Team:Groningen/Project/Transport
From 2009.igem.org
[http://2009.igem.org/Team:Groningen http://2009.igem.org/wiki/images/f/f1/Igemhomelogo.png]
|
|---|
Introduction
We are trying to find suitable systems capable of isolating heavy metals from the environment. There are several different mechanisms to achieve such a thing. We examined 3 kinds:
- Metal transporters, that transport the metal from the environment (ie. wastewater) to the cytoplasm
- Uncoupled
- Coupled with 'helper' compounds
- Metal binding proteins in the periplasm
We will investigate severals systems, to find which are suitable for the final design. the following systems are under concideration.
- Copper/zinc uptake via HmtA
- heavy metal uptake coupled to citrate via efCitH bsCitM
- Arsenite uptake via GlpF
- Periplasmic accumulation of heavy metals via Mer Operon.
HmtA
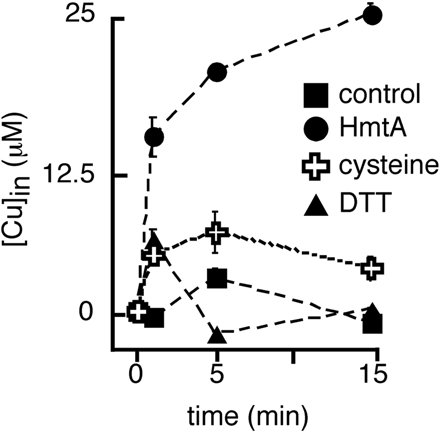
HmtA, heavy metal transporter A from Pseudomonas aeruginosa Q9I147 is a P-type ATPase importer. It mediates the uptake of Copper (Cu) and Zinc (Zn) and is functionally expressed in E.coli.
>gi|81857196|sp|Q9I147|Q9I147_PSEAE Probable cation-transporting P-type ATPase
| Enzyme | Number of Sites |
|---|---|
| EcoRI | 0 |
| XbaI | 0 |
| NotI | 0 |
| SpeI | 0 |
| PstI | 2 |
Missing information/To do
- Expression assesment
- Stability
- Level
- Functional assesment
- Uptake speed
- Affinity
- Electrolyte potential generating force
- Eliminate BioBrick restriction sites
Literature
Lewinson O., Lee A.T., Rees D.C. 2009. A P-type ATPase importer that discriminates between essential and toxic transition metals. PNAS. vol. 106, no. 12, p. 4677-4682.
Citrate coupled uptake
Citrate uptake coupled to heavy metals enables forcefeeding of the toxic compounds into the cell when citrate is the only carbound source available. This could be a very effecient strategy to accumelate vass ammounts of heavy metals. The current candidates are CitM from Bacilus subtilis and CitH form Enterococcus faecalis
Missing information/To Do
- Expression assesment
- Stability
- Level
- Functional assesment
- Uptake speed
- Affinity
- Electrolyte potential generating force
- Eliminate BioBrick restriction sites
Literature
Blancato, V.S., Magni, C. & Lolkema, J.S. Functional characterization and Me2+ ion specificity of a Ca2+–citrate transporter from Enterococcus faecalis. FEBS Journal 273, 5121-5130(2006). Bastiaan krom Citrate transporters of Bacilus subtilis Proefschrift. Jessica B. Warner Regulation and expression of the metal citrate transporter CitM Proefschrift.
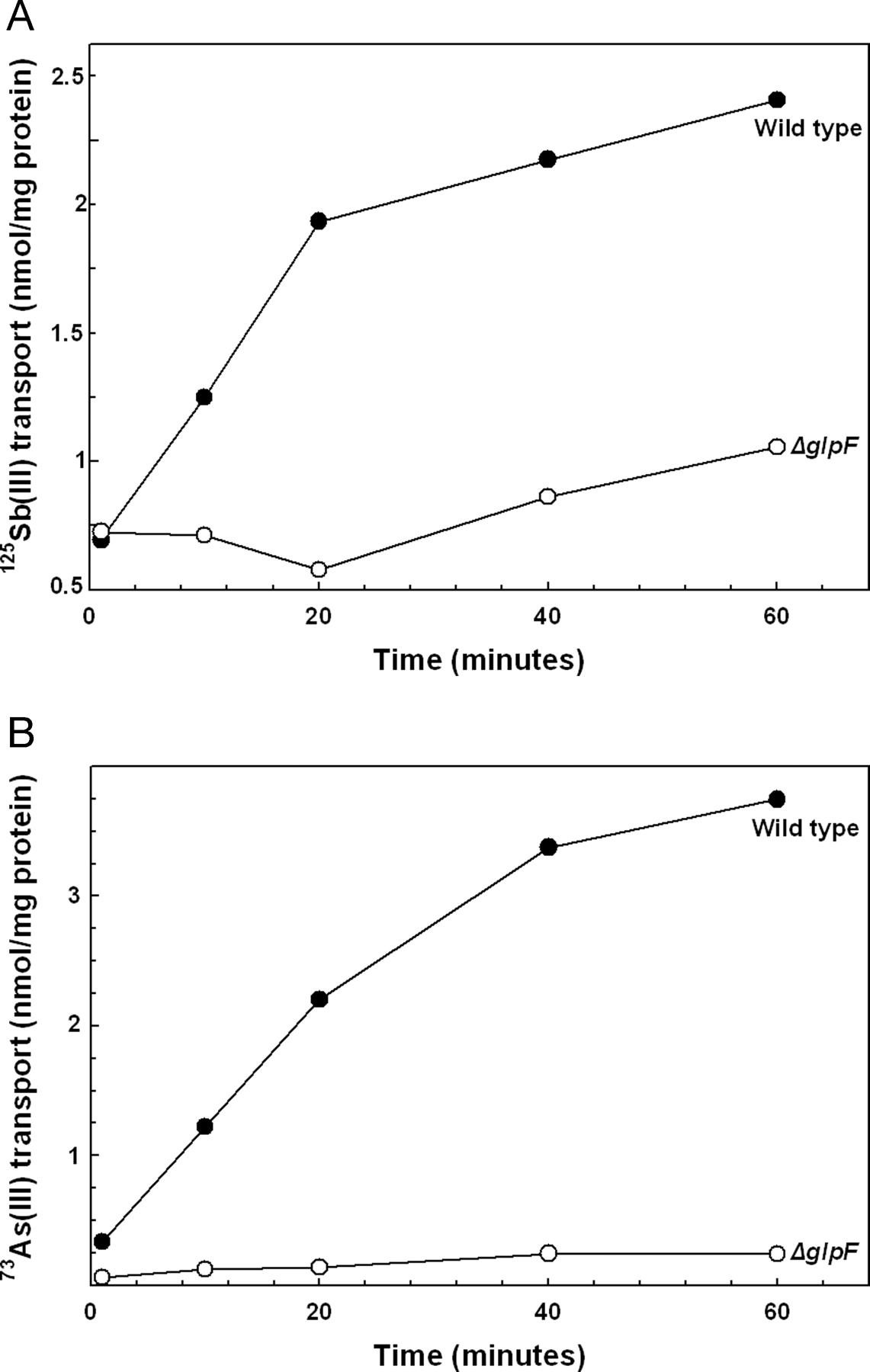
GlpF
Missing information/To Do
- Expression assesment
- Stability
- Level
- Functional assesment
- Uptake speed
- Affinity
- Electrolyte potential generating force
- Eliminate BioBrick restriction sites
Literature
1. Meng, Y., Liu, Z. & Rosen, B.P. As(III) and Sb(III) Uptake by GlpF and Efflux by ArsB in Escherichia coli. J. Biol. Chem. 279, 18334-18341(2004).
MerR family
Periplasmic accumulation of heavy metals via Mer proteins enables the harvesting of heavy metals from the medium. By binding the cytosolic and periplasmic metals to metallothionein and transporting the metal-protein complex into the periplasm. The MerR family consists of different proteins for one specific metal. (ie. PbrR(Lead), CueR(Copper), ZntR(Zinc), MerR(Mercury), ArsR(Arsenic), CadR(Cadmium))
Missing information/To Do
- Expression assesment
- Stability
- Level
- Functional assesment
- Uptake speed
- Affinity
- Electrolyte potential generating force
- Eliminate BioBrick restriction sites
Literature
Kao, W., Huang, C. & Chang, J. Biosorption of nickel, chromium and zinc by MerP-expressing recombinant Escherichia coli. J. Hazard. Mater 158, 100-106(2008).
Pennella, M.A. & Giedroc, D.P. Structural determinants of metal selectivity in prokaryotic metal-responsive transcriptional regulators. Biometals 18, 413-428(2005). "
"