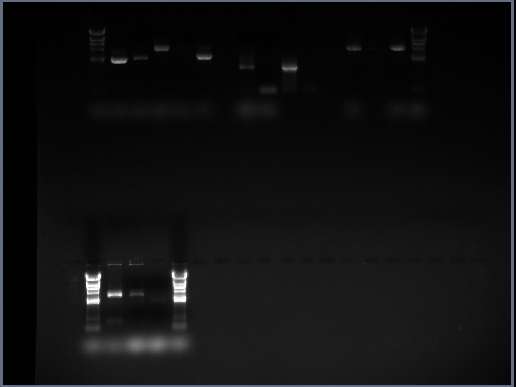
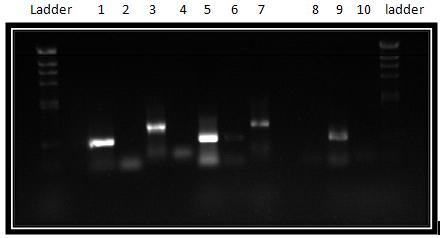
We took the decision to run 4 sets of DNA samples on 0.8% agarose gel - this includes the midi-prep samples (i.e. cotC, kinA, pGFP-rrnB, pMUTIN4 and pSB1AT3), products from a PCR reaction involving cotC, futher midi-preps of pMUTIN4 and digests from midi-prepped DNA the Stochastic Switch team have prepared (i.e. sac, ara and sleB) The resulting GelDoc photograph can be seen below:
The lanes are as follows:
Row 1
- Lane 1 = blank
- Lane 2 = HindIII DNA Ladder
- Lane 3 = [cotC midi-prep sample]
- Lane 4 = [kinA midi-prep sample]
- Lane 5 = [pGFP-rrnB midi-prep sample]
- Lane 6 = [pMUTIN4 midi-prep sample]
- Lane 7 = [pSB1AT3 midi-prep sample]
- Lane 8 = blank
- Lane 9 = cotC PCR reaction 1 sample
- Lane 10 = cotC PCR reaction 2 sample
- Lane 11 = cotC PCR reaction 3 sample
- Lane 12 = cotC PCR reaction 4 sample
- Lane 13 = blank
- Lane 14 = [pMUTIN4 midi-prep sample (from second attempt)]
- Lane 15 = leak-over of sample from lane 14
- Lane 16 = [pMUTIN4 midi-prep sample (from second attempt)]
- Lane 17 = HindIII DNA Ladder
Row 2
- Lane 1 = blank
- Lane 2 = HindIII DNA Ladder
- Lane 3 = sac midi-prep digest sample
- Lane 4 = ara midi-prep digest sample
- Lane 5 = sleB midi-prep sample
- Lane 6 = HindIII DNA ladder
To get an analysis of the bands seen in the wells click on the above hyperlinks highlighted in [light blue].
Conclusions
Today's lab session has been centred around the following list of objectives:
- Mini-prep kinA and cotC-GFP-smtA transformants
- Carry out the mini-prep processes
- Analyse the mini-prep DNA by treating with restriction enzymes.
Froze down kinA and cotC-GFP-smtA transformant cells instead
- PCR the cotC-GFP-smtA BioBrick with pMK15 and pMK16
- Set up the PCR
- Run the PCR for the correct duration
- Analyse PCR products through DNA gel electrophoresis
- Clean up PCR products if PCR reaction proves to be successful
- Carry out the five midi-preps.
- Quantify the midi-prep samples
- Run the midi-prep samples on agarose gel through DNA gel electrophoresis
- Run PCR products on gel
The objectives written in grey coloured text are objectives fulfilled in today's lab session, the objectives written in purple coloured text are objectives which were changed and the objectives left in black coloured text are objectives not achieved.
Tomorrow's lab session will be centred around the following objectives:
- Run products of PCR reaction involving the arsR BioBrick (BBa_J33206 in pSB1A2 missing promoter) and cadA promoter region
- If successful clean up product, run on agarose gel through electrophoresis, excise band and carry out gel extraction.
- Cut cleaned and excised fragment with BamHI and NheI
- Carry out ligation (possibly an overnight ligation)
- Cut pMUTIN4 and cotC (i.e. PCR product 3) with BamHI and HindIII
- Ligate the fragments (possibly overnight)
Stochastic Switch Team
We run the restriction digests stored in the freezer from Wednesday to see we really have the biobrick (sspB + pSB1AT3)
We had the results on the gel as we expected. We saw the bands for the plasmid backbone which were all equal in 11 columns. The 11th column had mCherry + pSB1AT3. mCherry has around 1000b and sspB has around 550bp which can also be confirmed from the gel.
This result meant we had our first manual biobrick! sspB + pSB1AT3 ligation worked and when we cut the ligated product we had the bands as we expected.
Today we did restriction digests using fast digest enzymes of pGFP-rrnb (EcoRI + NheI) pMKRQ+kinA (EcoRI + NheI) and James and Hanny's sleB (EcoRI + NheI). Gel extraction was done on pGFP-rrnb.
Sporulation Tuning/Chassis Team
Introduction
- Today's work is mini prep my single colony culture from yestoday, since jess has the mini prep plan too, she did mini prep for my cells as well.
- For the PCR result of cwlJ, we need to clean up gene before we redo PCR using orignal primers which contain Biobrick prefix and surfix.
- For the mini prep preparation, we picked 10 colonys this time for tomorrow's mini prep.
Experiment procedure
cwlJ PCR clean up
We used Sigma's PCR Clean Up Kit to precess our cwlJ PCR result.
- Follow the standard protocal from PCR Clean Up Kit.
cwlJ PCR with orignal primer
We got new PCR polymorase from another company -- BioTaq. We did PCR with two different Taq to check the reason why our PCR didn't work on 2nd september.
- Prepare buffer mix and Taq
1. For Go Taq
Reaction buffer 10ul x 6 = 60ul
dNTP 2ul x 6 = 12ul
--------------------------------
72ul
12ul for each reaction
GoTaq(5U/ul) 0.25ul x 4 = 1ul
ddH2O 9ul
--------------------------------
10ul
2.5ul for each reaction
2. For BioTaq
10x reaction buffer 5ul x 8 = 40ul
MgCl2 3ul x 8 = 24ul
dNTP 2ul x 8 = 16ul
------------------------------------
80ul
10ul for each reaction
BioTaq 0.5ul x 6 = 3ul
ddH2O 9ul
------------------------------------
12ul
2ul for each reaction
| No. | Water (ul) | PCR Mix (ul) | Forward Primer (ul) | Reverse Primer (ul) | Template (ul) | Polymerase used (ul) | Total Volume (ul) |
|---|---|---|---|---|---|---|---|
| 1 | 28.5 | 12 | cwlJForward 2.5ul | cwlJReverse 2.5ul | cwlJ PCR result 2ul | GoTaq 2.5ul | 50 |
| 2 | 30.5 | 12 | cwlJForward 2.5ul | cwlJReverse 2.5ul | 0 | GoTaq 2.5ul | 50 |
| 3 | 28.5 | 12 | Arun's primer 2.5ul | Arun's primer 2.5ul | Arun's template 2ul | GoTaq 2.5ul | 50 |
| 4 | 30.5 | 12 | Arun's primer 2.5ul | Arun's primer 2.5ul | 0 | GoTaq 2.5ul | 50 |
| 5 | 31 | 10 | cwlJForward 2.5ul | cwlJReverse 2.5ul | cwlJ PCR result 2ul | BioTaq 2ul | 50 |
| 6 | 33 | 10 | cwlJForward 2.5ul | cwlJReverse 2.5ul | 0 | BioTaq 2ul | 50 |
| 7 | 31 | 10 | Arun's primer 2.5ul | Arun's primer 2.5ul | Arun's template 2ul | BioTaq 2ul | 50 |
| 8 | 33 | 10 | Arun's primer 2.5ul | Arun's primer 2.5ul | 0 | BioTaq 2ul | 50 |
| 9 | 31 | 10 | cwlJForward 2.5ul | cwlJReverse 2.5ul | 168 DNA 2ul | BioTaq 2ul | 50 |
| 10 | 33 | 10 | cwlJForward 2.5ul | cwlJReverse 2.5ul | 0 | BioTaq 2ul | 50 |
- PCR procedure: STD50
95C 2min -->[95C 30S --> 50C 30S --> 75C 1min] 30 runs --> 75C 5min --> store at 4C
Culture transformation E.coli for mini prep
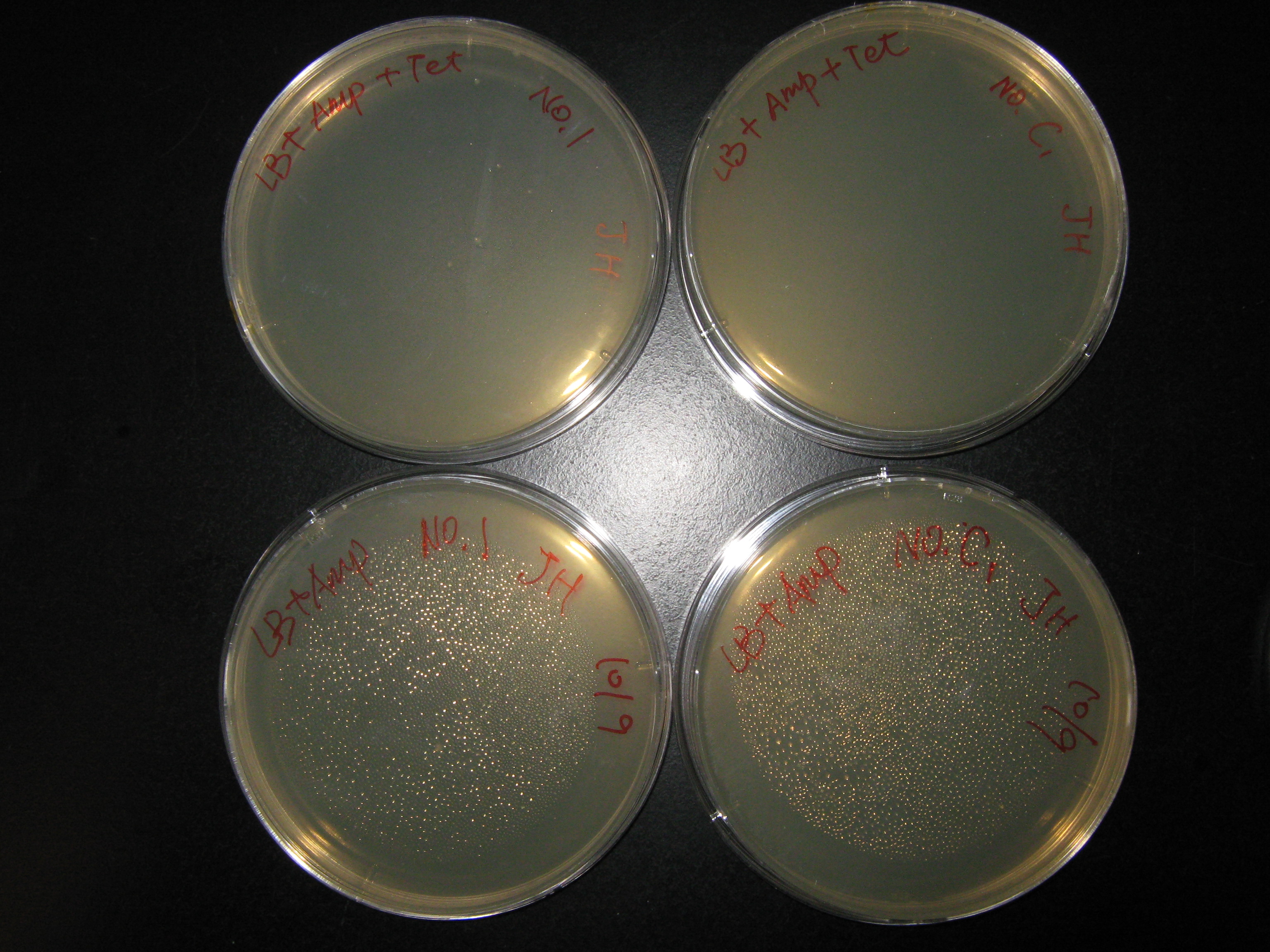
- Add 5ml LB+Amp+Tet liquid medium to 10 glass tubes.
- Use teeth stick to pick up single colony and put it into glass tubes.
- Put the tubes into shake incubator 37C overnight.
Conclusion
- The PCR result showed that BioTaq worked well for both cwlJ and sleB,even when we use the original designed primers and B.subtilis 168 DNA as template.
- We can't get PCR result when we used MotTaq. It may because our MotTaq has lost it's activity or it is not suitable for our template.
- GoTag works well when we applied PCR for sleB, but no PCR result for cwlJ when we used primer cwlJForward and cwlJReverse. After we got the PCR result of cwlJ fragment (by cwlJnoCut Primer) and use it as PCR template, GoTaq worked well.
Further plan
- Mini prep transformation of sleB:pSB1AT3.
- Midi prep the right clone of sleB.
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
News
Events
- 20 – 21 June 2009 - Europe workshop (London)
- 23 – 24 June 2009 - UK iGEM meetup (Edinburgh)
- 23 October Practice Presentation (Newcastle)
- 23 October T-shirts are ready
- 27 October Practice Presentation (Sunderland)
- 27 October Poster is ready
- 30 October – 2 November 2009 - Jamboree (Boston)
Social Net
- Newcastle iGEM Twitter
- [http://www.facebook.com/home.php#/group.php?gid=131709337641 Newcastle on Facebook]
- [http://www.youtube.com/user/newcastle2009igem Newcastle Youtube Channel]
 "
"