Notebook > AND Gate 1 > Core > Molecular cloning: lacP+SupD/GFP, Sal+SupD/GFP
Molecular cloning: lacP+SupD/GFP, Sal+SupD/GFP
Parts:
lacP (R0010)+SupD-term (K228001+B0015)=lacP-SupD-term (K228822)
lacP (R0010)+GFP (E0840)=lacP-GFP (K228821)
Sal+SupD-term (K228001+B0015)=Sal-SupD-term
Sal+GFP (E0840)=Sal-GFP
Resource:
lacP: from parts, myself,
SupD-term: from Lin Min, has digested by EcoR1 and Xba1,
GFP: from Lin Min, has digested by EcoR1 and Xba1;
Sal: from a plasmid from Lin Min;
2009.8.17
Plasmid mini prep:
lacP
Double digest:
LacP:
| EcoR1 | 1uL
|
| plasmid | 10uL
|
| Buffer | 2uL
|
| water | 6uL
|
37 ℃ 4 hour
PCR: (sal)
| System | 20 uL
|
| pfu enzyme | 1ul
|
| primer (with standard prefix and suffix) | 1uL each
|
| Buffer | 2 uL
|
| water | 10uL
|
| template (the plasmid content Sal) | 1uL
|
| dNTP | 4uL
|
Gel electrophoresis:
Products of double digest of L and PCR
Marker: 100bp 250bp 500bp 750bp 1kb 2kb 3kb 5kb
Loading buffer and DNA dye: 6×
Voltage and time: 60V 5min; 120V 15min
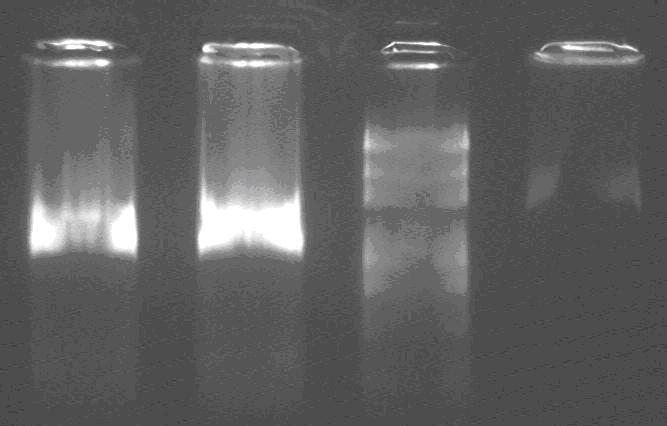
Lane 1&2: Sal, PCR product,
Lane 3: Marker,
Lane 4: lacP, digest product
Sal should be about 1.4kb, and the result is correct!
After digested, lacP should have a 200bp one, but I can not see it. So I decided to repeat to amplify the Ecoli and mini prep.
DNA Gel purification:
Sal
Double digest:
Sal:
| Spe1 | 1uL
|
| EcoR1 | 1uL
|
| plasmid | 10uL
|
| Buffer | 2uL
|
| water | 6uL
|
37 ℃ overnight
2009.8.18
PCR product purification:
Sal
DNA ligation:
| System | 10uL
|
| Insert | 6uL
|
| vector | 2uL
|
| buffer | 1uL
|
| T4 DNA ligase | 1uL
|
16℃ 4 hour
Insert: Sal;
Vector: GFP and SupD
Transformation:
Products of ligation (Sal-SupD, Sal-GFP), competent cells 50uL each,
Smear to LB plate with Amp
Plasmid mini prep: (again)
lacP
Double digest: (again)
LacP:
| Spe1 | 1uL
|
| EcoR1 | 1uL
|
| plasmid | 10uL
|
| Buffer | 2uL
|
| water | 6uL
|
37 ℃ overnight
2009.8.19
Gel electrophoresis:
Products of double digest of lacP,
Marker: 100bp 250bp 500bp 750bp 1kb 2kb 3kb 5kb
Loading buffer and DNA dye: 6×
Voltage and time: 60V 5min; 120V 15min
Lane1: lacP: insert 200bp;
Lane2: Marker
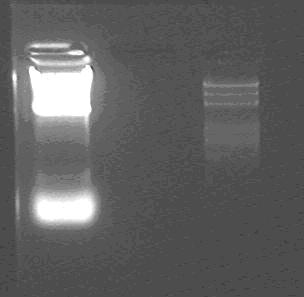
We can find that the smallest but bright one may be RNA, and a little larger and dim one is the insert.
DNA Gel purification:
Insert of lacP
DNA ligation:
| System | 10uL
|
| Insert | 6uL
|
| vector | 2uL
|
| buffer | 1uL
|
| T4 DNA ligase | 1uL
|
16℃ 4 hour
Insert: lacP;
Vector: GFP and SupD
Transformation:
Products of ligation (lacP-SupD, lacP-GFP), competent cells 50uL each,
Smear to LB plate with Amp
PCR: (colony PCR to check the Sal-GFP and Sal-SupD)
| System | 10 uL
|
| Master mix | 5ul
|
| primer (standard primers) | 0.5uL each
|
| water | 4uL
|
| template |
|
Double digest: (prepare lacP as a vector)
LacP:
| Spe1 | 1uL
|
| Pst1 | 1uL
|
| plasmid | 10uL
|
| Buffer | 2uL
|
| water | 6uL
|
37 ℃ 4 hour
Gel electrophoresis:
Products of double digest of lacP, and PCR Sal
Marker: 100bp 250bp 500bp 750bp 1kb 2kb 3kb 5kb
Loading buffer and DNA dye: 6×
Voltage and time: 60V 5min; 120V 15min
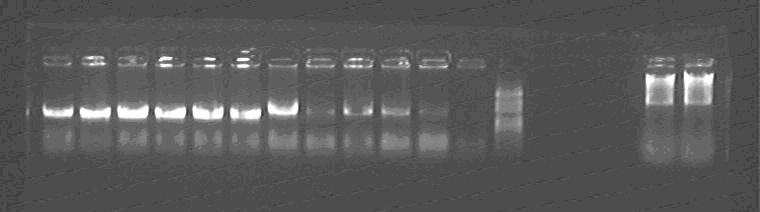
Lane1~6: Sal-GFP1~6, should be 2.4kb, all are wrong;
Lane7~11: Sal-SupD1~5, should be 1.7kb, all are correct;
Lane12: negative control;
Lane13: Marker;
Lane14,15: lacP vector, should be 2.3kb, but there are some pollution plasmids?
2009.8.20
Plasmid mini prep:
Sal-SupD
DNA Gel purification:
lacP vector;
The plates of lacP-SupD and lacP-GFP are very good, and the colonies on the lacP-GFP are green. So I do not need to confirm it.
PCR: (colony PCR to check the lacP-SupD)
| System | 10 uL
|
| Master mix | 5ul
|
| primer (standard primers) | 0.5uL each
|
| water | 4uL
|
| template |
|
Gel electrophoresis:
Products of PCR lacP-SupD
Marker: 100bp 250bp 500bp 750bp 1kb 2kb 3kb 5kb
Loading buffer and DNA dye: 6×
Voltage and time: 60V 5min; 120V 15min
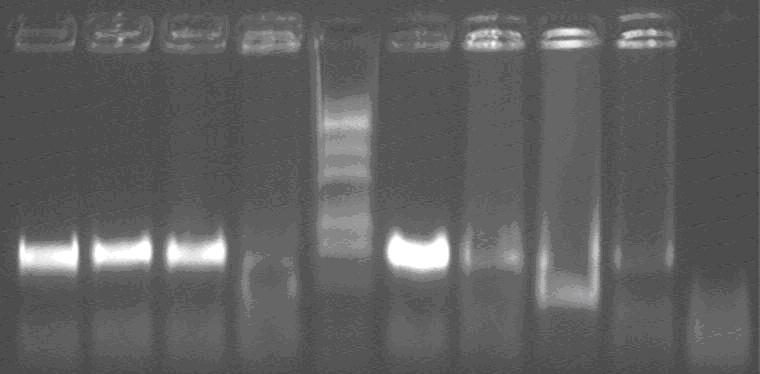
Lane1~4, 6~9: lacP-SupD 1~7;
Lane5: Marker;
Lane10: negative control;
The correct one should about 600bp, and we can find that 1, 2, 3 and 5 are correct.
Result
I successfully constructed the clone: lacP-SupD-term (K228822), lacP-GFP (K2288221) and Sal-SupD-term (which turn out to be wrong after a few days, for more detail, refer to Haoqian Zhang’s notes).
I failed to construct Sal-GFP.
Work transfer
Sal transfers to Haoqian Zhang to do, and lacP vector transfers to ShenShan.
^Top
|