Analysis Methods
Tutorials
The following section is a kind of tutorial, which describes step by step how to obtain our different results.
Maxwell-Boltzmann Energy Distribution
Click here to show/hide
Here we will confirm that the kinetic energy distribution of the atoms in a system corresponds to the Maxwell distribution for a given temperature.
1. In VMD, load the .psf file. Browse for the restart velocity file (in our case, it was 2v0w_hydr_wb_i_eq.restart.vel), the type of file need this time to be selected. In the Determine file type: pull-down menu, choose NAMD Binary Coordinates, and load again.
The molecule looks terrible! That is because VMD is reading the velocities as if they were coordinates, but how it looks doesn't matter: we just need VMD to make a file containing the masses and velocities for every atom.
2. We create a selection with all the atoms in the system, by typing in the TkCon window:
set all [atomselect top all]
Open a file energy.dat for writing:
set fil [open energy.dat w]
3. For each atom in the system, calculate the kinetic energy, and write it to a file :
foreach m [$all get mass] v [$all get {x y z}] {
puts $fil [expr 0.5 * $m * [vecdot $v $v] ]
}
Close the file:
close $fil
All these steps can be avoided by typing in the Terminal Window:
vmd -dispdev text -e get energy.tcl
We now have a file of raw data that can be used to fit the obtained energy distribution to the Maxwell-Boltzmann distribution. The temperature of the distribution can be obtained from that fit.
1. In a Terminal window, type xmgrace. In the xmgrace window, choose the Data → Import → ASCI I menu item. In the Files box, select the file energy.dat. Click on the OK button. You should see a black trace which correspond to the raw data.
2. To look at the distribution of points, we will make a histogram of this data. Choose the Data → Transformations → Histograms menu item. In the Source → Set window, click on the first line, in order to make a histogram of the data just loaded.
Click on the Normalize option.
Fill in Start: 0, Stop at: 10, and # of bins: 100, and Apply.
 3.
3. The plot has been created, but cannot be seen yet. Use the right button of the mouse to click on the first set (in the Source → Set window).
Click on Hide. Now, go to the main Grace window and click on the button labeled AS: it will resize the plot to fit the existing values. This is the distribution of energies.
Now, we will fit a Maxwell-Boltzmann distribution to this plot and find out the temperature that the distribution corresponds to.
4. Choose the Data → Transformations → Non-linear curve fitting menu item. In the Source → Set window, click on the last line, which corresponds to the histogram you created.
In the Main tab, type in the Formula window:
y = (2/ sqrt(Pi * a0 ∧3 )) * sqrt(x) * exp (-x / a0)
This will fit the curve and get a fit for the parameter a0, which corresponds to kB T in units of kcal/mol.
 5.
5. In the Parameters drop-down menu, choose 1. You can give an initial value to A0, so that the iterations will look for a value in the vicinity of your initial guess. In the A0 window, type 0.6, which in kcal/mol corresponds to a temperature of ∼ 300K.

Click on the Apply button. This will open a window with the new value for a0, as well as some statistical measures, including the Correlation Coefficient, which is a measure of the fit.

The value of a
0 obtained corresponds to k
BT . Obtain the temperature T for this distribution with k
B = 0.00198657 kcal
/mol /K.

Energies
Click here to show/hide
Here we will calculate the averages of the various energies (kinetic and the different internal energies: bonded (bonds,
angles and dihedrals) and non-bonded (electrostatic, van der Waals)) over the course of a simulation.
1. We start with a file obtained from NAMD:
http://www.ks.uiuc.edu/Research/namd/utilities/ and download namdstat.tcl
2. In the VMD TkCon window, type :
source namdstats.tcl
data avg ../namd_log 101 last
The second line will call a procedure which will calculate the average for all output variables in the log file from the first logged timestep after 101 to the end of the simulation.
Temperature distribution
Click here to show/hide
Objective: Simulate our protein in an NVE ensemble, and analyze the temperature distribution.
In order to obtain the data for the temperature from the log file we will again use the script namdstats.tcl, which was already sourced. Type in a terminal window:
data_time TEMP namd_log
It will store each timestep and its corresponding temperature in the file TEMP.dat.
Density
Click here to show/hide
In order to obtain the data for the volume from the log file we will again use the script ''namdstats.tcl'', which was already sourced. Type in a terminal window:
data_time VOLUME namd_log
It will store each timestep and its corresponding temperature in the file TEMP.dat.
Pressure as a function of simulation time
Click here to show/hide
In order to obtain the data for the pressure from the log file we will again use the script namdstats.tcl, which has been updated for the occasion.
The file can be found here . Please rename to namdstats.tcl after download.
Here are the steps to use this script:
source namdstats.tcl
data_time DATA_NAME LOG_FILE
It will extract data from LOG_FILE, creating a DATA_NAME.dat containing values and time informations.
Available DATA_NAME can be: BOND, ANGLE, DIHED, IMPRP, ELECT, VDW, BOUNDARY, MISC, KINETIC, TOTAL, TEMP, TOTAL2, TOTAL3, TEMPAVG, PRESSURE, GPRESSURE, VOLUME, PRESSAVG, GPRESSAVG.
So, to extract pressure from our first simulation, the command is: data_time PRESSURE namd_log
RMSD for individual residues
Click here to show/hide
We aim at finding the average RMSD over time for each residue in the protein using VMD.
1. We load the .psf and .dcd files obtained after the first round of simulation.
2. In the TkCon window type:
source residue_rmsd.tcl
set sel_resid [[atomselect top "protein and alpha"] get resid]
This will get all the residues number of all alpha-carbons in the protein. Since there is just one and only one α-carbon per residue, it is a good option.
3. Now we will calculate the RMSD values of all atoms in the newly created selection:
rmsd_residue_over_time top $sel resid
At the end of the calculation, we have a list of the avergae RMSD per residue (in the file residue_rmsd.dat)
Remark: we updated the script residue_rmsd.tcl to be able to specify on which frames the rsmd has to be computed. Please have a look on this wiki for a more up-to-date version of the file... Command is:
rmsd_residue_over_time top $sel resid FIRST_FRAME LAST_FRAME
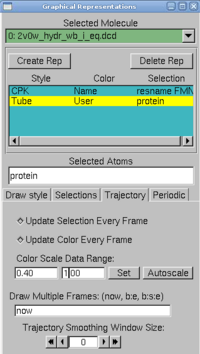
4. In VMD, in Graphics → Representations, do the following actions:
- create two replicates: protein and resname FMN in selected atoms
- for the FMN, choose CPK as a drawing method
- for the protein, choose tube as drawing method, and user as coloring method. Now click on the Trajectory tab, and in the Color Scale Data Range, type 0.40 and 1.00.
The protein is colored according to its average RMSD values. The residues displayed in blue are more mobile while the ones in red move less.
Here is a movie with the protein colored according to average RMSD values:
video
5. Now we can plot the RMSD value per residue by typing in a Terminal window :
xmgrace residue rmsd.dat

RMSD of selected atoms compared to initial position along time
Click here to show/hide
This script was highly updated, please go to the script page if you encounter a problem!!!
Selections are not precise here!
We made a small TCL script to calculate RMSD from selected atoms compared to their initial position along timestep.
The file can be found here. Please rename to Residue_rmsd_igem09.tcl after download.
Example to run the script:
load ''.dcd'' + ''.psf'' on VMD
source residue_rmsd_igem09.tcl
set sel_resid [[atomselect top "protein and alpha"] get resid]
rmsd_residue_over_time top $sel_resid 0 0
We tried to select only backbone from protein + FMN → change
set sel_resid [[atomselect top "backbone"] get resid]
The script was updated to be able to define reference frame and first frame were RMSD will be calculated. We usually don't need to compute RMSD during heating, for instance. RMSD takes a lot of time. In our first run 1 frame = 100 timesteps * 2 fs*timesteps^-1 = 200 fs
complete form for run is:
rmsd_residue_over_time top $sel_resid FIRST_FRAME REFERENCE_FRAME
For our first run, if we want to select only the 295°K NPT plateau, and set its first frame as reference, we have to launch:
rmsd_residue_over_time top $sel_resid 1115 1115
Here is how the script processes:
- calculate how many frames are in .dcd
- for each timestep, the script aligns (best fit) the backbone of the protein to the eference position to minimize RMDS. (Test: "and not mass 1,008000" == and noh was added in selection to remove hydrogen)
- for each residue (selected by sel_resid), RMSD is computed and the sum of all RMSD (one for each residue) is stored for current timestep
- script's output is data_rmsd.dat

Salt bridges
Click here to show/hide
As we wanted to redo the analysis from Schulten's article, we looked for salt bridges. VMD can easily compute this, it even propose an easy GUI. Standard configuration is just fine for now. You'll have a log file containing the list of nitrogen-oxygen susceptible of forming a salt bridge. You'll also get a file for each bridge containing the distance between both atoms along the simulation.
In the light state, we have 9 salt bridges witin the protein and 12 if we consider the protein and the flavin (use "protein or resname FMN" as selection).
ASP471-ARG467
GLU409-ARG442
FMN450-FMN450
ASP540-LYS544
ASP432-ARG442
FMN450-ARG451
GLU457-LYS489
GLU444-LYS485
ASP522-ARG521
ASP424-ARG451
GLU475-LYS533
FMN450-ARG467

RMSF
Click here to show/hide
After changing the script [see here], we perform an interesting analysis from these 2 files. First, we have to correct the RMSF, that can be linked to beta factor using this equation:

If you plot beta factor and RMSF, you get such a thing.


Scripts used
A few scripts were needed to analyze the trajectory file (.dcd) and log files from namd. We'll list here what we made and how they can be used. All scripts are in TCL but don't show an elevated syntax.
Some informations about the simulation are stored in the log file. A script is really useful to extract them before plotting.
We updated the file available on namd website. Here is our version of namdstats.tcl. Please rename to namdstats.tcl after download.
Here are the steps to use this script:
- source namdstats.tcl
- data_time DATA_NAME LOG_FILE
It will extract data from LOG_FILE, creating a DATA_NAME.dat in the working directory (type pwd to see where you are) containing values and time informations.
Available DATA_NAME are: BOND, ANGLE, DIHED, IMPRP, ELECT, VDW, BOUNDARY, MISC, KINETIC, TOTAL, TEMP, TOTAL2, TOTAL3, TEMPAVG, PRESSURE, GPRESSURE, VOLUME, PRESSAVG, GPRESSAVG.
So, to extract pressure from our first simulation, the command is:
- data_time PRESSURE namd_log
Sum of RMSD for each residue
- Basically, this script will calculate the RMSD at each frame. The ouptut is residue number and the sum of its RMSD at each specified frame.
- You have to provide a list or residue ID in the $sel_resid. There is no check for duplicated ID, so choose carefully your selection ;). (The .dcd and .pdb must be loaded before). You probably want to select each residue composing the protein only once, that's why we use "protein and alpha", there is only one CA per residu. This selection is only to define which residues are taken in consideration, it has no impact on wich atoms are selected.
- If you want to change which atoms are taken in consideration for RMSD, you'll have to update the selection within the script.
- The RMSD will calculate the RMSD for each non-hydrogen atom of the residue at each step and sum all these results. That's why the process is really heavy: #Residues * #AtomsPerResidue * #Frames * #CostOfRMSD
- Output is residue_rmsd.dat
In the TkCon window type:
- source residue_rmsd_v2.tcl
- set sel_resid [[atomselect top "protein and alpha"] get resid]
- rmsd_residue_over_time top $sel resid
Remark: we updated the script residue_rmsd.tcl that was originaly available from namd website to be able to specify on which frames the rsmd has to be computed. Please have a look on this wiki for a more up-to-date version of the file... Command is:
- rmsd_residue_over_time top $sel resid FIRST_FRAME LAST_FRAME
Mean RMSD along time
We used this particular representation of RMSD to visualize if our simulation is reaching an equilibrium. It also helps a lot to see if the parameters are correctly set.
As we wrote the script, take a special care at the selections you make ;)
- The script will go through the frames and sum RMSD of each selected residues (in fact it will sum RMSD of every atom composing the residue)
- The sum of RMSD for each frame is then divided by the number of residues, to "normalize" the value
- As before, you have to provide the list of residues.
- Output is data_rmsd.dat
In the TkCon window type:
- source residue_rmsd_igem09.tcl
- set sel_resid [[atomselect top "protein and alpha"] get resid]
- rmsd_residue_over_time_igem top $sel_resid 0 0
The script was updated to be able to define reference frame and first frame were RMSD will be calculated. We usually don't need to compute RMSD during heating, for instance. RMSD takes a lot of time. In our first run 1 frame = 100 timesteps * 2 fs*timesteps^-1 = 200 fs
complete form for run is:
- rmsd_residue_over_time top $sel_resid FIRST_FRAME REFERENCE_FRAME
RMSF from namd .dcd
Root mean square fluctuation can be compared to the beta factor observed during chrystallography. The beta factor has to be loaded from the .pdb. This can be achieved with a few bash commands:
- cat 2v0u.pdb | grep CA > 2v0u_prot_CA.pdb
We then wrote a script to get RMSF.
- RMSF is calculated on a selected window of frames (between FIRST_FRAME and LAST_FRAME).
- There is no reference frame, the function return the fluctuation around the average in the window.
- It returns one value per alpha carbon.
- Output is data_rmsf_igem.dat
In the TkCon window type:
- source rmsf_igem09.tcl
- set sel_resid [[atomselect top "protein and alpha"] get resid]
- rmsf_CA top $sel_resid FIRST_FRAME LAST_FRAME

 "
"







