Team:Cambridge/Notebook/Week4
From 2009.igem.org
(→Amplification) |
(→Monday) |
||
| Line 61: | Line 61: | ||
====Codon Optimisation for B.Subtilis and E.Coli==== | ====Codon Optimisation for B.Subtilis and E.Coli==== | ||
| + | |||
| + | ==Tuesday== | ||
| + | |||
| + | ===Wet Work=== | ||
| + | |||
| + | ===Dry Work=== | ||
<!--Do not remove the first and last lines in this page!--><div id="contentbox_bottom"></div></div> | <!--Do not remove the first and last lines in this page!--><div id="contentbox_bottom"></div></div> | ||
Revision as of 08:56, 4 August 2009
Categories :
Project :
-
Overview
Sensitivity Tuner
--- Characterisation
--- Modelling
Colour Generators
--- Carotenoids (Orange/Red)
--- Melanin (Brown)
--- Violacein (Purple/Green)
The Future
Safety
Notebook :
Team Logistics :
Week 4 - Development
Monday
Wet Work
Melanin
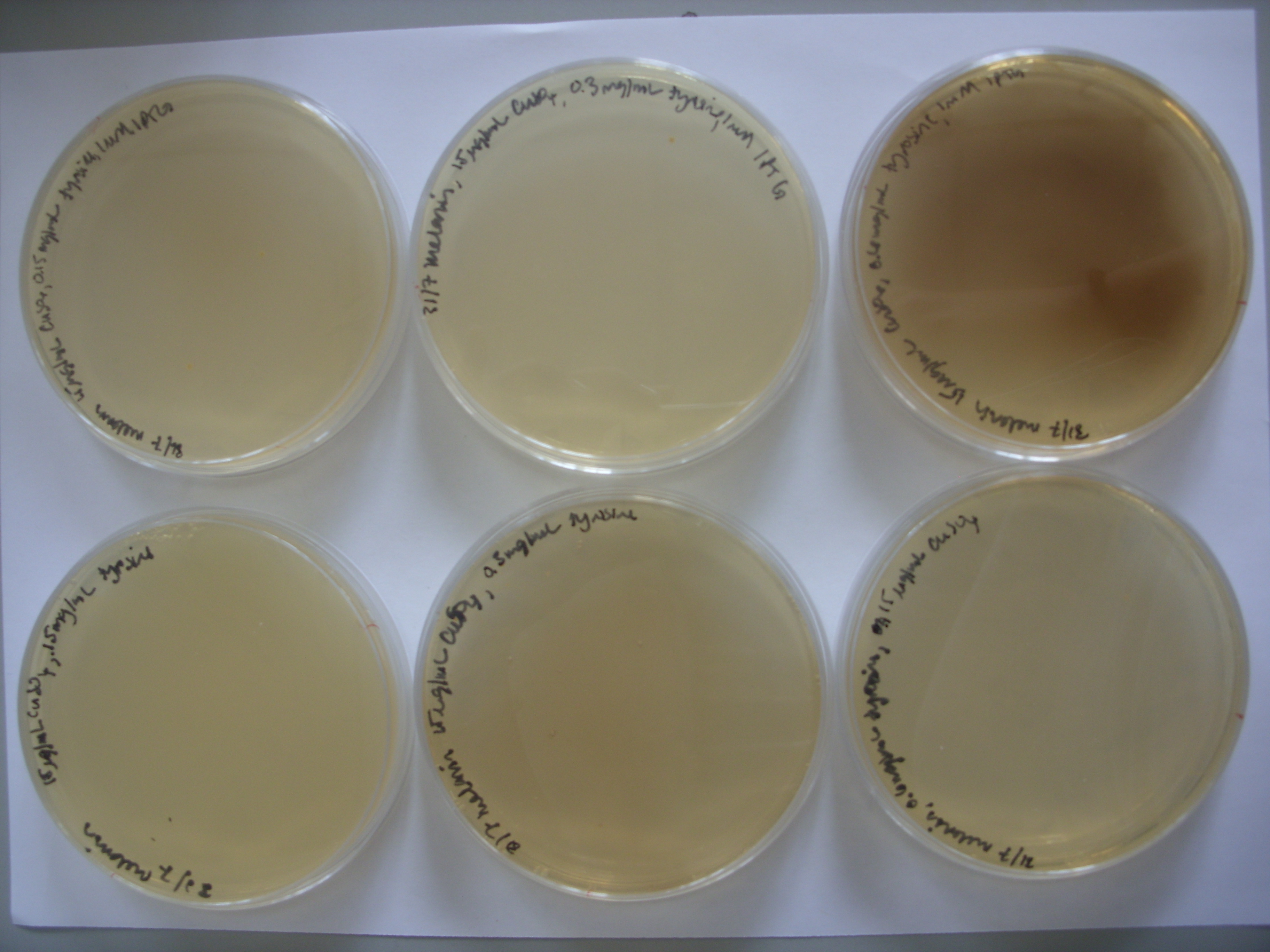
- Results from weekend plates:
From left to right, top row: 1mM IPTG with 0.075 mg/mL tyrosine, 1mM IPTG and 0.3 mg/mL tyrosine, and then 1mM IPTG and 0.6 mg/mL tyrosine From left to right, bottom row: 0.075 mg/mL tyrosine, 0.3 mg/mL tyrosine, and then 0.6 mg/mL tyrosine.
It appears that the bacteria were much too high a concentration; they formed lawns. As we saw on the first plates, pigment production is reduced at high bacterial concentrations. However, it is encouraging that the darkest plate is the one with the greatest tyrosine concentration and IPTG.
To follow up, the experiment was repeated with 1/100 and 1/1000 dilutions of bacterial culture on the following plates:
100ug/ml Ampicillin, 15 ug/ml CuSO4, 0.3 mg/mL tyrosine
100ug/ml Ampicillin, 15 ug/ml CuSO4, 0.6 mg/mL tyrosine
100ug/ml Ampicillin, 7.5 ug/ml CuSO4, 0.075 mg/mL tyrosine, 0.5mM IPTG
100ug/ml Ampicillin, 15 ug/ml CuSO4, 0.3 mg/mL tyrosine, 1mM IPTG
100ug/ml Ampicillin, 15 ug/ml CuSO4, 0.6 mg/mL tyrosine, 1mM IPTG
- Unfortunately, the overnight cultures for the plate readers were contaminated, so a single colony was once again incubated overnight in 10 mL of 100ug/ml Ampicillin, 15 ug/ml CuSO4, 0.3 mg/mL tyrosine, 1mM IPTG in preparation for pigment characterisation using the plate reader.
Violacein
As described in the resource cited below, violacein can be extracted using butanol. A single colony was incubated overnight in LB with trimethoprim. We hope to achieve pigment production in liquid cultures and then extract pigment later in the week.
P.R. August, T.H. Grossman, C. Minor, M.P. Draper, I.A. MacNeil, J.M. Pemberton, K.M. Call, d. Holt, and M. S. Osbourne, Sequence Analysis and Functional Characterization of the Violacein Biosynthetic Pathway from Chromobacterium violaceum, J. Mol. Microbiol. Biotechnol. (2000) 2(4): 513-519. [http://www.horizonpress.com/jmmb/v2/v2n4/26.pdf]
Amplification
- Chose 4 activator / promoter combinations to continue work with:
- P2 activator with Psid promoter (I746374)
- P2 activator with PLL promoter (I746375)
- PSP3 activator with PF promoter (I746380)
- phiR73 with PO promoter (I746391)
- Of the above, on plates all showed leaky expression of GFP and RFP, suggesting that last weeks transformation was successful.
- In preparation for a plate reader assay tomorrow, incubated single colonies overnight of the above constructions as well as the following controls
- I3540 (pBad and GFP) - taken from 2007 glycerol stocks
- I3620 (pBad and RFP) - taken from 2007 glycerol stocks
- untransformed Arabinose strain colony
- Plate reader assay will have triple replicates of all of the above constructs induced with arabinose at 0 uM, 10 uM, 100 uM, and 1mM concentrations.
Dry Work
Promoter response to input concentration
- The pBAD promoter currently used in the amplification system is sensitive to different concentrations of arabinose, although this effect could be considered limited, so this is probably not the best promoter to use to get a sequence of outputs at different concentrations [].
 "
"