Molecular cloning: Pcat-2M-lacI/tetR-term+lacP/tetP
Parts: K228815/16+R0010/R0040=K228817/18
Resource:
Pcat-2m-lacI/tetR-term (K228815/16): myself, plasmid, rename as L, T.
lacP: part R0010, from He Siheng, already digested;
tetP: part R0040, myself, already digested (July 20th)
2009.7.30
Double digest:
L, T:
| Spe1 | 1uL
|
| Pst1 | 1uL
|
| plasmid | 10uL
|
| Buffer | 2uL
|
| water | 6uL
|
37 ℃ 4 hour
Gel electrophoresis:
Products of double digest of L, T
marker: 100bp 250bp 500bp 750bp 1kb 2kb 3kb 5kb
loading buffer and DNA dye: 6×
voltage and time: 60V 5min; 120V 30min
lane1: digested product of T;
lane2: digested product of L;
lane3: marker;
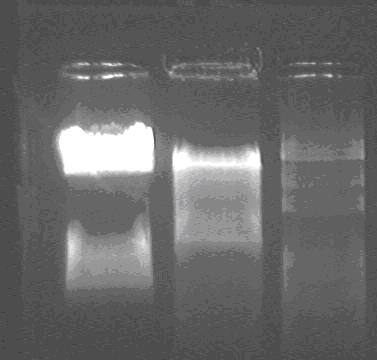
The insert of T is about 900bp.
The insert of L is about 1.4kb.
DNA Gel purification:
Inserts of L, T.
DNA ligation:
| System | 10uL
|
| Insert | 6uL
|
| vector | 2uL
|
| water | 3uL
|
| buffer | 1uL
|
| T4 DNA ligase | 1uL
|
16℃ overnight.
Insert: T *2;
Vertor: tetP (has already digested by EcoR1 & Xba1)
2009.7.31
Transformation:
Products of ligation (T+tetP *2), competent cells 50uL each,
Smear to LB plate with Amp
DNA ligation:
| System | 10uL
|
| Insert | 6uL
|
| vector | 2uL
|
| water | 3uL
|
| buffer | 1uL
|
| T4 DNA ligase | 1uL
|
16℃ overnight.
Insert: L *2;
Vertor: lacP (has already digested by EcoR1 & Xba1, by He Siheng)
Transformation:
Products of ligation (L+lacP *2), competent cells 50uL each,
Smear to LB plate with Amp
2009.8.1
Every plate is very well: more than 100 clones
PCR: (colony PCR, T-tetP)
| System | 10 uL
|
| Master mix | 5ul
|
| primer (standard primer) | 0.5uL each
|
| water | 4uL
|
| template | 10 colonies of T-tetP
|
Gel electrophoresis: (help by Lin Min)
Refer to Lin Min’s notes,
All of 10 colonies are wrong!!!
Repeat!!!
Double digest: (again, tetP)
tetP:
| EcoR1 | 1uL
|
| Xba1 | 1uL
|
| plasmid | 4uL
|
| Buffer | 2uL
|
| water | 12uL
|
37 ℃ 4 hour
Transfer colonies: (L-lacP)
Transfer 6 colonies (L-lacP) into 5ml LB, and amplify the Ecoli.
Plasmid mini prep: (L-lacP)
6 colonies of L-lacP
Double digest: (to check the correct L-lacP)
6 L-lacP:
| EcoR1 | 1uL
|
| Pst1 | 1uL
|
| plasmid | 4uL
|
| Buffer | 2uL
|
| water | 12uL
|
37 ℃ overnight
2009.8.2
Gel electrophoresis: (check the correct L-lacP)
Products of double digest
Marker: 100bp 250bp 500bp 750bp 1kb 2kb 3kb 5kb
loading buffer and DNA dye: 6×
Voltage and time: 60V 5min; 120V 15min

I forgot to add the Marker, but from the result we can easily find that all these 6 colonies are wrong!!!
PCR: (colony PCR, L-lacP)
| System | 10 uL
|
| Master mix | 5ul
|
| primer (standard primer) | 0.5uL each
|
| water | 4uL
|
| template | 24 colonies of L-lacP
|
Gel electrophoresis: (check the correct L-lacP)
Products of PCR
Marker: 100bp 250bp 500bp 750bp 1kb 2kb 3kb 5kb
loading buffer and DNA dye: 6×
Voltage and time: 60V 5min; 120V 15min

From the lane 5 to the last one are 24 results of L-lacP PCR.
Since there is not any DNA larger than 1kb, all of these 24 colonies are wrong!!!
All these DNA are about 200bp. So they are the result of self link of lacP!!! There must be something wrong with the digested lacP!!!
Double digest: (again, lacP)
lacP:
| EcoR1 | 1uL
|
| Xba1 | 1uL
|
| plasmid | 4uL
|
| Buffer | 2uL
|
| water | 12uL
|
37 ℃ overnight!
DNA ligation (again T+tetP):
| System | 10uL
|
| Insert | 6uL
|
| vector | 2uL
|
| water | 3uL
|
| buffer | 1uL
|
| T4 DNA ligase | 1uL
|
16℃ 4 hours
Insert: T *2;
Vertor: tetP (digested on Aug.1st)
Transformation: (again T+tetP)
Products of ligation (T+tetP *2), competent cells 50uL each,
Smear to LB plate with Amp
2009.8.3
PCR product purification:
lacP (digested yesterday)
DNA ligation (again L+lacP):
| System | 10uL
|
| Insert | 6uL
|
| vector | 2uL
|
| water | 3uL
|
| buffer | 1uL
|
| T4 DNA ligase | 1uL
|
16℃ 4 hours
Insert: L *2;
Vertor: lacP (digested on Aug.2nd )
PCR: (colony PCR, T-tetP)
| System | 10 uL
|
| Master mix | 5ul
|
| primer (standard primer) | 0.5uL each
|
| water | 4uL
|
| template | 10 colonies of T-tetP
|
Gel electrophoresis:
Products of PCR
Marker: 100bp 250bp 500bp 750bp 1kb 2kb 3kb 5kb
loading buffer and DNA dye: 6×
Voltage and time: 60V 5min; 120V 15min
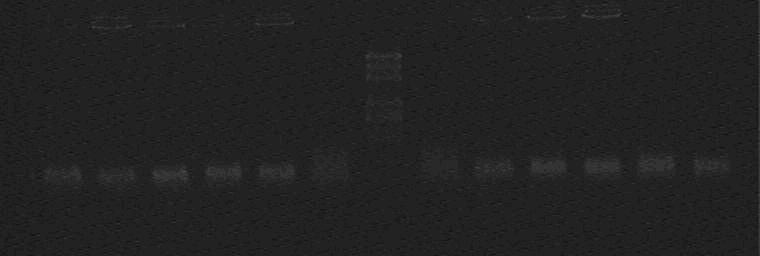
Since there is not any DNA larger than 1kb, all of these 10 colonies are wrong Again!!!
Bad Luck!!!!!
Transformation: (again L+lacP)
Products of ligation (L+ lacP *2), competent cells 50uL each,
Smear to LB plate with Amp
Double digest: (the 3rd time! tetP and T)
tetP:
| EcoR1 | 1uL
|
| Xba1 | 1uL
|
| plasmid | 4uL
|
| Buffer | 2uL
|
| water | 12uL
|
T:
| pe1 | 1uL
|
| Pst1 | 1uL
|
| plasmid | 10uL
|
| Buffer | 2uL
|
| water | 6uL
|
37 ℃ overnight!
2009.8.4
PCR product purification:
tetP (digested yesterday)
Gel electrophoresis:
Products of double digest of L, T
marker: 100bp 250bp 500bp 750bp 1kb 2kb 3kb 5kb
loading buffer and DNA dye: 6×
voltage and time: 60V 5min; 120V 30min
lane1: digested product of T;
lane2: marker

The insert should be 900 bp, and it is correct!
DNA Gel purification:
Insert of T.
DNA ligation (the 3rd time T+tetP):
| System | 10uL
|
| Insert | 6uL
|
| vector | 2uL
|
| water | 3uL
|
| buffer | 1uL
|
| T4 DNA ligase | 1uL
|
16℃ 4 hours
Insert: T *2 (new);
Vertor: tetP (new);
Transformation: (the 3rd time T+tetP)
Products of ligation (T+tetP *2), competent cells 50uL each,
Smear to LB plate with Amp
PCR: (the 2nd time colony PCR, L-lacP)
| System | 10 uL
|
| Master mix | 5ul
|
| primer (standard primer) | 0.5uL each
|
| water | 4uL
|
| template | 12 colonies of L-lacP
|
Gel electrophoresis: (check the correct L-lacP)
Products of PCR
Marker: 100bp 250bp 500bp 750bp 1kb 2kb 3kb 5kb
loading buffer and DNA dye: 6×
Voltage and time: 60V 5min; 120V 15min
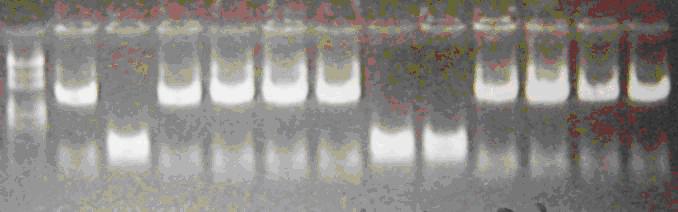
The insert of correct L-lacP is about 1.4kb!!!
9 of 12 colonies are correct!!!!!
2009.8.5
PCR: (the 3rd time colony PCR, T-tetP)
| System | 10 uL
|
| Master mix | 5ul
|
| primer (standard primer) | 0.5uL each
|
| water | 4uL
|
| template | 12 colonies of T-tetP
|
Gel electrophoresis: (check the correct T-tetP)
Products of PCR
Marker: 100bp 250bp 500bp 750bp 1kb 2kb 3kb 5kb
loading buffer and DNA dye: 6×
Voltage and time: 60V 5min; 120V 60min
Lane 1~12: T-tetP 1~12
Lane 13: Marker
The insert is about 1kb, and 9 of these 12 colonies are CORRECT!!!!

Result
At last, I successfully constructed: Pcat-2M-lacI/tetR-term+lacP/tetP, and they are the parts K228817/18.
Experience
The vector is very important in this cloning. We should digest completely all the vectors, in order to prevent the self-linkage of the vectors. My experience is that if you want to digest 4ul plasmid as vector, you had better digest it overnight. If you want to quick such as in two hours, reduce the amount of plasmid.
^Top
|
 "
"







