Team:Brown/Project Implications
From 2009.igem.org
Human Practices
Safety Issues
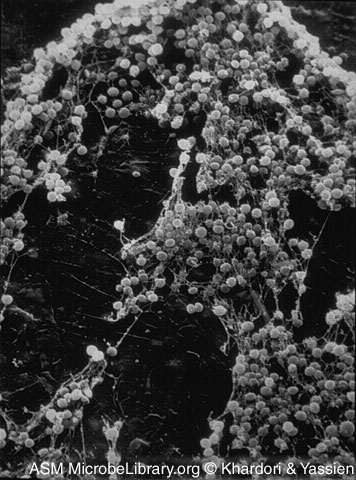
This project raises safety issues due to the use of Staphylococcus epidermidis for producing and secreting the histamine binding protein, EV131. Although S. epidermidis is one of the more benign species of Staphylococcus, it can form infectious biofilms that are impervious to antibiotic treatment if its cell density becomes too great. This could pose a grave risk to researchers involved and the public for whom Allergene aims to serve.
Institutional Biosafety Regulation
Any member of the Brown University faculty, staff, post-doctoral, and student bodies who plans to initiate research involving human subjects must submit a protocol for review and approval by the Institutional Review Board (IRB) prior to beginning the project. The Research Protections Office (RPO) staff provides administrative support to researchers who are preparing protocols for presentation to the IRB.
The Institutional Animal Care & Use Committee (IACUC) ensures the health, well-being, and humane use of animals used in research at Brown, following the regulations and guidance of the U.S. Department of Agriculture and the Department of Health and Human Services. IACUC staff is available to answer administrative questions, and campus veterinarians can provide consultation and assistance on animal care and questions regarding experimental protocols. For our project, however, we have not yet proceeded to preclinical testing stage. For in vivo research, the Office of Environmental Health and Safety (EHS) at Brown provides compliance tools to optimize laboratory and facility conditions across research projects, from safe handling of potentially hazardous materials to biosafety training. EHS also has created guidelines to describe its emergency response system, environmental strategies, and lab equipment rules.
We presented and proposed our project to the Office of Environmental Health and Safety at Brown University. Initially, we ran into concerns about the Biosafety Level 2 classification of the proposed bacteria, Staphylococcus aureus, that we planned to use for expression of the histamine binding protein. With further consideration of the biohazard implications of S. aureus, we decided instead to utilize Staphylococcus epidermidis for the expression of the histamine binding protein. Using S. epidermidis provided us with two major benefits:
== 1) S. epidermidis is endogenous to the nasal flora and thus, very well-suited for accomplishing the eventual goal of our project.
2) S. epidermidis is Biosafety Level 1, eliminating the fears that would have been associated with a Biosafety Level 2 bacterium. ==
 "
"