Team:UNIPV-Pavia/Notebook/Week5Jul
From 2009.igem.org

|
|
|
|
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
Week from July 27th, to August 1st, 2009
 Previous Week
Previous Week
|
Next Week

|
July, 27th
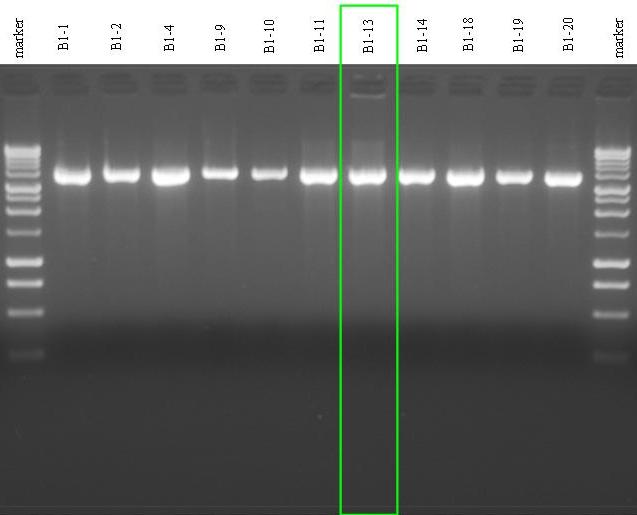
- Screening for the 11 miniprepped DNA samples for B1: digestion S-P for all.
- Gel results:
- Samples 1, 4, 10, 11, 14, 19 and 20 showed an extra band for the non ligated plasmid;
- Samples 2, 9, 13 and 18 were pure! we decided to keep B1-13 (lane 8) to perform future ligations.
- NOTE: we had a pure sample for B1 (i.e. B1-13)and three almost-pure sample for B2. Anyway, we decided to perform ligation reactions for these samples and the extra band of B2 will be eliminated during gel cut/purification. WE DECIDED TO KEEP B2-5. Next weeks we will think about purifying B2-5 itself.
- We transformed 20 pg of B1-13 purified DNA (stored at -20°C) in TOP10 in order to prepare a glycerol stock for this construct. We incubated the plate at 37°C overnight.
Preparation of experiment with Tecan F200
- We infected 5 ml of LB + Amp with 10 ul of A14pg, A8pg and A9pg glycerol stocks.
- We also infected 5 ml of LB + Amp with a single colony taken from B0030 native plate (stored at +4°C).
- We incubated the inocula at 37°C, 220 rpm overnight.
July, 28th
- We streaked LB agar plates + suitable antibiotic with iGEM stabs:
| K116001 | K116002 | K112405 |
| P0412 | I746902 | I746903 |
| K101017 | F2620MIT1 | F2620MIT2 |
- We incubated these "single colonies" plates at 37°C overnight.
- We picked a single colony from B1-13 plate to infect 1 ml of LB + Amp and incubated this inoculum for 5 hours and 1/2.
- We prepared a glycerol stock for B1-13.
- We aliquoted the remaining 250 ul of B1-13 bacterial culture in two different falcon tubes and re-filled them with 5 ml of LB + Amp.
- We also infected 5 ml of LB + Amp with 10 ul of B0015(X2) and B2-5(X2) glycerol stocks.
- We incubated these six cultures at 37°C, 220 rpm overnight.
- We received sequencing results for:
- A12-2: sequence ok!
- A12-3: sequence ok!
Preparation of experiment with Tecan F200
- We diluted 1:1000 the overnight cultures of A14pg, A8pg, A9pg and B0030.
- We incubated the diluted cultures for 5 hours (37°C, 220 rpm).
- After 5 hours, we adjusted the OD600 at 0.025.
Experiment with Tecan F200
- Description
- Purpose:
- Materials & Methods
- Protocol
- Results
July, 29th
- Miniprep for:
- B1-13
- B1-13bis
- B2-5
- B2-5bis
- B0015
- B0015bis
- Digestion:
- B1-13(E-S)
- B1-13bis(E-S)
- B2-5(E-S)
- B2-5bis(E-S)
- B0015(E-X)
- B0015bis(E-X) - 500ng
- Gel run/cul/purification for:
- B1-13(E-S) - ONLY FOR CHECK, NOT PURIFIED
- B1-13bis(E-S) - ONLY FOR CHECK, NOT PURIFIED
- B2-5(E-S)
- B2-5bis(E-S)
- B0015(E-X)
- B0015bis(E-X)
- Precipitation with sodium acetate for:
- B1-13(E-S)
- B1-13bis(E-S)
- We had good yields for all, except from B0015bis(E-X). We will use the other vector (i.e. B0015(E-X)) for ligation.
- Ligation:
- B3 = B1(E-S) + B0015(E-X) in pSB1AK3 (50 ng of vector)
- B4 = B2(E-S) + B0015(E-X) in pSB1AK3 (26 ng of vector)
- keeping the sample that gave the higher yield.
- We incubated the ligations at 16°C overnight.
- All the streaked plates showed single colonies! we prepared an inoculum for all of them picking a single colony and infecting 1 ml of LB + suitable antibiotic. We incubated these inocula for 5 hours and 1/2.
- Then, we prepared a glycerol stock for each of them and re-filled the remaining 250 ul of bacterial culture with 3 ml of LB + suitable antibiotic.
- We incubated the cultures at 37°C, 220 rpm overnight.
July, 30th
- We transformed B3 (50 pg) and B4 (20 pg) ligations. We incubated the plates at 37°C overnight.
- Miniprep for the overnight cultures of:
| K116001 | K116002 | K112405 |
| P0412 | I746902 | I746903 |
| K101017 | F2620MIT1 | F2620MIT2 |
- We sent purified DNA to BMR Genomics for sequencing (VF2 and VR for all of them, except from K112405, whose plasmid does not have VF2 annealing site).
- We also sent purified DNA of B1-13 and B2-5 (stored at -20°C) to BMR Genomics for sequencing.
- We received sequencing results for A11: lacI sequence showed a deletion...we will repeat A11 ligation from BOL1 and R0011 parts.
Experiment with Tecan F200
- Description
- Purpose:
- Materials & Methods
- Protocol
- Results
Preparation of experiment with Tecan F200 (for the following day!)
- We inoculated 10 ul of J23100 and J23100-E0240 parts form UNIPV iGEM 2008 stocks.
- We incubated these two inocula at 37°C, 220 rpm overnight.
July, 31st
- B3 and B4 plates showed colonies! We picked 7 colonies for each plate and infected 1 ml of LB + Kan. We incubated these 14 inocula for 5 hours and 1/2. Then we prepared glycerol stocks and re-filled the remaining 250 ul of bacterial culture with 5 ml of LB + Kan.
- We incubated these cultures at 37°C, 220 rpm overnight.
Preparation of experiment with Tecan F200
- We diluted 1:100 J23100 and J23100-E0240 parts form UNIPV iGEM 2008 stocks overnight cultures.
- We incubated these two cultures at 37°C, 220 rpm for about 3 hours.
Experiment with Tecan F200
- Description
- Purpose:
- Materials & Methods
- Protocol
- Results
August, 1st
- Miniprep for the 7 colonies of B3 and for the 7 colonies of B4. We stored purified DNA at -20°C and next Monday we will perform screening for these 14 samples.
 Previous Week
Previous Week
|
Next Week

|
 "
"