Team:SJTU-BioX-Shanghai/Characterization
From 2009.igem.org

Project introduction. Inspired by the natural regulator of circadian bioclock exhibited in most eukaryotic organisms, our team has designed an E.coli-based genetic network with the toxin-antitoxin system so that the bacterium oscillates between two states of dormancy and activity (more...)
Part characterization
- Information about RelE toxin+Double terminater([http://partsregistry.org/wiki/index.php?title=Part:BBa_K185004 BBa_K185004])
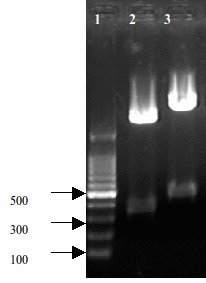
It is a compsite part composed by BBa_K185047 and BBa_B0015. The creator of this part is Alex Jiang. When we insert different promoter ahead of this part, it will express RelE protein on different levels, depending on the strength of promoters. Its length is 443 bp, which can be verified by gel electrophoresis.
- Fig 1. Construct results of RelE+Double terminator
- Lane2: [http://partsregistry.org/wiki/index.php?title=Part:BBa_K185000 BBa_K185000] RelE toxin+Rbs30 311bp
- Lane3: [http://partsregistry.org/wiki/index.php?title=Part:BBa_K185004 BBa_K185004] RelE toxin+Double terminator 443bp
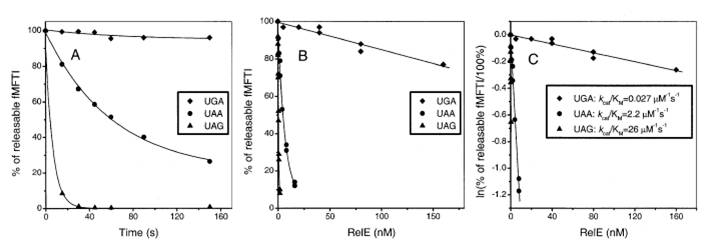
The stop codon of wild type RelE gene is TGA. Here we mutated it into TAA, according to inhibition specifity towards different stop codons: UAG>UAA>UGA. The rates of the inhibition reactions in all cases were proportional to the relE concentration and thus determined by kcat/Km. The kcat/Km Values (s-1μM-1) for RelE Cleavage of 3 stop codons in the Ribosomal A Site have been measured previously. (UGA:0.078, UAA:2.2, UAG:26).
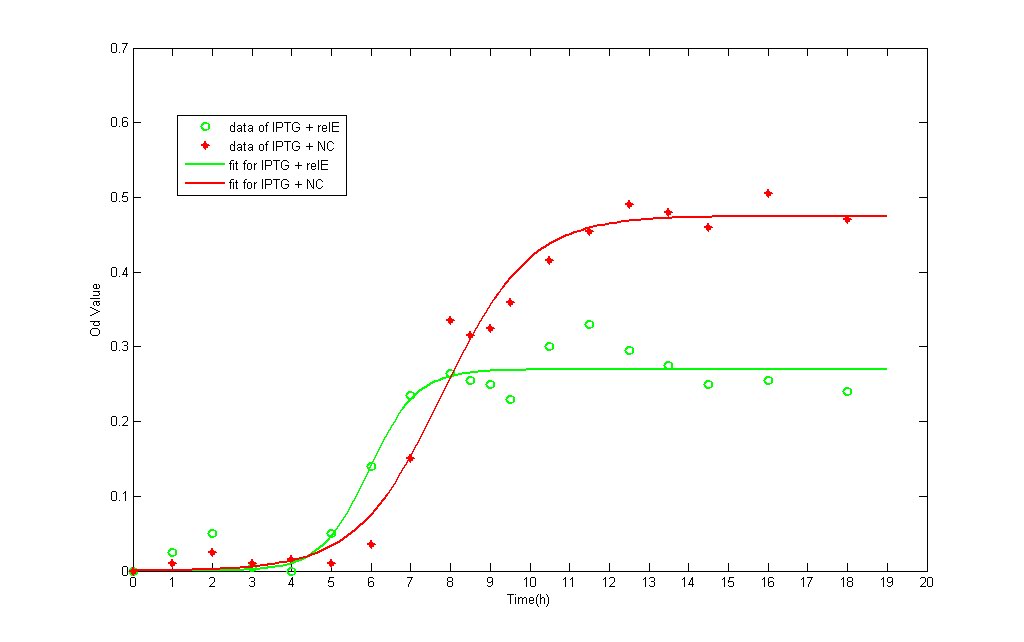
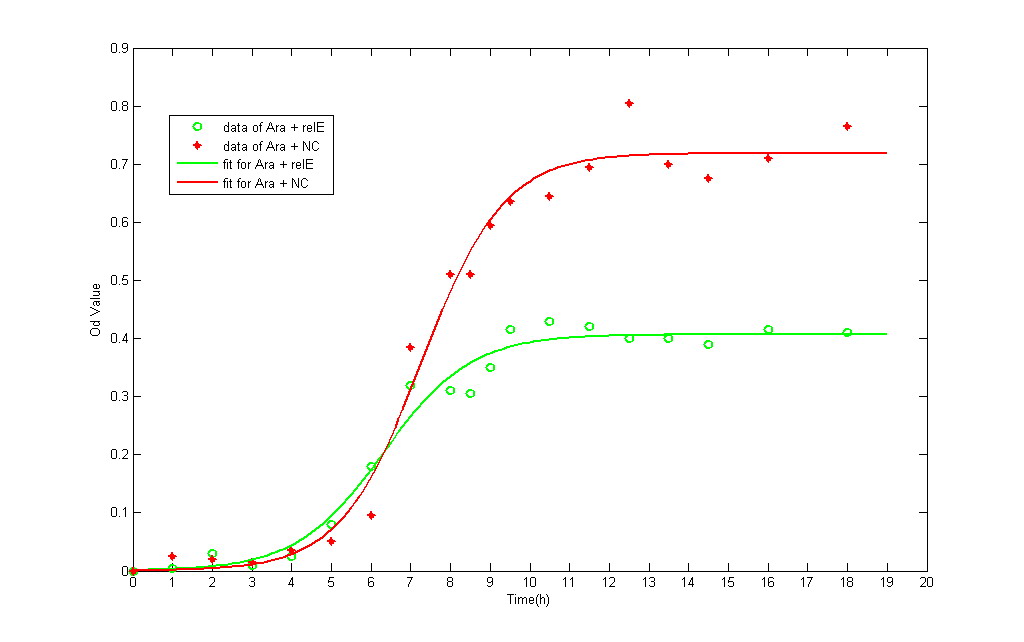
We have test this part under two different promoters: pLac and pBAD. After identification of reconstruced plasmid, we transfected the plasmid into E.coli BL21 strain and then incubated it into M9 medium. All the liquid M9 medium contains 0.1% IPTG and 0.1% Amp(In pBAD strain, 0.2% L-arabinose and 0.1% Amp), in order to keep the same growing conditions. The total amount of bateria is detected by spectrophotometer. It provides the real-time detecting data of bacteria amount under OD600.
- The growth data and fitting curve (according to logistic formulation) are demonstrated below:
- The green line represents the OD value of NC, whereas the red one represents RelE strain.'
- The green line represents the OD value of NC, whereas the red one represents RelE strain.
The growth curve of both NC and RelE strain fit the typical bacteria growth pattern, including the lag phase, exponential phase, stationary phase, and death phase. However, the exponential phase (the most rapid phase in which bateria grows) in RelE strain shows a shorter period than in NC group. The shorter exponential period eventually results in lower concentration of bacteria in stationary phase. Consequensely, we can verify that expression of the relE gene has been shown to severely inhibit bacteria growth and prevent colony formation.
Next, go to Human practice of our project.
 "
"