Notebook > AND Gate 1 > Core > Molecular cloning for AND GATE: Sal_T7ptag + AraC_SupD
Molecular cloning for AND GATE: Sal_T7ptag + AraC_SupD
2009.8.26
Receive Sal insert digested by two enzymes EcoRI and SpeI, several low-copy backbone plasmids (1-9C,Tc+;1-7G,K+;1-1M,A+) and AraC_SupD plasmid.
Digest 1-9C backbone plasmid into vector
| 1.5μL | EcoRI
|
| 1.5μL | XbaI
|
| 2μL | 10×M buffer
|
| 7μL | plasmid
|
| 8μL | ddH2O
|
| 20μL | Total
|
2009.8.27
Electrophoresis to identify 1-9C vector digested from the plasmid
Result (from left to right)
1-9C①, 1-9C plasmid control①, Marker, 1-9C②, 1-9C plasmid control②
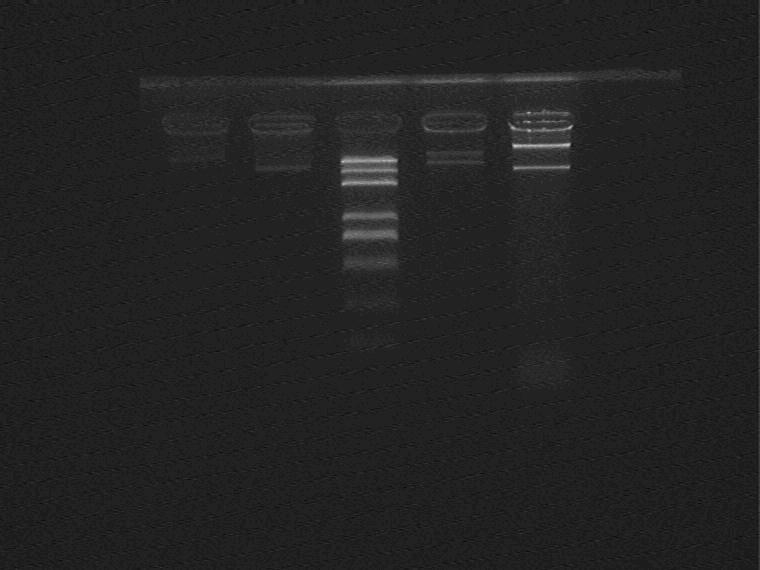
The result is strange, change another backbone
Digest 1-7G backbone plasmid into vector
| 1.5μL | EcoRI
|
| 1.5μL | XbaI
|
| 2μL | 10×M buffer
|
| 5μL | plasmid
|
| 10μL | ddH2O
|
| 20μL | Total
|
Electrophoresis to identify 1-7G vector digested from the plasmid
Result (from left to right)
Marker, 1-7G①, 1-7G②, 1-7G plasmid control
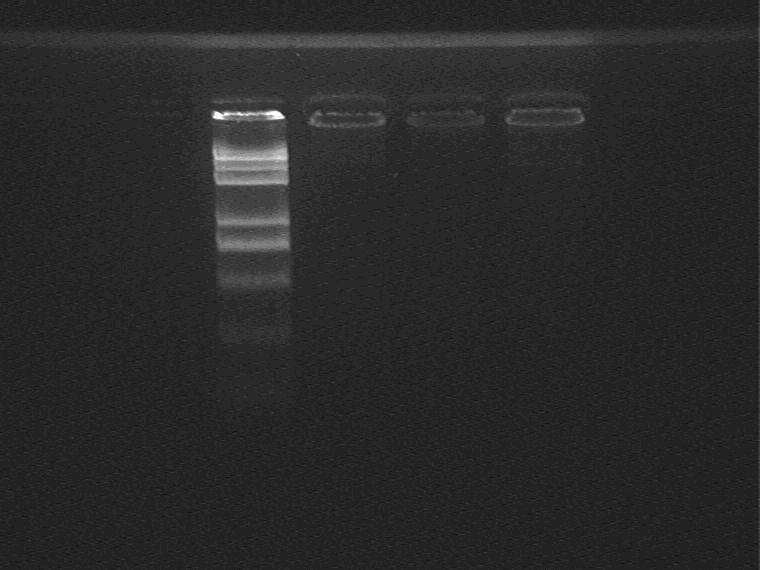
The concentration of 7G plasmid is too low, so the electrophoretic bands are invisible.
2009.8.28
Digest 1-7G backbone vector again and electrophoresis to identify
Result (from left to right)
1-7G, 1-7G plasmid control, Marker,
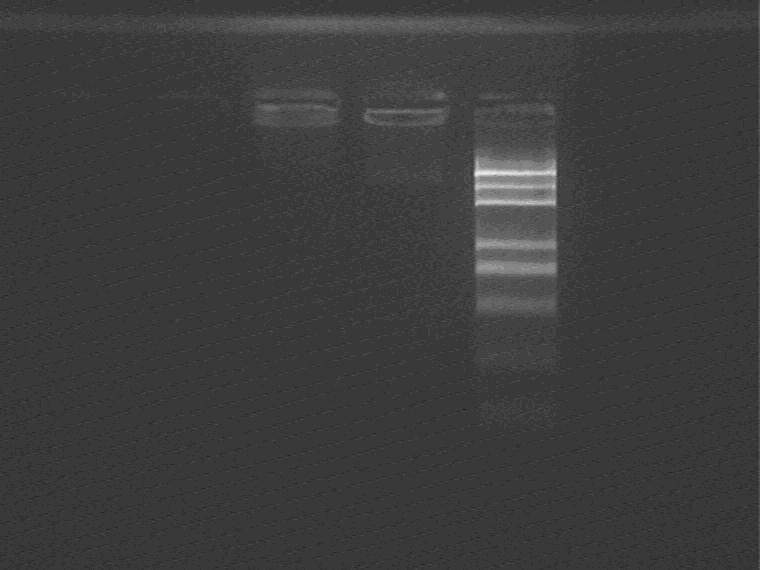
Still failed.
Digest 1-1M backbone plasmid into vector
| 1.5μL | EcoRI
|
| 1.5μL | XbaI
|
| 2μL | 10×M buffer
|
| 15μL | plasmid
|
| 0 | ddH2O
|
| 20μL | Total
|
Electrophoresis to identify 1-1M vector digested from the plasmid
Result (from left to right)
1-1M, 1-1M plasmid control, Marker
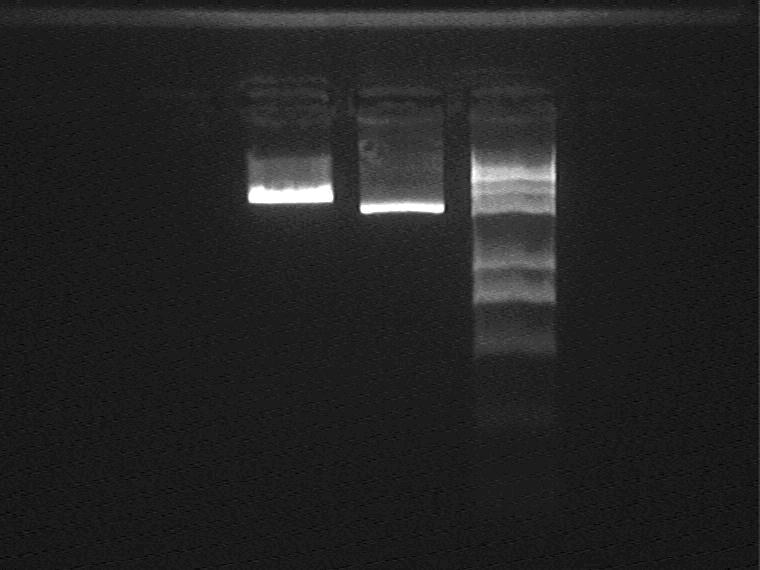
DNA purification to extract 1-1M vector
Ligate the Sal insert and 1-1M vector
System
| 3μL | Sal insert
|
| 1μL | 1-1M vector
|
| 1μL | 10× Ligase buffer
|
| 1μL | Ligase
|
| 4μL | ddH2O
|
| 10μL | Total
|
Transformation for the ligation product: Sal insert + 1-1M vector
2009.8.29
A mistake was made when digesting 1-1M vector: there is a RFP part between restriction site XbaI and SpeI, so 1-1M vector should be digested by EcoRI and SpeI!
Digest 1-1M vector again
| 1.5μL | EcoRI
|
| 1.5μL | SpeI
|
| 2μL | 10×H buffer
|
| 15μL | plasmid
|
| 20μL | Total
|
DNA purification to extract 1-1M vector, ligate the Sal insert and 1-1M vector and transform the ligation product: Sal insert + 1-1M vector
2009.8.31
Miniprep for Sal+1M plasmid
Digest Sal+1M vector
| 1.5μL | SpeI
|
| 1.5μL | PstI
|
| 2μL | 10×H buffer
|
| 6μL | plasmid
|
| 9μL | ddH2O
|
| 20μL | Total
|
The electrophoretic result is strange, so prepare to miniprep again tomorrow.
2009.9.1
Miniprep for Sal+1M plasmid again
Determine the concentration of T7ptag plasmid by spectrophotometer
Sal+1M plasmid A: 5.0174ng/μL×50=250.87ng/μL
Sal+1M plasmid B: 4.1509ng/μL×50=207.55ng/μL
Sal+1M plasmid C: 3.2412ng/μL×50=162.06ng/μL
Digest Sal+1M vector again
Electrophoresis to identify the Sal+1M vector
Result (from left to right)
Sal_1M A, Sal_1M B, Sal_1M C, plasmid control, Marker
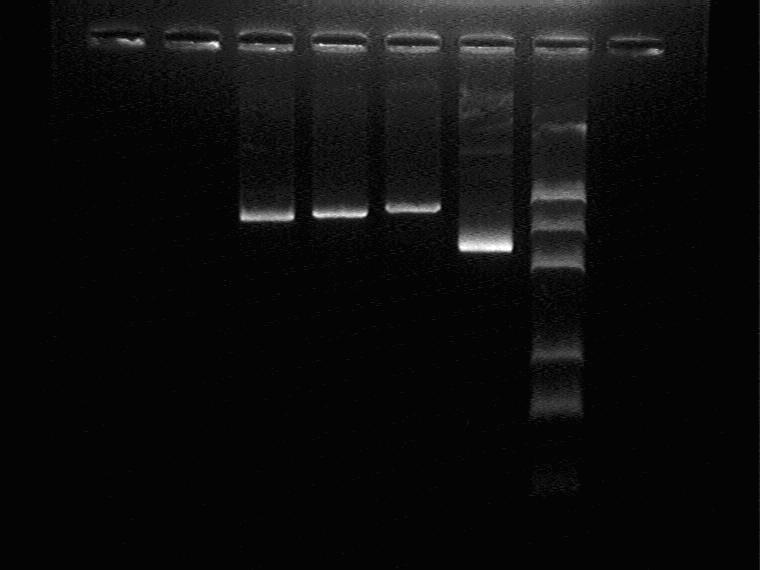
The positions of three digestion products are a little different, so digest the plasmid with XbaI and PstI to identify.
| 1μL | SpeI
|
| 1μL | PstI
|
| 1μL | 10× buffer
|
| 3μL | plasmid
|
| 4μL | ddH2O
|
| 10μL | Total
|
Electrophoretic result (from left to right)
Sal_1M A, Sal_1M B, Marker, Sal_1M C
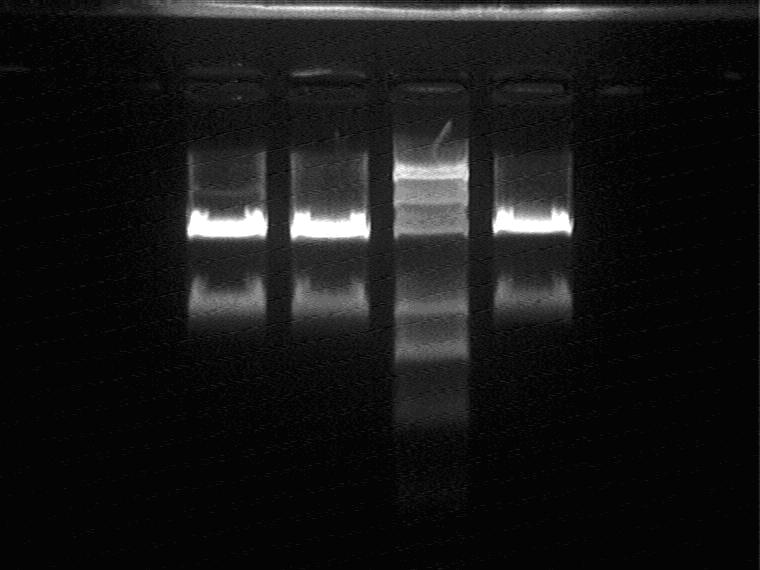
Ligate the RBS+T7ptag+Terminator insert and Sal+1M vector
System
| 3μL | insert
|
| 1μL | vector
|
| 1μL | 10× Ligase buffer
|
| 1μL | Ligase
|
| 4μL | ddH2O
|
| 10μL | Total
|
2009.9.2
Transformation for the ligation product: RBS+T7ptag+Terminator insert and Sal+1M vector
Identify the colonies on the plates (Sal_1M+T7ptag) by PCR
System
| 5μL | Master mix
|
| 0.25μL | Forward primer
|
| 0.25μL | Reverse primer
|
| 4.5μL | ddH2O
|
| | Template (Bacteria Colony)
|
| 10μL | Total
|
2009.9.3
Electrophoresis to identify the PCR product
Result 1 (from left to right, from top to bottom)
1H①~⑤,1J①~④,Marker,1J⑤,2G①~⑤,2I①~②
2I③~⑤, 2K①~⑤,Marker, 2M①,Marker,2M②~⑤,5J①~④
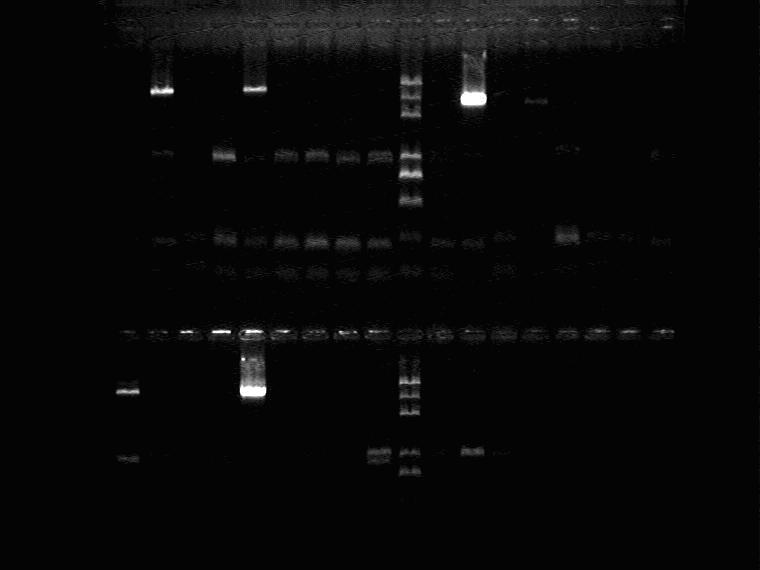
Result 2 (from left to right)
5J⑤,5N①~⑤,Marker,11N①~⑤
Choose 27 samples to miniprep
1H②④⑤,1J①②④,2G②③④,2I②③⑤,2K①②③,
2M①②③,5J③④⑤,5N①②④,11N①②⑤
Double digestion for Sal+T7ptag insert
| 1μL | XbaI
|
| 1μL | PstI
|
| 2μL | 10×M buffer
|
| 5μL | Sal+T7ptag plasmid
|
| 11μL | ddH2O
|
| 20μL | Total
|
Double digestion for AraC_SupD vector
| 1.5μL | SpeI
|
| 1.5μL | PstI
|
| 2μL | 10×H buffer
|
| 4μL | plasmid
|
| 11μL | ddH2O
|
| 20μL | Total
|
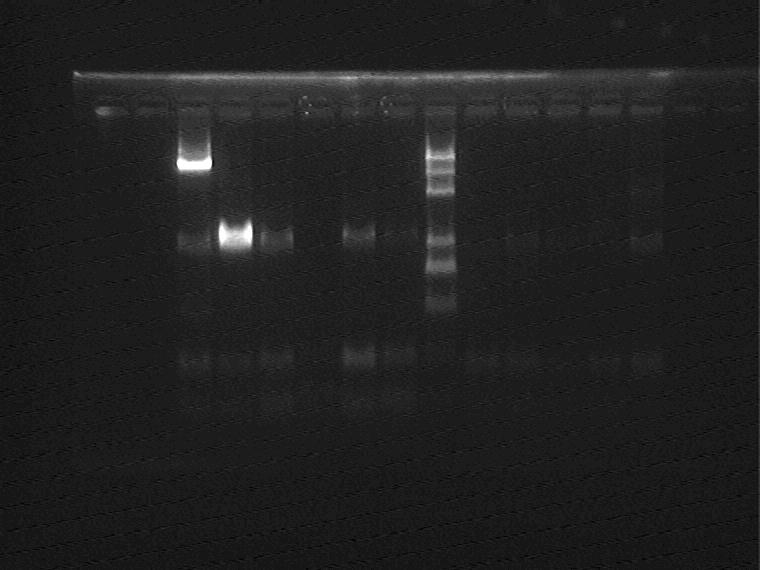
Electrophoresis and Gel Extraction for Sal+T7ptag insert
Result (from left to right, from top to bottom)
1H②④⑤,1J①②④,2G②,Marker,2G③④,2M①②③,2I②③
2I⑤,2K①②③,5J③④⑤,Marker,5N①②④,11N①②⑤,AraC_SupD vector
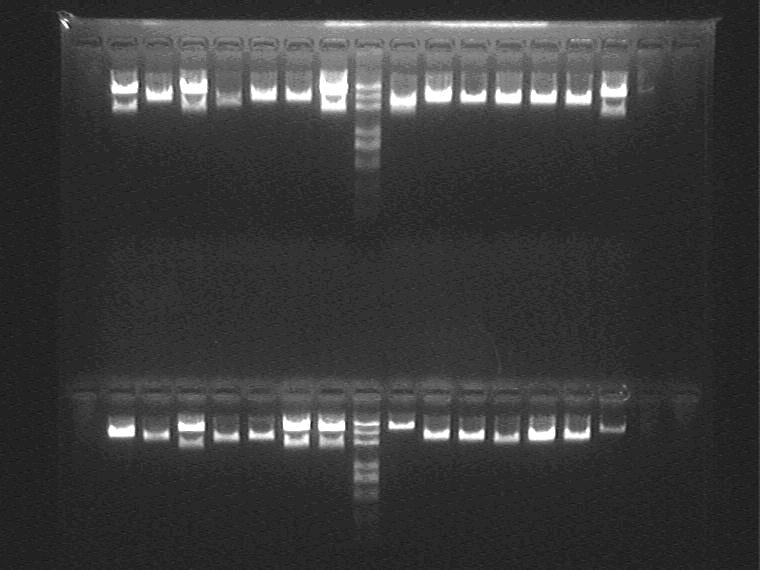
Extract 1H⑤,2G②,2I③,2K②,5J④,5N① from the gel
DNA purification to extract AraC_SupD vector
Ligate the Sal_RBS_T7ptag insert and AraC_SupD vector
System
| 3μL | insert
|
| 1μL | vector
|
| 1μL | 10× Ligase buffer
|
| 1μL | Ligase
|
| 4μL | ddH2O
|
| 10μL | Total
|
2009.9.4
Transformation for the ligation product: Sal_RBS_T7ptag insert + AraC_SupD vector
Choose three other colonies for 5N, 11N, 1J, 2M and shake them
12 samples in total: 5NA\B\C, 11NA\B\C, 1JA\B\C, 2MA\B\C
Miniprep for 12 samples
2009.9.5
Double digestion for Sal_RBS (5N, 11N, 1J, 2M)_T7ptag insert
| 1.5μL | XbaI
|
| 1.5μL | PstI
|
| 2μL | 10×M buffer
|
| 5μL | plasmid
|
| 10μL | ddH2O
|
| 20μL | Total
|
Electrophoretic result (from left to right)
5NA\B, 1JB\C, Marker, 11NA\B, 2MB\C
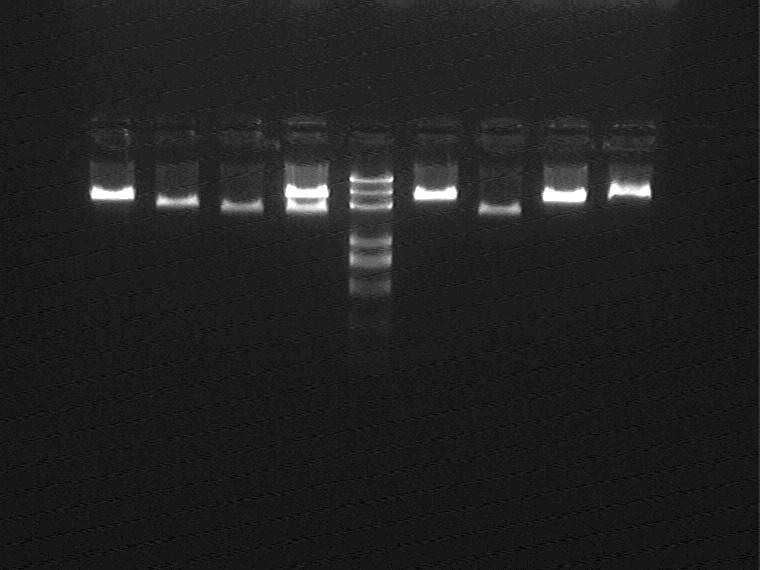
None correct ligation products can be extracted.
Double digestion for Sal_RBS (1H, 2G, 2I, 2K, 5J, 5N)_T7ptag+AraC_SupD insert
| 1.5μL | EcoRI
|
| 1.5μL | PstI
|
| 2μL | 10×H buffer
|
| 4μL | plasmid
|
| 11μL | ddH2O
|
| 20μL | Total
|
Electrophoresis and Gel Extraction for Sal_RBS (1H, 2G, 2I, 2K, 5J, 5N)_T7ptag+AraC_SupD insert
Result (from left to right)
1H①②③, 2I①②③, 2G①, Marker, 2G②③, 5J①②③, 2K①②③
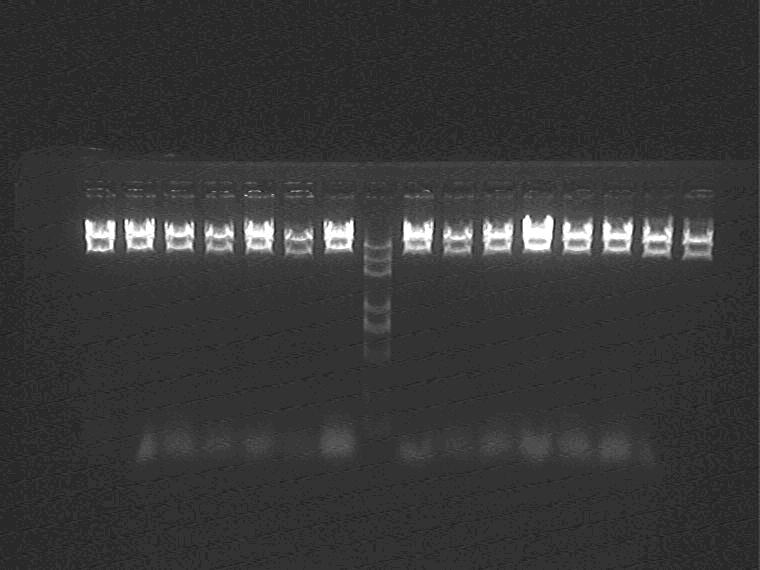
Extract 1H②,2I②,2G①,5J②,2K① from the gel
2009.9.6
Ligate the Sal_T7ptag+AraC_SupD insert(EP) and 1-7G(K+) backbone
System
| 5μL | insert
|
| 1μL | backbone
|
| 1μL | 10× Ligase buffer
|
| 1μL | Ligase
|
| 2μL | ddH2O
|
| 10μL | Total
|
I mistake the plasmid for backbone, so ligate again and transform the ligation product.
I made a great mistake, the length of Sal_RBS_T7ptag+AraC_SupD insert should be a little larger than 5k and the backbone be 3k, which is not in accordance with the results I got yesterday, indicating that the ligation products are abnormal, so I have to rework.
Double digestion for Sal_RBS (1H⑤, 2G②, 2I③, 2K②, 5J④)_T7ptag insert again
| 1.5μL | XbaI
|
| 1.5μL | PstI
|
| 2μL | 3 buffer
|
| 5μL | plasmid
|
| 10μL | ddH2O
|
| 20μL | Total
|
Shift AraC_SupD to low-copy backbone 1-7G
| 1.5μL | EcoRI
|
| 1.5μL | PstI
|
| 2μL | EcoRI buffer
|
| 4μL | plasmid
|
| 11μL | ddH2O
|
| 20μL | Total
|
Electrophoresis and Gel Extraction for Sal_RBS_T7ptag insert
Result (from left to right)
1H, 2G, 2I, 2K, 5J, Marker, AraC_SupD
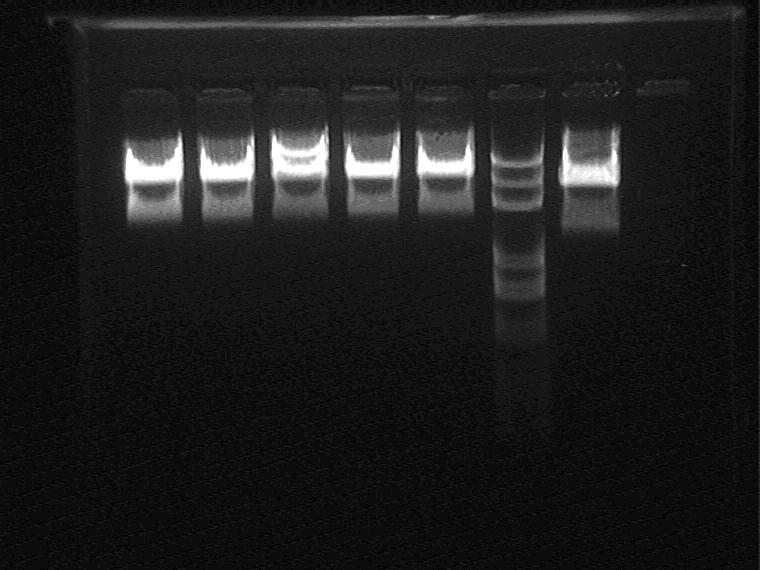
2009.9.8
Miniprep for 1J①②③, 5N①②③, 11N①②③, 2M①②③(Sal_RBS_T7ptag)
Ligate the AraC_SupD insert(EP) and 1-7G(K+) backbone again.
2009.9.9
Double digestion for Sal_RBS (1J, 5N, 11N, 2M)_T7ptag insert
| 1.5μL | XbaI
|
| 1.5μL | PstI
|
| 2μL | 3 buffer
|
| 6μL | plasmid
|
| 9μL | ddH2O
|
| 20μL | Total
|
Electrophoresis and Gel Extraction for Sal_RBS_T7ptag insert
Result (from left to right)
1J①②③, 2M①②③, Marker, 5N①②③, 11N①②③
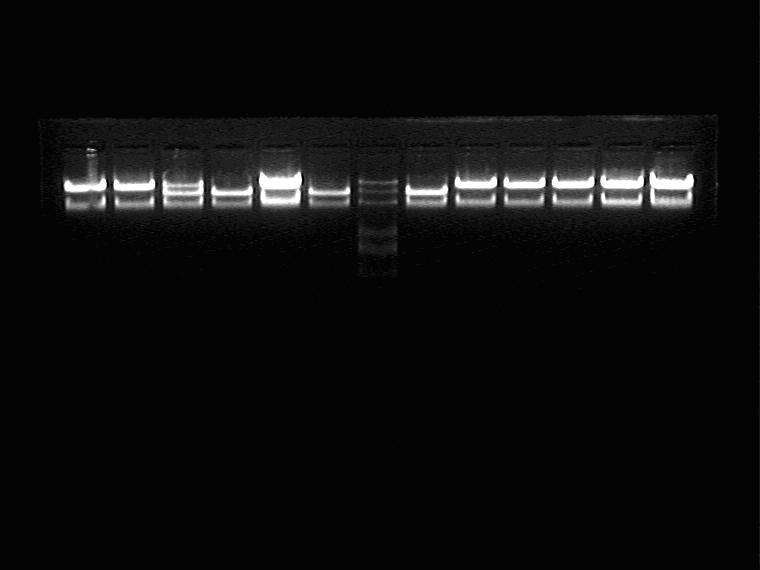
2009.9.10
Electrophoresis and Gel Extraction for Sal_RBS_T7ptag(2G/5J) insert
Result (from left to right)
2G, Marker, 5J
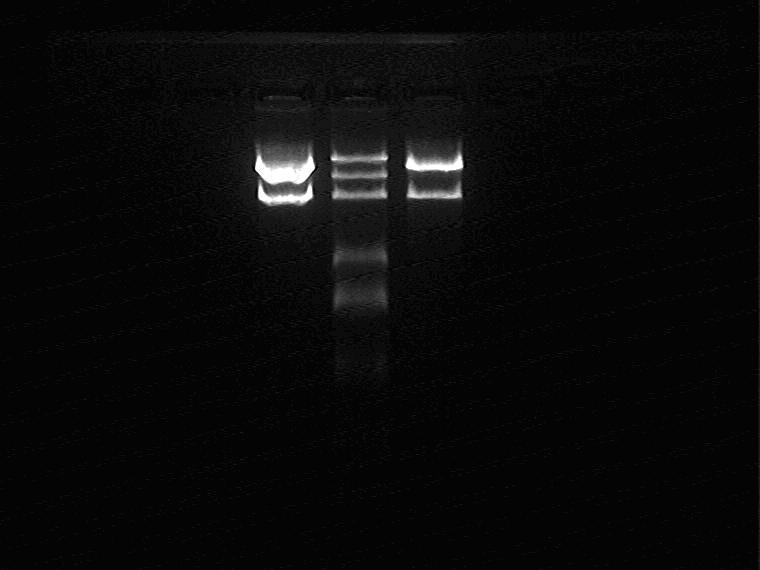
There is something wrong with 2G insert, so only extract 5J inset.
Gel: Sal_5J_T7ptag insert 0.025gPN volume: 75μL
Receive reverse mutated T7ptag bacteria liquid from Lin Min, shake it and store the strain.
Miniprep for T7ptag plasmid
Determine the concentration of T7ptag plasmid by spectrophotometer
4.6422ng/μL×50=232.11ng/μL
^Top
|