From 2009.igem.org
|

|
December 2008
| M | T | W | T | F | S | S |
|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 |
|
| 8 | 9 | 10 | 11 | 12 | 13 | 14 |
|
| 15 | 16 | 17 | 18 | 19 | 20 | 21 |
|
| 22 | 23 | 24 | 25 | 26 | 27 | 28 |
|
| 29 | 30 | 31 | | | | |
|
|
March 2009
| M | T | W | T | F | S | S |
|
| | | | | | | 1 |
|
| 2 | 3 | 4 | 5 | 6 | 7 | 8 |
|
| 9 | 10 | 11 | 12 | 13 | 14 | 15 |
|
| 16 | 17 | 18 | 19 | 20 | 21 | 22 |
|
| 23 | 24 | 25 | 26 | 27 | 28 | 29 |
|
| 30 | 31 | | | | | |
|
|
April 2009
| M | T | W | T | F | S | S |
|
| | | 1 | 2 | 3 | 4 | 5 |
|
| 6 | 7 | 8 | 9 | 10 | 11 | 12 |
|
| 13 | 14 | 15 | 16 | 17 | 18 | 19 |
|
| 20 | 21 | 22 | 23 | 24 | 25 | 26 |
|
| 27 | 28 | 29 | 30 | | | |
|
|
May 2009
| M | T | W | T | F | S | S |
|
| | | | | 1 | 2 | 3 |
|
| 4 | 5 | 6 | 7 | 8 | 9 | 10 |
|
| 11 | 12 | 13 | 14 | 15 | 16 | 17 |
|
| 18 | 19 | 20 | 21 | 22 | 23 | 24 |
|
| 25 | 26 | 27 | 28 | 29 | 30 | 31 |
|
|
|
|
|
|
|
|
|
November 2009
| M | T | W | T | F | S | S |
|
| | | | | | | 1 |
|
| 2 | 3 | 4 | 5 | 6 | 7 | 8 |
|
| 9 | 10 | 11 | 12 | 13 | 14 | 15 |
|
| 16 | 17 | 18 | 19 | 20 | 21 | 22 |
|
| 23 | 24 | 25 | 26 | 27 | 28 | 29 |
|
| 30 | | | | | |
|
|
|
|
Week from June 22nd, to June 28th, 2009
June, 22nd
- This week we planned to ligate the GFP protein generator under the control of Ptet in A4 and A6 constructs, in order to have:
- an aTc->GFP measurement system (A6-E0240 = J23100-RBS-tetR-TT-Ptet-RBS-GFP-TT) and
- a control construct that should always express GFP(A4-E0240 = RBS-tetR-TT-Ptet-RBS-GFP-TT)
- Moreover, we planned to begin the construction of a lysis actuator with K117000 (=celB) BioBrick and also a test construct with J23118 to assay GFP expression.
- We infected 5 ml of LB + Amp with 10 ul of these glycerol stocks (to prepare samples for this week's ligations):
| K117000
| E0240 (X5)
| J23118
|
| A4
| A6
| B0015
|
- We incubated the 5 ml cultures overnight at 37°C, 220 rpm.
- We transformed BOL1 plasmid in TOP10 and plated transformed bacteria in a LB + Amp agar plate. We incubated the plate overnight at 37°C.
- We ordered pdc and adhB coding sequences from Zymomonas mobilis to Mr Gene. They had the following specifications:
- the sequences were taken from the online data banks, considering the most recent entries;
- the organism of interest was Z. mobilis CP4 (ATCC 31821);
- the sequences were codon-optimized for E. coli;
- prefix and suffix sequences were added, considering that both genes start with ATG;
- an additional stop codon (TAA) was addad at the end of the coding sequence, just before the suffix, in order to have a double stop codon;
- restriction sites EcoRI, XbaI, SpeI, PstI and NotI had to be avoided during codon optimization (while the original coding sequences didn't have any of them).
June, 23rd
- BOL1 plate showed colonies! We picked a colony from BOL1 plate and infected 1 ml of LB + Amp. We incubated the inoculum at 37°C, 220 rpm for 5 and 1/2 hours.
- Glycerol stock for the grown culture of BOL1.
- We re-filled the remaining 250 ul of BOL1 culture with 5 ml of LB + Amp and incubated the culture overnight at 37°C, 220 rpm. The next day it will be miniprepped and sent to BMR for sequencing!
- Miniprep for: E0240 (5 samples), A4, A6, B0015, J23118 and K117000
- Digestions:
| E0240(X-P) (5 samples)
| J23101(E-S)
| K117000(E-S)
|
| A4(S-P)
| A6(S-P)
| B0015(E-X)
|
- The bands were cut from the gel and the following ligations were performed:
- A7 = J23118(S-P) + E0240(X-P)
- A8 = A4(S-P) + E0240(X-P)
- A9 = A6(S-P) + E0240(X-P)
- A10 = K117000(E-S) + TT(E-X)
- The ligation reactions were incubated overnight at 16°C.
|
|
 Elisa and Letizia working on our wiki. |
June, 24th
- Miniprep for BOL1. We also sent the purified plasmid to BMR Genomics for sequencing.
- We resuspended R0011 (=Plac) and K112808 (=lysis actuator from T4 phage) from the iGEM plates with 15 ul of RNAse free water.
- Trasformations in TOP10 bacteria were done for: A7, A8, A9, A10, K112808 and R0011. We plated the transformed bacteria and incubated the plates overnight at 37°C.
- Team meeting
- We received sequencing results for:
- K131009 - MIT sequencing was confirmed: celB had a point mutation which changes 1 amino acid L->M, while both prefix and suffix had a nucleotide deletion (anyway, the restriction sites were not corrupted). We will document it on K131009 page, but by now we don't know if it is a good idea to use this BioBrick...
- A3 - correct! (long part, but lacZ had already been successfully tested)
- A4 - correct!
- A5? - correct! (long part, but lacZ had already been successfully tested), we were very lucky: the colony had been chosen randomly from the A5 plate:):) Now "A5?" will be called "A5"!
- A6 - correct!
June, 25th
- All the overnight plates showed colonies!
- We picked a colony from R0011 and K112808 plates to infect 1 ml of LB + Amp. We incubated the two inocula at 37°C, 220 rpm for 5 and 1/2 hours.
- Glycerol stocks for R0011 and K112808. The remaining 250 ul of R0011 grown culture had been re-filled with 5 ml of LB + Amp to grow an overnight culture (37°C, 220 rpm).
- We also infected 5 ml of LB + Amp with 10 ul of BOL1 glycerol stock to grow an overnight culture (37°C, 220 rpm).
- Colony PCR for:
- A7 - 3 colonies (few colonies because we picked non red coloured colonies)
- A8 - 8 colonies (important ligation!)
- A9 - 12 colonies (important ligation!)
- A10 - 3 colonies (it's quite unuseful to screen this ligation, because probably we are not able to discriminate the positive and the negative transformants)
- The picked colonies used in the PCR were inoculated in 1 ml of LB + Amp and incubated at 37°C, 220 rpm, waiting for the gel results.
- We performed two different PCR programs:
- one for A7 (expected size of positive plasmid: ~1 Kb) and A10 (expected size of positive plasmid: ~600 bp), with 3 min elongation time
- and a different one for A8 and A9 (expected size of positive plasmids: ~2 Kb for both).
- Electrophoresis for the resulting reactions.
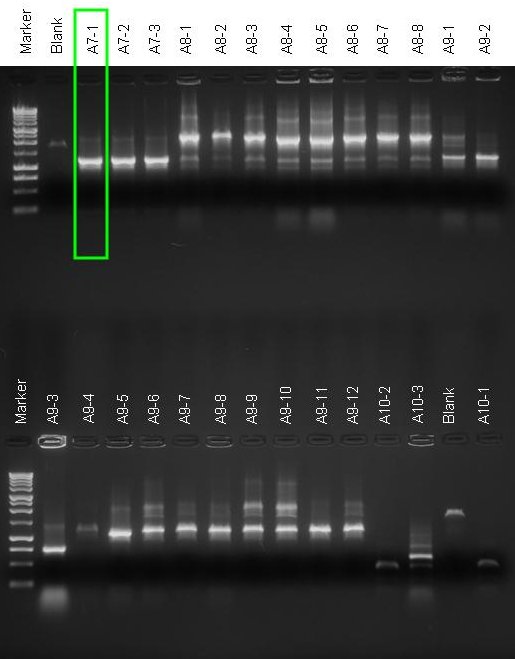 Colony PCR on A7, A8, A9, A10: we chose the 1st screened colony for A7 (A7-1), while we did not choose any colony for the other ligations because they did not look correct. |
- Gel results:
- A7 - all colonies were good, we kept A7-1 to store a glycerol stock.
- A8, A9 and A10 unfortunately showed extra bands. So, we didn't keep any colony for them.
- Blank reaction also showed a contaminant band.
- We planned:
- not to care about A10 at the moment because the priority was to test aTc sensing device with A8 and A9;
- to re-examinate the gel results to explain the lengths of the extra bands (on Monday);
- to re-transform the ligations (stored at -20°C) because probably more than one plasmid had been incorporated in the screened colonies...
Preparation of experiment with Tecan F200
- We infected 5 ml of LB + Amp with 10 ul of:
- T9002 (the following day it will be diluted and induced with different concentrations of 3OC6-HSL)
- E0240 (negative control)
- A1 (positive control)
- glycerol stocks. We incubated the inocula overnight at 37°C, 220 rpm.
June, 26th
 Susanna resuspending pellets during miniprep. |
- Miniprep for R0011 and BOL1. The purified plasmids were stored at -20°C ready to be cut!
|
- We diluted 1:20 the A8 and A9 ligations, which were stored at -20°C, and transformed 1 ul of the dilution in TOP10 E. coli. We plated the transformed bacteria on LB + Amp agar plates and incubated the two plates at 37°C overnight.
|
|
Preparation of experiment with Tecan F200
- We diluted 1:1000 the overnight cultures of:
- T9002 (24 different 5 ml cultures)
- E0240 (one 5 ml culture)
- A1 (one 5 ml culture)
- We induced the 24 cultures of T9002 with eigth different concentrations of 3OC6-HSL (three 5 ml cultures for each concentration): 0 nM, 0.1 nM, 1 nM, 10 nM, 100 nM, 1 uM, 10 uM, 100 uM.
- We incubated the cultures overnight at 37°C, 220 rpm for 3 hours.
June, 27th
- The two overnight plates showed colonies. We put the two grown plates at +4°C: next week they will be screened!
Experiment with Tecan F200

 Previous Week
Previous Week

 Previous Week
Previous Week

 "
"



