Molecular cloning: strong promoter+Pcat-2M-lacI-term-lacP-GFP
Parts: J61100+K228819
Resource:
Pcat-2M-lacI-term-lacP-GFP: myself, colonies, renamed as L1, L2, L3.
Promoter (J61100): from Lin Min, vector (has already digested by Pst1 and Spe1)
2009.8.8
Plasmid mini prep:
L1, L2, L3
Double digest:
L1, L2:
| Pst1 | 1uL
|
| plasmid | 10uL
|
| Buffer | 2uL
|
| water | 6uL
|
37 ℃ 4 hour
2009.8.9
Gel electrophoresis:
Products of double digest of L1, L2,
marker: 100bp 250bp 500bp 750bp 1kb 2kb 3kb 5kb
loading buffer and DNA dye: 6×
voltage and time: 60V 5min; 120V 60min
lane2: Marker
lane1,3: L1, L3;
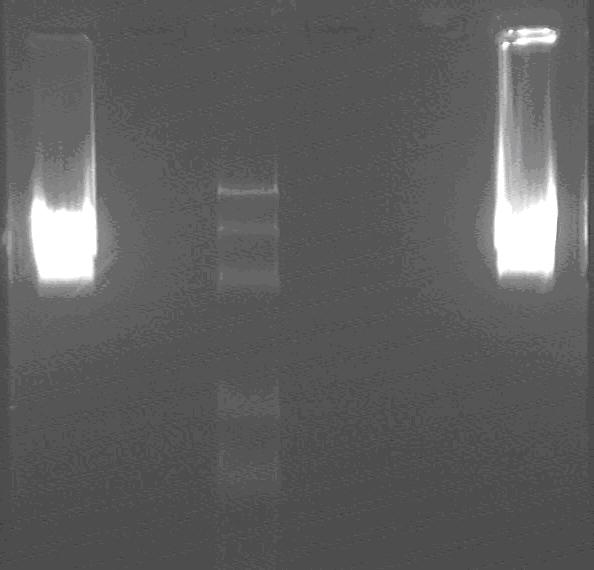
The insert is about 2.4kb, and the backbone is 2.1kb.
DNA Gel purification:
The insert of L1 and L2.
DNA ligation:
| System | 10uL
|
| Insert | 6uL
|
| vector | 2uL
|
| water | 3uL
|
| buffer | 1uL
|
| T4 DNA ligase | 1uL
|
16℃ 4 hour
Insert: L1, L2;
Vertor: strong promoter, from Lin Min.
Transformation:
Products of ligation, competent cells 50uL each,
Smear to LB plate with Amp
2009.8.10
Every plate (P+L1/L2) is very well: more than 100 clones
But many colonies are become green under the blue light, which means that the expression of LacI can not fully repressed the promoter lacP.
Result & Discussion
I successfully constructed clone: Promoter-Pcat-2M-lacI-term-lacP-GFP .
However, I disappointed to find that these clones are not work very well, because the GFP express significantly even on the plate (without induce)!!!
The expression of lacI is still not enough to repress the lacP.
^Top
|
 "
"

