Notebook > AND_Gate_2 > Min Lin's Note
2009.9.3
PCR T3polymerase
| Phusion | 0.5ul
|
| Primer F | 1.25ul
|
| Primer R | 1.25ul
|
| Template | 1ul
|
| HF Buffer | 10ul
|
| ddH2O | 32ul
|
| dNTP | 4ul
|
PCR a gradient 58, 60, 62, 65, 67
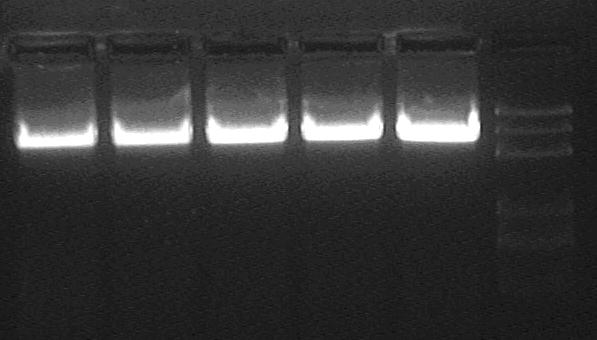
GEL Purification.
Enzyme Digestion
| PCR product | 10ul
|
| EcoRI | 1ul
|
| PstI | 1ul
|
| 10xH Buffer | 2ul
|
| ddH2O | 6ul
|
2009.9.4
Purify the product of enzyme digestion.
Ligation:
pSB1K3 backbone
T3 polymerase
Pick colonies of the T7p-CI 1-1M.
PCR assessment:
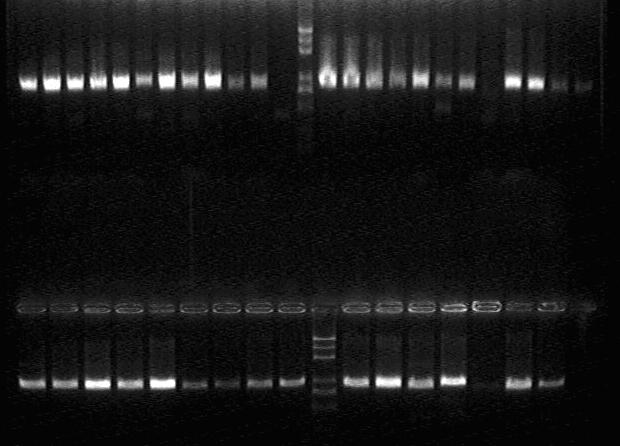
Shake each one of the correct colonies in the incubator.
2009.9.5
Miniprep 6 of the T3polymerase colonies.
5 of them are red.
Enzyme Digestion assessment of the remaining one.
At the same time PCR with the Sequencing primer for assessment.
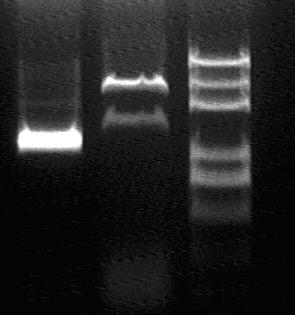
Enzyme Digestion again to confirm:
XhoI, NotI, (XbaI, SpeI), (XbaI, SpeI, HindIII) for assessment
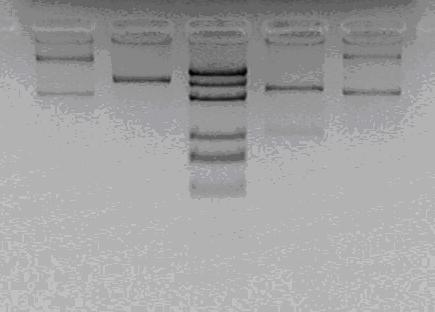
It is not a correct colony.
2009.9.6
PCR again the 3 counter plasmid. Same protocol.
At the same time use the PCR product of last time to do nested PCR with Standard primer.
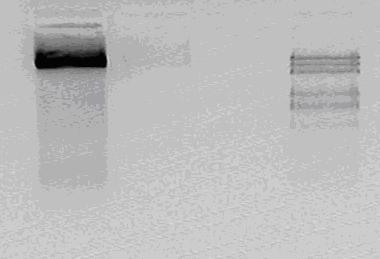
No result for the nested PCR.
Again PCR 3 counter plasmid
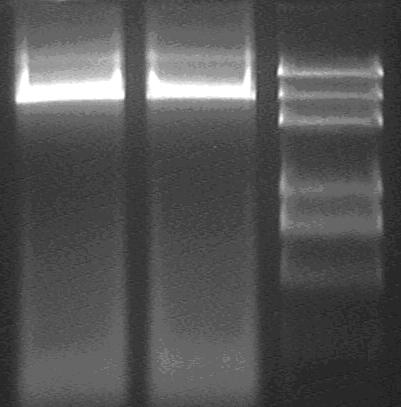
Purification;
Enzyme Digestion one the PCR products with EcoRI and SpeI.
Ligation:
T3 polymerase PCR product into pEASY-BLUNT.
T3 polymerase PCR product after digestion into pSB1A2(ES).
Transformation.
Enzyme Digestion of the T7p-CI into 1-1M for Enzyme digestion assessment:
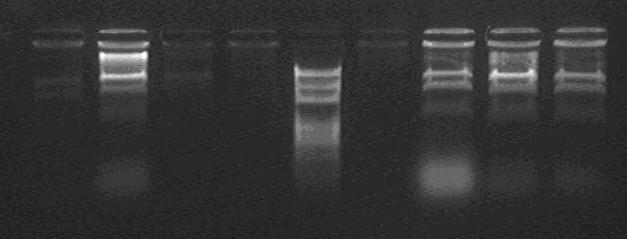
Looks like no one is correct in size.
2009.9.8
Pick 10 colonies from the pEASY-BLUNT plate for assessment.
Pick 5 colonies from the pSB1A2 plate.
Miniprep
Enzyme Digestion with EcoRI and PstI
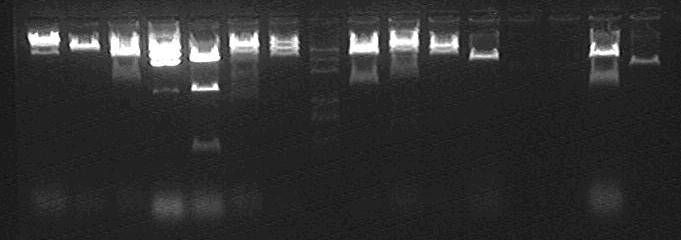
None is correct!!!
2009.9.9
Enzyme Digestion of the 3 Counter plasmid with EcoRI and NheI, Gel purification of the correct insert that contains the T3 pol.
PCR using the purified insert as a template.
PCR 2 tubes with Phusion, one without DMSO the other with DMSO.
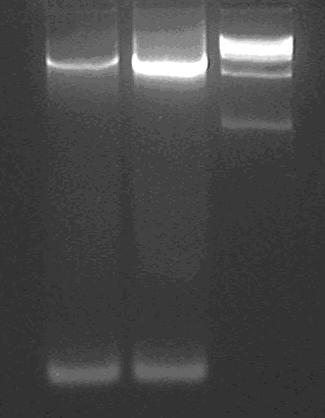
Gel purification of the two bands.
PCR again using the Standard primer and product of the first cycle of PCR as template.
GEL:

It seems that the size is not correct.
Purification and use some to confirm the size.
It turns out that the size is actually correct.
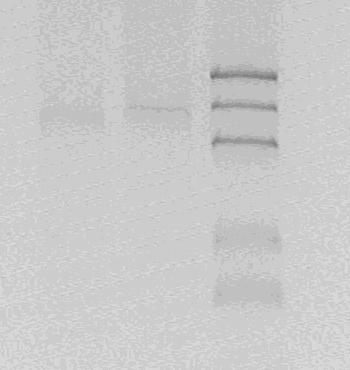
The product is digested with by XbaI and PstI directly.
Ligation:
T3 pol(XP) into pSB1A2
Transformation.
2009.9.11
Pick 10 colonies of the T3pol clone. No band
Mniprep 5 plasmids.
Digest with EcoRI and PstI for assessment
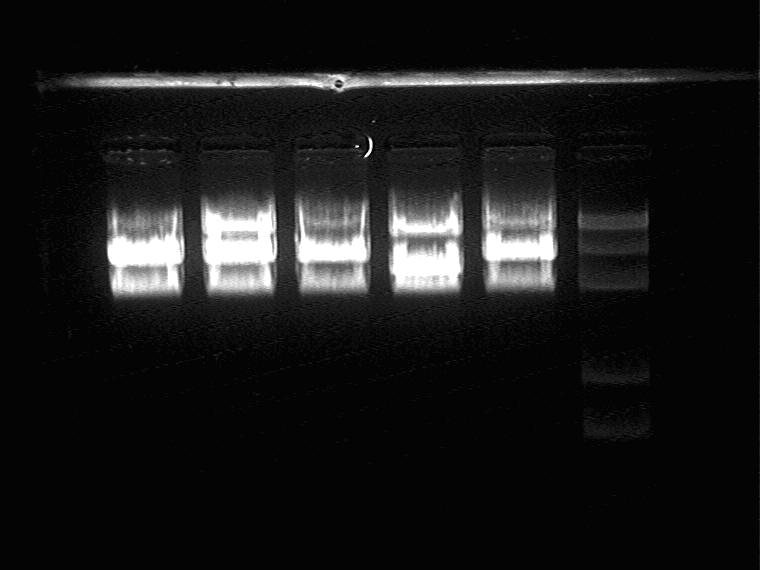
It seems right for NO. 1,2,3,5.
Digest with:
(ExoRI and PstI)
(EcoRI and SpeI)
(XbaI and PstI)
(XbaI and SpeI)
PCR with:
Sequencing primer
Standarlize primer
Universal Primer
For assessment
PCR
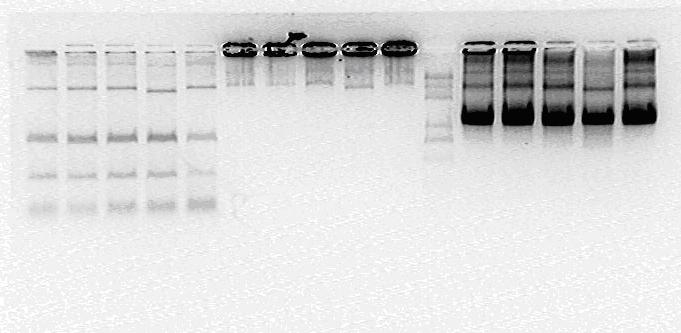
Enzyme Digestion
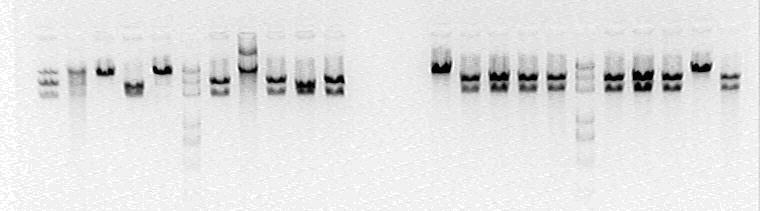
All phenomenon shows that this time it is correct.
Send for sequence, However, the forward primer has no signal, and the reverse primer shows more than one binding site. It is weird.
2009.9.15
Transformation from the part distribution the P2 activator PhiR73 delta and one of its promoter PO.
Plate overnight.
2009.9.16
Pick one of the PhiR73 delta colony and PO promoter colony, shake in the incubator.
Miniprep PhiR73 delta and PO promoter plasmid.
Use Phusion to PCR the PhiR73 delta plasmid over night.
| Phusion | 0.5ul
|
| PointMutation For | 1.25ul
|
| PointMutation Rev1 | 1.25ul
|
| Template | 1ul
|
| HF Buffer | 10ul
|
| ddH2O | 32ul
|
| dNTP | 4ul
|
| Phusion | 0.5ul
|
| PointMutation For | 1.25ul
|
| PointMutation Rev2 | 1.25ul
|
| Template | 1ul
|
| HF Buffer | 10ul
|
| ddH2O | 32ul
|
| dNTP | 4ul
|
The first PCR introduces one point mutation and the second introduces two.
2009.9.17
GEL of the PCR product
Purification of the GEL.
Blunting kination reaction 70 centigrade for 10min then 37 centigrade for 10min
Ligation.
Transformation:
PointMutation1, PointMutation2.
Plate
2009.9.18
Pick from PointMutation1, PointMutation2 plate each 5 colonies. Shake in the incubator.
Miniprep the 11 tubes.
Enzyme Digestion:
Digest PointMutation plasmids with EcoRI and PstI for assessment.
2009.9.19
Send pointmutation plasmid for sequencing.
2009.9.21
Enzyme Digestion PM9 with XbaI and PstI.
GEL. Purification.
Ligation:
Sal promoter(sp) vector
PM9(XP)
Transformation
2009.9.22
Pick colonies for PCR assessment. Get the correct colony.
Get Ara-SupD from GRC.
Enzyme Digestion with EcoRI SpeI.
Shake the Sal-PM9 colony in the incubator.
Shake the pSB4K5 1-7G strain in the incubator.
Gel: get Ara-SupD (ES insert)
2009.9.23
Miniprep the two plasmids.
Enzyme Digestion pSal-PM9 with XbaI and PstI
Digest pSB4K5 with EcoRI and PstI
Gel: get pSal-PM9 (XP insert), pSB4K5 EP backbone.
Ligation:
pSal-PM9(XP insert) 3.5ul
ara-supD(ES insert )3.5ul
pSB4K5 EP vector 1ul
ligase 1ul
ligation buffer 1ul.
Ligation over night.
2009.9.24
Transformation.
2009.9.25
Pick colonies PCR assessment. No result. Maybe due to low copy plasmid.
Shake in the incubator.
2009.9.26
Miniprep.
Enzyme Digestion of the plasmids.
Gel assessment.
Get the correct colony.
2009.9.27
Second plasmid transformation:
Shake the PO-GFP strain in the incubator to OD 0.4
Suspend with CaCl2 and make competent cells.
Transformation with the correct ara-supD-pSal-PM9 plasmid.
Plate over night.
2009.9.28
Shake the transformation from last night.
Shake the ORgate-GFP strain.
Induction.
^Top
|