Notebook > AND Gate 1 > Core > Molecular cloning of Sal + AraC AND GATE
Molecular cloning of Sal + AraC AND GATE
2009.8.20
Get Sal plasmids, ES digested insert and EX digested GFP vector from Shuke Wu.
Ligate the Sal pro to GFP part.
Transform the ligated Sal pro + GFP part.
2009.8.21
Pick up and amplify the transformed colonies.
2009.8.22
Induce the amplified colonies
2009.8.23
Test the Sal promoter, but the PBS is not good, thus the testing failed.
2009.8.24
Ligation of Sal promoter to GFP vecot.
ES double digest the Sal promoter+supD part.
Transform the ligated plasmid.
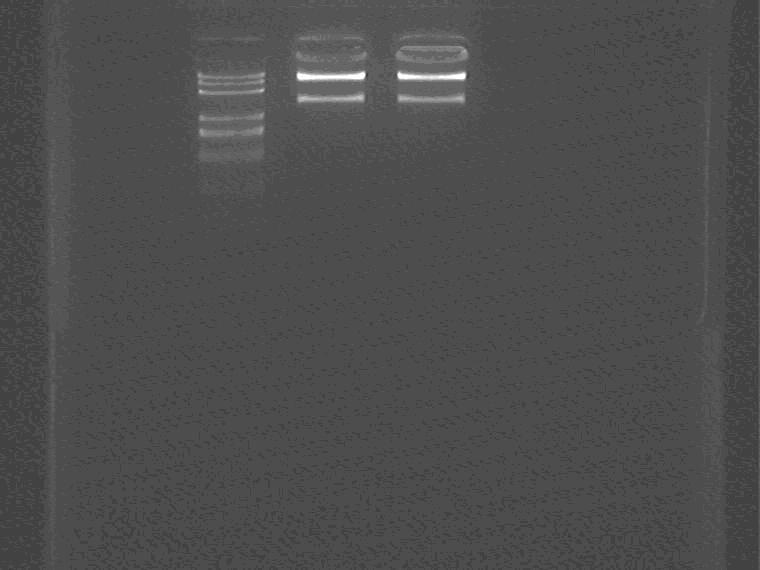
Electrophoresis the ES digested Sal pro+supD insert.
Samples loading order: Marker, control, digested plasmid.
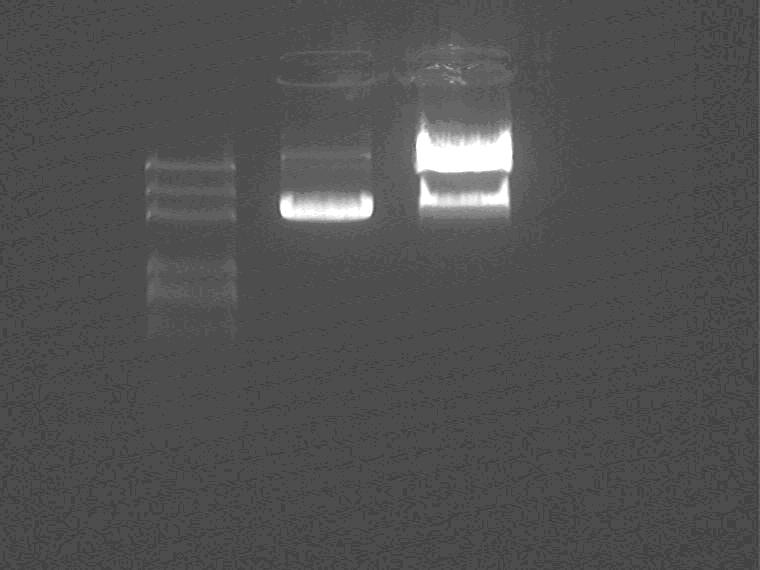
Cut gel extract the insert.
Ligation of Sal pro + supD to 1-1M vector.
2009.8.25
Transforming the ligated Sal pro +supD +1-1M vector
Pick up and amplify Sal pro + GFP colonies.
Test the OD value of the bacteria liquid.
Make a gradient of Sal induction.
Pick up and amplify 5 colonies of Sal pro+supD+1-1M ligated product
2009.8.26
Amplify the colonies of Sal pro + GFP1 again to test the promoter.
Miniprep the Sal pro+supD+1-1M1,4 samples
SP digest the Sal pro + supD + 1-1M 1. 4 plasmids.
2009.8.27
EP digest the Sal pro+supD+1-1M1, 4 plasmids
Electrophoresis the digested 4 samples yesterday.
Samples loading order: Control Sal4 plasmid, Sal4 insert, Marker, control Sal1 plasmid, Sal1 EP insert.
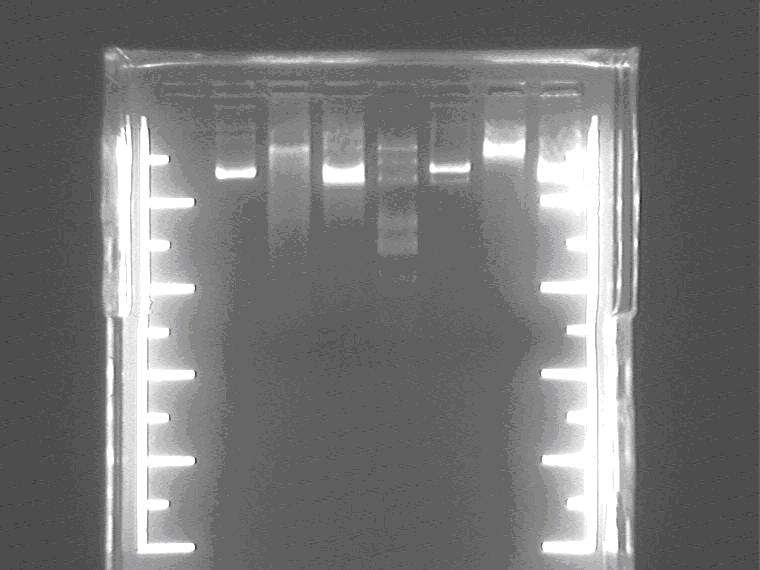
Pick up and amplify 5 colonies of Sal pro +supD+1-1M
Amplify 1-1M, 7E 7G, 9C backbone plasmids.
2009.8.28
Miniprep the Sal pro + supD+1-1M 1-5 and 1M, 7G, 7E, 9C samples.
Electrophoresis to identify the minipreped plasmid.
Samples loading order: 7G, 9C, 7E, 1M, Sal pro + supD+1-1M1-5, Marker
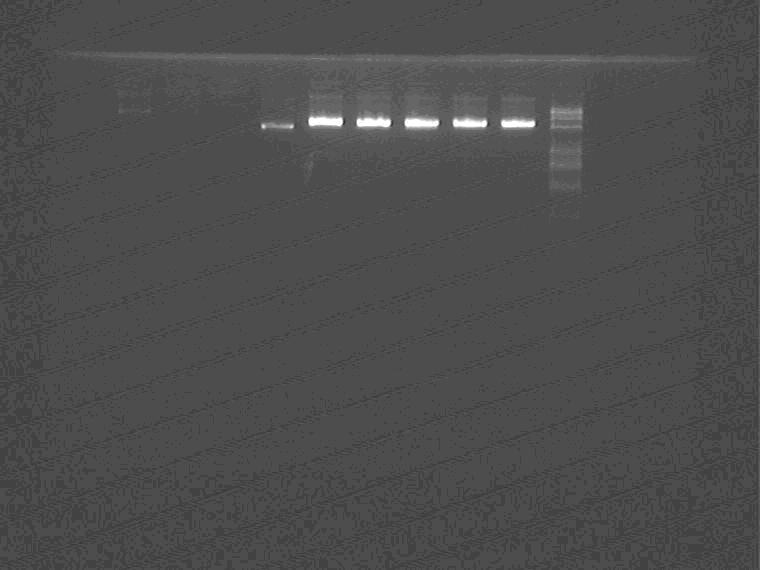
SP digest the Sal pro + supD + 1-1M1-5
Electrophoresis the digested Sal pro+supD+1-1M1-5 samples.
But the digestion itself failed.
I decided to try another strategy of ligation.
SP digest the Sal pro+supD plasmid.
Repick up and amplify 1-7G, 7E, 1M, 9C colonies.
2009.8.29
Electrophoresis the SP digested Sal pro+supD plsmid.
Samples loading order: Marker Sal pro+supD vector Control.
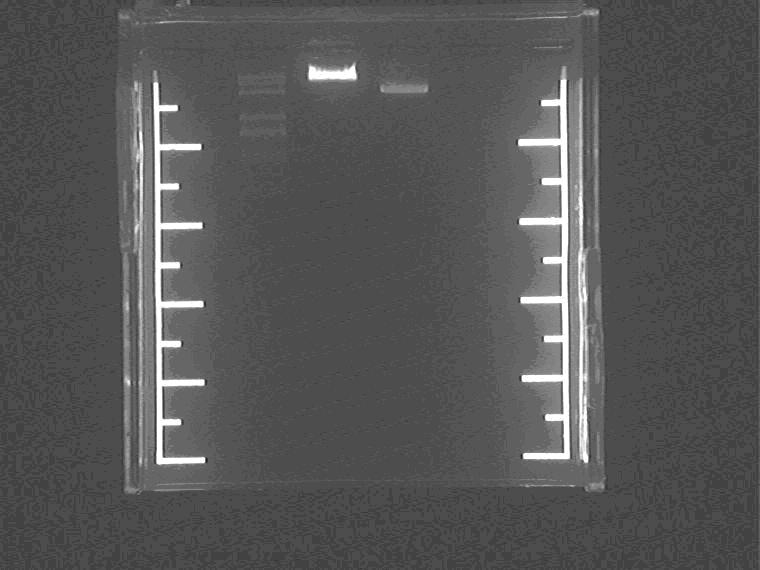
Cut gel and extract the vector.
Miniprep 1-7E, 7G, 9C, 1M plasmids with our own protocol.
Electrophoresis the 1-7G, 9C, 1M plasmids. The result shows that the miniprep failed.
Ligate Sal pro+supD vector to AraC insert(different RBS)
2009.8.30
Transforming the ligation product with DH5α strain.
Pick up and amplify the colonies of Sal+AraC AND GATE(5 colonies for each RBS).
2009.8.31
Miniprep 45 samples. Sal pro+supD+AraC+T7ptag
1-1H 1-2M 1-1J 1-5J 1-11N 1-5N 1-2G 1-2K 1-2I
Electrophoresis the plasmid samples above:
Samples loading order: 1H1-5 1J1-5 2G 1-5 2I1-5 2K1-5 2M1-5 5J1-5 5N1-5 11N1-5
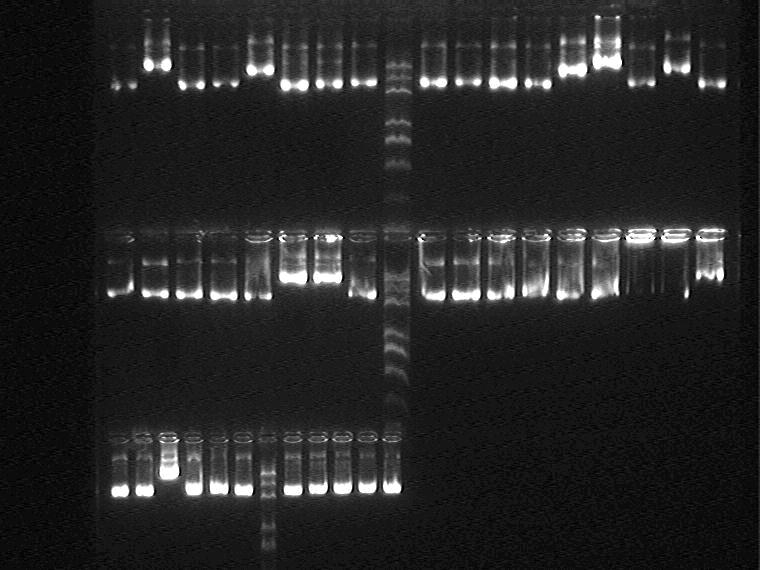
2009.9.1
EP digest to identify 1H2,5 2G3, 4 2I1, 2K, 4 5J4, 5N2
Pick up and amplify these 15 colonies.
PCR identification of these 15 colonies
Electrophoresis the double digested samples.
Samples loading order: 2G3, 4 2K3, 4 1H2, 5 5J4 2I1 5N2 1M Marker Control, 1M plasmid control.
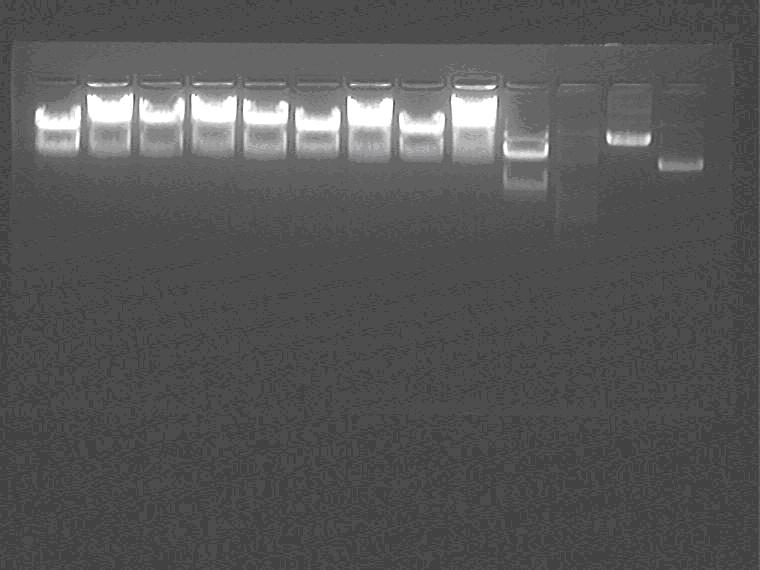
Cut gel and extract 1-1M EP digested vector.
Electrophoresis the PCR identification product.
Samples loading order: 2M1-5 11N1-5 1J15432 Marker
The PCR result shows how serious the pollution is.
2009.9.2
ES double digest of Sal pro+supD; EX double digest of AraC+T7ptag(9 RBS)
Add CIAP before the end.
Electrophoresis AraC+T7ptag(9 RBS) vector
Sal pro+supD insert
Samples loading order: 5J 1H 11N 5N 2K 2I 2G 1J 2M Marker, vector control, insert control, Sal pro insert
Midi purification the vectors(9 RBS)
Ligate Sal pro + supD to AraC+T7ptag(9 RBS)
2009.9.3
Transforming the ligation product yesterday. Sal pro + supD + AraC + T7ptag(9 RBS)
Pick up 45 colonies (5 for each RBS).
PCR identification of these 45 colonies.
2009.9.4
Electrophoresis the PCR identification product
Samples loading order: 2G1-4, 1H1-4 1J1-4, Marker, 5J1-4, 2I1-4, 11N1-4, Marker, 2K1-4, 5N1-4, Marker, 2M1-4
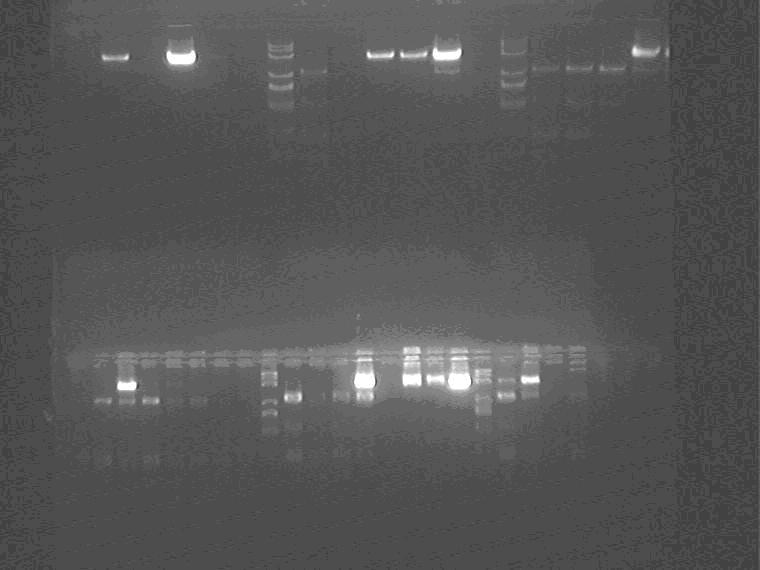
Miniprep 18 samples. 2M4 5N4 2K1 2I3 5J4 2G4 1H1-4 1J1-4 11N1-4
Tansform the ligation of Sal pro+supD+AraC t7ptag 1-2k again.
Pick up and amplify 5 colonies.
PCR identification 1-2K1-5 colonies.
2009.9.5
Electrophoresis the Sal pro+supD+AraC+t7ptag 1-2K1-5 PCR identification product.
Samples loading order: 4 2 1 3 5 marker
EP double and single digest 2M4 5N4 2I4 5J4 2G4 plasmid.
Electrophoresis the double and single digested samples, 12 samples in total.
Samples loading order: 2M4 5N4 M 2I3 5J4 M 2G4
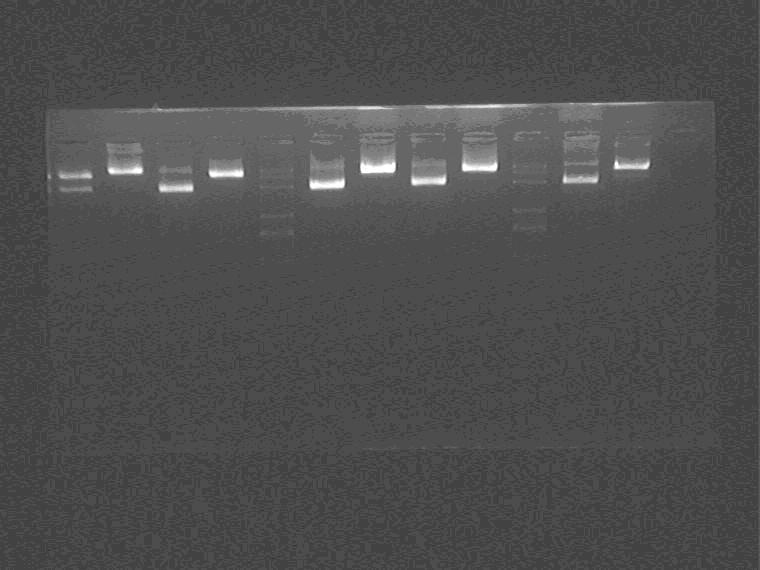
Cut gel and extract AND GATE EP digested insert.
2009.9.7
HSH has digested the relegated Sal pro+supD plasmids.
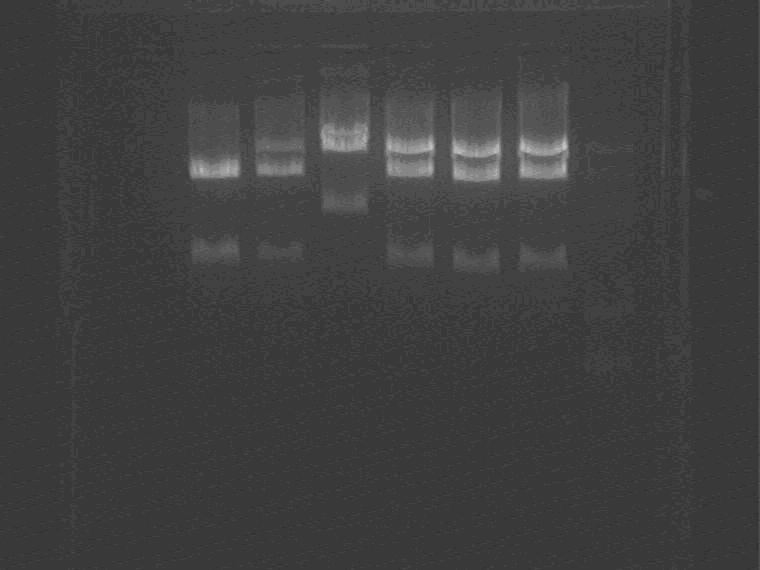
Electrophoresis them.
Samples loading order: Marker 3ES 2ES
Cut gel and extract the Sal pro+supD insert.
Ligating Sal pro+supD insert to AraC+T7ptag, 9 samples in total.
Miniprep Sal pro+supD4, 5 plasmids.
Transform Sal pro+supD insert and AraC+t7ptag ligation product.
2009.9.8
The transformation yesterday didn’t succeed, so I decide to do transformation again.
Pick up and amplify 27 colonies(5 for each RBS, especially 1for 1-1J and 1-2G).
Amplify the 1-7G spawn
PCR identification of these 27 colonies, but the samples were ruined by Lin Min.
ES digest of Sal pro+supD and XP digest of AraC+T7ptag plasmid for insert.
2009.9.9
Miniprep 1-7G plasmid.
Electrophoresis 10 samples in total
Sal pro+supD insert (ES digested); AraC+t7ptag insert(XP digested)
Samples loading order: Sal pro+supD, Sal control, 11N, 1H, 2K, 5J, AraC plasmid Marker 1J 5N 2M 2I 2G
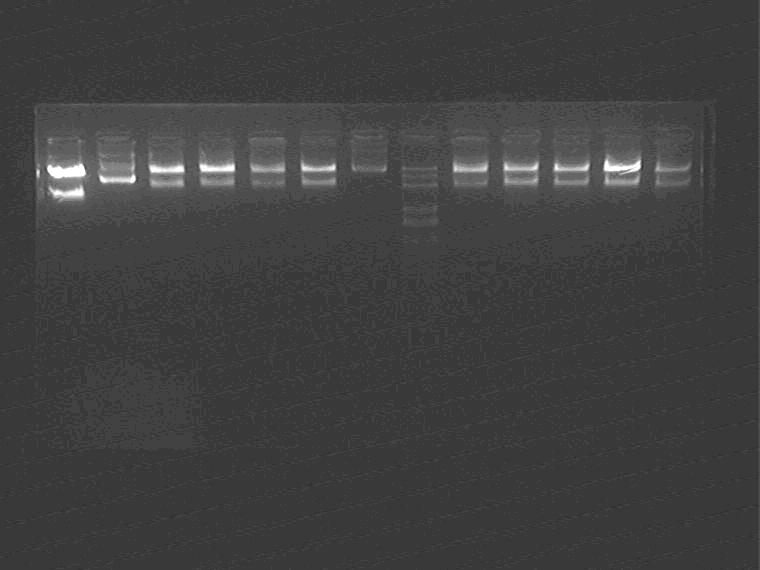
Cut gel and extract the inserts.
EP double digest the 1-7G plasmids.
Electrophoresis 1-7G backbone
Samples loading order: 1, 2, 3, 4, 5, 6, Marker
Ligation of Sal pro+supD, AraC+T7ptag to 1-7G vector.
Miniprep 27 samples of Sal pro+supD+AraC+T7ptag(among which one for 1J and 2G)
Transforming the ligation product: Sal + AraC AND GATE
2009.9.10
EP digest the 27 samples(SA AND GATE 5J1-5 2I1-5 2M1-5 11N1-5 2K1-5 2G1 1J1)
ES digest of Sal pro + supD plasmid(3 indetical samples)
Amplify the Sal pro + GFP plasmid.
Electrophoresis the digested samples
Samples loading order: EP digested 5J1-5 2I1, 2 M 2I3-5 2M1, 2
EP digested 2M 3-5 11N1-3 M 11N4, 5 2K1-4
EP digested 2K5 1J1 2G1 ES digested Sal pro + supD1-3 M
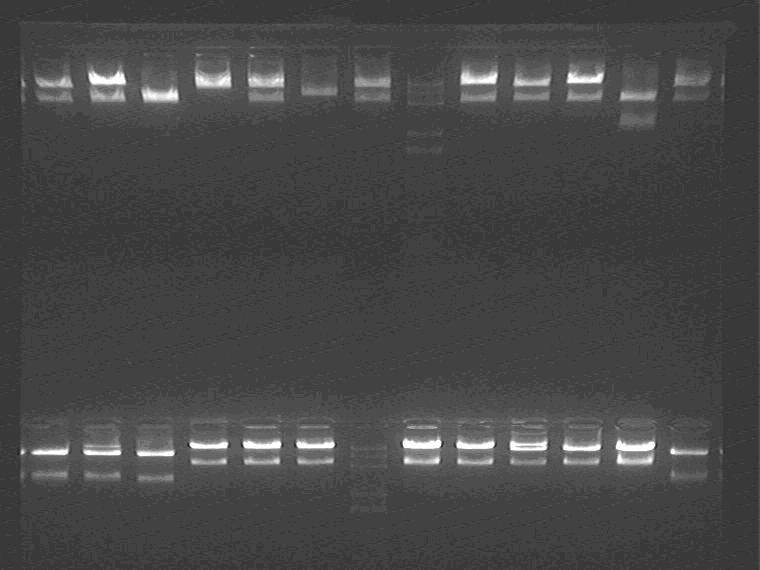
Cut gel and extract the inserts.
2009.9.11
Miniprep T7p+9RBS+CI+T plasmids
EP double digest of T7p+9RBS+CI+T plasmids.
Extract the gel cut yesterday.
Electrophoresis to identify the extracted 12 samples (9 ANF GATEs and 3 Sal pro + supD ES inserts)
Samples loading order: 11N3, 4, 1, 2 2K2, 3 2I3, 5 5J2 Sal1, 2, 3 Marker
Ligate 11N1, 2K2, 2I3, 5J2 to low copy backbone 1-7G
Ligate Sal pro + supD ES insert and AraC+T7ptag insert to 1-7G backbone.
2009.9.12
Transform the ligation done yesterday, 13 samples in total.
2009.9.13
Pick up 3 colonies for each plate of 9 triligation product, and 4 AND GATE low copy backbone ligation product.
PCR identification of these 39 samples
Electrophoresis the PCR identification product. The result shows that the insert may be a large fragment.
Miniprep the 39 samples of AND GATEs
Electrophoresis the PCR identification of AND GATE samples loading order:
2G1-3, 5J1-3, M, 11N1-3, 2K1-3, M, 5N1-3
1J1-3, 2I1-3, M, 1H1-3, 2M1-3, M, 2I1-3
11N1 1-3, M, 2K2 1-3 M, 5J2 1-3
2009.9.14
XP digest of AND GATEs (31 samples)
Electrophoresis the digested samples, 31 samples in total.
Samples loading order: 2K22, 2K21 1H3 2I3 2I1 5J1 2I3 M 5J23 2I2 11N2 2K23 5J2 M 5N2 2K2 11N1 2I43 1H2 5J2 11N3 1J2 M 2K3 5J2 5J21 11N1 M 11N1 5N3 5J3 2M3 2G1 M 2I32 2M2
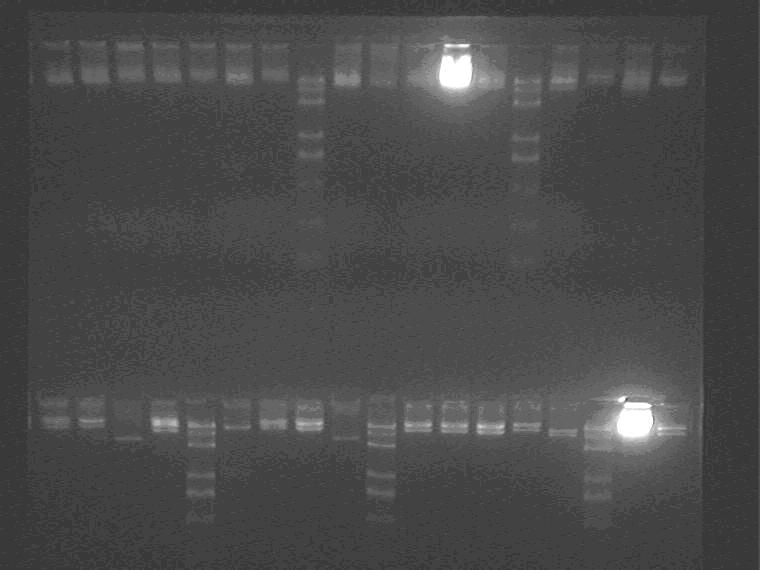
EP digest of 1-7G plasmids.
Make competent cells to transform
Transform 1H3 2I31 11N2 5J2 5N22 2M2 5J21 2I32
2009.9.15
Retransform 1H3 2I31 11N2 5J2 5N2 2M2 5J21 2I32.
Miniprep these plasmids.
^Top
|