Team:Groningen/Notebook/16 September 2009
From 2009.igem.org
[http://2009.igem.org/Team:Groningen http://2009.igem.org/wiki/images/f/f1/Igemhomelogo.png]
|
|---|
Wet
GVP Cluster
Planning
- → TODO work out the wiki page for GVP
- → DONE make a doodle for presentation planning (1-19 oct.)
- → TODO media attention
- → DONE place an ethics survey link on twitter
- → TODO clone pArsR-GVP into pSB2K3
- → TODO clone repeat out of GVP cluster
- → TODO make glycerol stocks of constructs
- → TODO enter info on part registry
Plates
| Name | Plasmid Used | Antibiotics on Plasmid | No. of Colonies | Date |
| pArsR-GVP (low) | pSB2K3 | Kanamycin | ~25 | 15 sept. |
| pArsR-GVP (high) | pSB2K3 | Kanamycin | ~120 | 15 sept. |
| Negative (low) | x | x | 0 | 15 sept. |
| Negative (high) | x | x | 0 | 15 sept. |
- → The plates showed a low amount of colonies, and no colonies in the case of the negative plates.
- → The two plates were stored in the fridge for further use.
Cultures
The overnight cultures with LB-amp100 medium of colonies E.coli TOP10 with pLacI-GVP (no.1 and 2), pZntR-GVP (glycerol), pCueO-GVP (glycerol), GlpF (no.1 and 2), and pLacI (pSB1AC3) all showed expected growth of bacteria.
Plasmid isolation
Plasmid isolation was performed on the cultures of E.coli TOP10 containing the above mentioned plasmids with the "Sygma-Aldrich™ [http://www.sigmaaldrich.com/life-science/molecular-biology/dna-and-rna-purification/plasmid-miniprep-kit.html GenElute™] Plasmid Miniprep Kit".
- From each tube 4mL of culture was collected in a 2.0mL cup (tubes from pArsR-GVP, GVP and pNL29 were combined), and the cells were pelleted by centrifugation for 1 min. at max. speed and the supernatant discarded.
- Plasmids were eluted with 30μL MQ and stored in the fridge
Concentrations
| Plasmid | Conc. ng/μL | 260/280 | 260/230 | -20 box (michael | Restriction Control |
| pCueO-GVP (pSB2K3) | 53.3 | 1.90 | 1.88 | x | Yes (EcoRI/PstI) |
| pZntR-GVP (pSB2K3) | 49.8 | 1.96 | 2.16 | x | Yes (EcoRI/PstI) |
| pLacI-GVP (pSB1A2, no.1) | 526.7 | 1.83 | 2.32 | x | Yes (EcoRI/PstI) |
| pLacI-GVP (pSB1A2, no.2) | 493.3 | 1.83 | 2.34 | x | Yes (EcoRI/PstI) |
| GlpF (pSB1AC3, no.1) | 118.7 | 1.84 | 1.57 | x | Yes (EcoRI/PstI) |
| GlpF (pSB1AC3, no.2) | 96.7 | 1.92 | 2.03 | x | Yes (EcoRI/PstI) |
| pLacI (pSB1AC3) | 158.3 | 1.90 | 2.24 | x | Yes (EcoRI/PstI) |
Restriction for Control
The plasmids from the o.n. precultures of pArsR-GVP and pSB2K3 (earlier last week) were cut with PstI and EcoRI to cut out the entire part between the pre- and suffix. The plasmids with PstI were cut with PstI for control.
| Plasmid | Amount μL | MQ μL | Fast digest buffer | EcoRI fast digest enzyme | XbaI fast digest enzyme | SpeI fast digest enzyme | PstI fast digest enzyme |
| pCueO-GVP | 16.0 | x | 3.0 | 1.0 | x | x | 1.0 |
| pZntR-GVP | 16.0 | x | 3.0 | 1.0 | x | x | 1.0 |
| pLacI-GVP no.1 | 2.0 | 14.0 | 3.0 | 1.0 | x | x | 1.0 |
| pLacI-GVP no.2 | 2.0 | 14.0 | 3.0 | 1.0 | x | x | 1.0 |
| GlpF no.1 | 10.0 | 6.0 | 3.0 | 1.0 | x | x | 1.0 |
| GlpF no.2 | 10.0 | 6.0 | 3.0 | 1.0 | x | x | 1.0 |
| pLacI (pSB1AC3) | 8.0 | 8.0 | 3.0 | 1.0 | x | x | 1.0 |
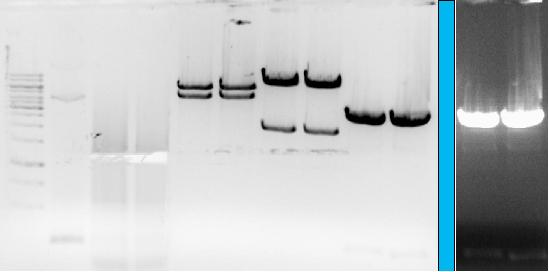
- → From left to right: 1kB ladder, three samples of SJ, pCueO-GVP (pSB2K3), pZntR-GVP (pSB2K3), pLacI-GVP (pSB1A2, no.1), pLacI-GVP (pSB1A2, no.2), GlpF (pSB1AC3), pLacI (pSB1AC3)
- → The additional two lanes are a darker image of the GlpF (pSB1AC3), pLacI (pSB1AC3) lanes, to check for GlpF and pLacI insert.
Transporters
Metal Accumulation
Vectors
Dry
|
|
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 "
"