Team:SJTU-BioX-Shanghai/Result eucaryote
From 2009.igem.org

Project introduction. Inspired by the natural regulator of circadian bioclock exhibited in most eukaryotic organisms, our team has designed an E.coli-based genetic network with the toxin-antitoxin system so that the bacterium oscillates between two states of dormancy and activity (more...)
Experiments on eukaryotic cell
The circadian clock is an endogenous biological oscillator found widely throughout the tree of life. Circadian rhythms impact many behavioral and physiological processes, allowing for the anticipation of daily changes in the environment, the coordination of events that need to occur simultaneously or sequentially, and the segregation of activities that should be separated. At the organism level, these 24-hour cycles are manifested diversely, occurring in the movement of leaves and the opening of flowers, spore formation in Neurospora, and blood pressure and body temperature, as well as hunting, foraging, mating, and sleeping behavior in mammals. Clearly, because of the widespread occurrence of the circadian clock and its effects at all levels of organization, this system is an essential component of biological knowledge.
Inspired by the natural regulator of circadian bioclock exhibited in most eukaryotic organisms, our team has designed an E.coli-based genetic network with the toxin-antitoxin system so that the bacterium oscillates between the states of dormancy and activity. We want to control the circadian rhythm of bacteria using this artificial bioclock. Interestingly, we think that this system may be used in mammalian cell to realize many human practices including delaying aging, controlling the progress of cancer and curing the depression et al. Therefore, we have tried to test the relE-relB system effect in the mammalian cell line.
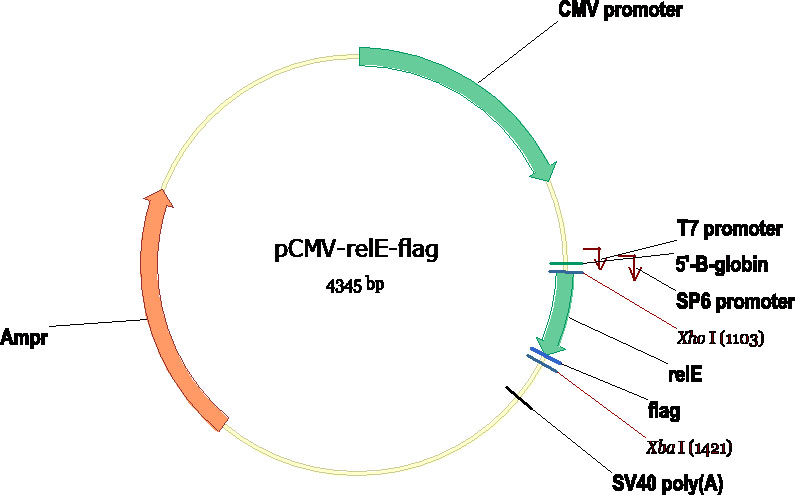
We have generated two constructs including pCMV-relE-flag and pIND-relB-flag (Fig. 1), the latter plasmid will be induced to express the relB protein in EcR-CHO cells (Invitrogen) when adding the Ponasterone A (Fig.2). These two plasmids were co-transfected in EcR-CHO cell line, then the expression of relB were induced and uninduced. If uninducing, the expecting result is that the cells only with relE protein will gradually show the apoptosis phenotype. However, if inducing with PA, the expression of relB will rescue the apoptosis. We can not get the final result as the limitation of time, but we will continue to conduct the experiment to identify the hypothesis if the relE-relB system indeed can be used in mammalian cell to realize the human practices.
- Fig. 1 The constructs of pIND-relB-flag and pCMV-relE-flag.
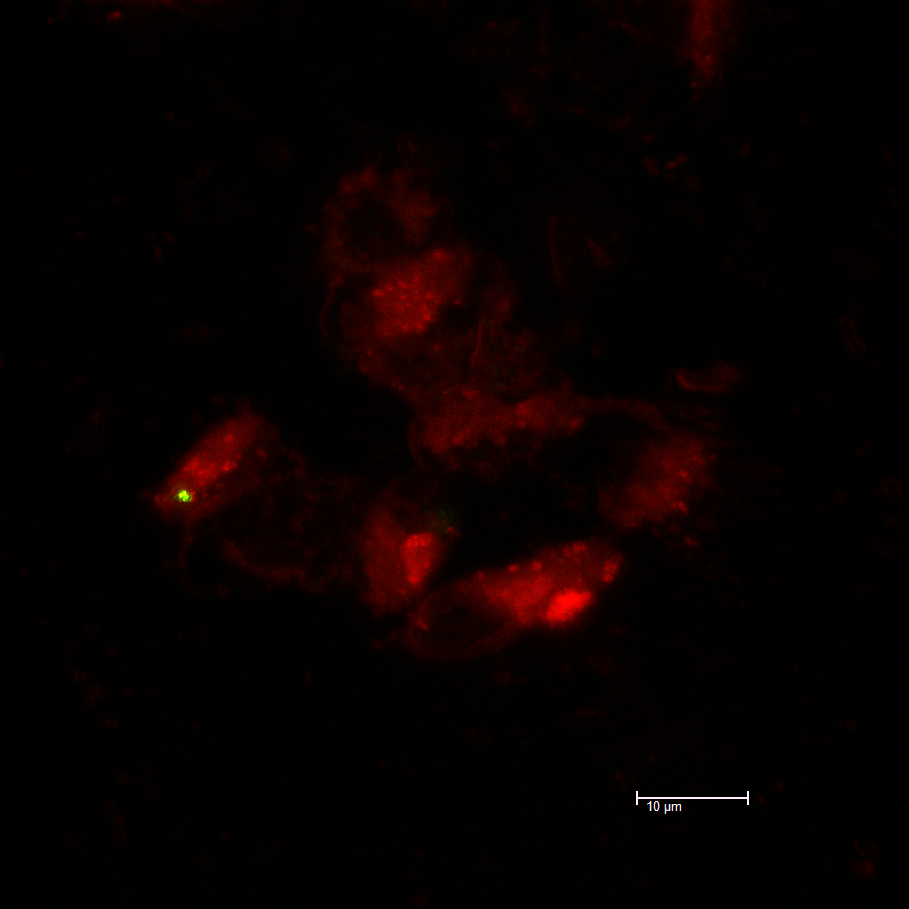
- Fig. 2 The immunofluorescent assay of relB and relE in the EcR-CHO cells.
EcR-CHO cells were grown on glass slides to reach 90% confluency and transfected with relB and relE in a pIND vector and pCMV vector respectively, and after 24h fixed in 80% acetone, and were then incubated for 60min at 37°C in the PBS with anti-flag monoclonal antibody. FITC-labeled goat anti-mouse IgG (Santa Cruz Biotechnology) was used as a secondary antibody. The immunofluorescent staining signal was identified by confocal microscopy (Leica TCS SP5). The cells were stained with Evans Blue. The white scale bars represent 10μm.
Next, go to Parts & Devices.
 "
"