Team:Newcastle/IntroductoryLabwork/14 July 2009
From 2009.igem.org
Introductory Lab Session: 14th July 2009
Introduction
Today, DNA gel electrophoresis was carried out on the restriction digests of the plasmid DNA. Based on the observations from the gel electrophoresis, two cultures were then taken and used to innoculate LB broth.
Procedure
- Prepare the agarose gel by melting it in the microwave
- Let the agarose cool down for a while
- While the agarose cools down, prepare the plate to run the gel
- Get the samples containing the DNA treated with restriction enzymes out of the fridge. The samples are as follows:
- First set - treated with EcoRI
- Culture 2 (GFP(BBa_I13522)) - Lane 3
- Culture 3 (RFP(BBa_J04450)) - Lane 4
- Culture 4 (RFP(BBa_J04450)) - Lane 5
- Culture 5 (RFP(BBa_J04450)) - Lane 6
- Culture 6 (RFP(BBa_J04450)) - Lane 7
- Culture 7 (RFP(BBa_J04450)) - Lane 8
- Second set - treated with EcoRI and PstI
- Culture 2 (GFP(BBa_I13522)) - Lane 9
- Culture 3 (RFP(BBa_J04450)) - Lane 10
- Culture 4 (RFP(BBa_J04450)) - Lane 11
- Culture 5 (RFP(BBa_J04450)) - Lane 12
- Culture 6 (RFP(BBa_J04450)) - Lane 13
- Culture 7 (RFP(BBa_J04450)) - Lane 14
- First set - treated with EcoRI
Plasmid with GFP(BBa_I13522) is resistant to AmpR and KanR and the other one with RFP (BBa_J04450) is resistant to AmpR and TetR. Both contain one restriction site for EcoRI and one for PstI.
By just adding EcoRI we expected DNA to be linear whereas in tubes having both restriction enzymes we expected two pieces of DNA.
- Add 1ul of loading buffer to each DNA sample to make the DNA heavier and centrifuge for one minute.
- In the meantime, poor the agarose gel onto the tray, place the comb on the tray, and let the gel solidify.
- After the centrifugation, place the DNA samples on ice
- Prepare the lambda HindIII markers
- Once the gel is ready, transfer the tray to the gel electrophoresis area
- Fill with buffer
- Gently get the comb off the gel
- Start loading the lamba DNA ladder in Lane 1
- Then load Lane 2 to 14
- Finally load Lane 15 with the lamba DNA ladder
- Set the voltage to 120 and run the gel electrophoresis
- After 5mins, drop the voltage to 90V.
Because the DNA is negatively charged, this voltage moved the DNAs on the gel towards the positive side. We then looked at the DNAs on the gel by ultraviolet light.
Results

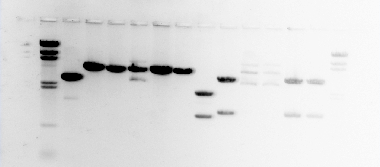
The photographs above show the DNA forming bands within the lanes of the agarose gel.
Next Stage
It was decided that larger plasmid preps should be carried out on cultures 2 and 3 (containing GFP and RFP respectively). The E.coli cells from these cultures were taken from premade agar plates containing all ten cultures and used to innoculate 50ml of LB each. The E.coli within the LB were then grown overnight.
News
Events
- 20 – 21 June 2009 - Europe workshop (London)
- 23 – 24 June 2009 - UK iGEM meetup (Edinburgh)
- 23 October Practice Presentation (Newcastle)
- 23 October T-shirts are ready
- 27 October Practice Presentation (Sunderland)
- 27 October Poster is ready
- 30 October – 2 November 2009 - Jamboree (Boston)
Social Net
- Newcastle iGEM Twitter
- [http://www.facebook.com/home.php#/group.php?gid=131709337641 Newcastle on Facebook]
- [http://www.youtube.com/user/newcastle2009igem Newcastle Youtube Channel]
 "
"




